Revolutionizing GC-24 and Furegrelate Production with Sustainable Nickel Catalysis Technology
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is detailed in Chinese Patent CN111646889B, which discloses a novel green synthesis method for two critical drug active molecules: GC-24, a thyroid hormone receptor β-selective analog, and Furegrelate, a highly selective thromboxane synthase inhibitor. This patent represents a paradigm shift from traditional multi-step syntheses reliant on hazardous reagents to a streamlined, nickel-catalyzed reductive coupling strategy. By utilizing cheap and readily available benzyl alcohols and aryl electrophiles activated by dimethyl oxalate, this technology constructs the key diarylmethane framework in a single step. For R&D directors and process chemists, this innovation offers a compelling alternative to legacy methods, promising not only improved step economy but also a drastically simplified purification profile and enhanced safety standards during manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of GC-24 has been plagued by inefficiency and safety hazards, typically requiring a cumbersome 10-step sequence that involves multiple protection and deprotection cycles. As illustrated in prior art such as ACS Catal. 2017, 7, 1108, the conventional route necessitates the preparation of specialized benzyl reagents and relies heavily on the use of highly corrosive and volatile chemicals like hydrogen bromide (HBr) and boron trichloride (BCl3). Furthermore, these legacy processes often depend on expensive palladium metal catalysts and sophisticated carbene ligands, which drive up the cost of goods sold (COGS) and complicate the removal of heavy metal residues to meet stringent regulatory limits. Similarly, the traditional manufacturing of Furegrelate, based on techniques dating back over 30 years (J. Med. Chem. 1986, 29, 1461), involves a perilous 6-step pathway. This outdated method utilizes strong corrosive reagents such as nitric acid, sulfuric acid, and trifluoroacetic acid, and critically, includes flammable and explosive processes like catalytic hydrogenation and diazotization. The initial nitration step in particular suffers from poor selectivity and low yields (around 22%), creating significant bottlenecks in production capacity and waste management.
The Novel Approach
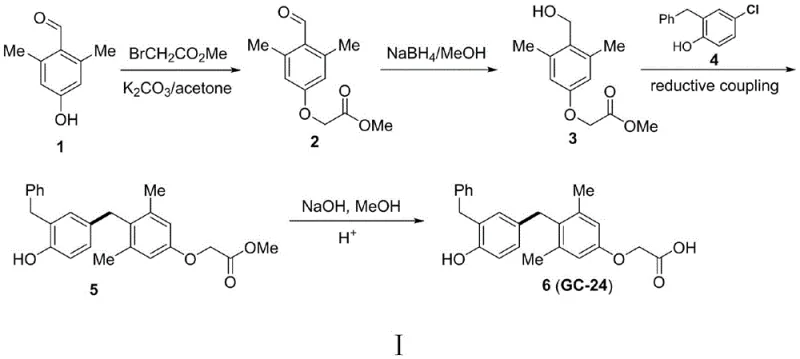
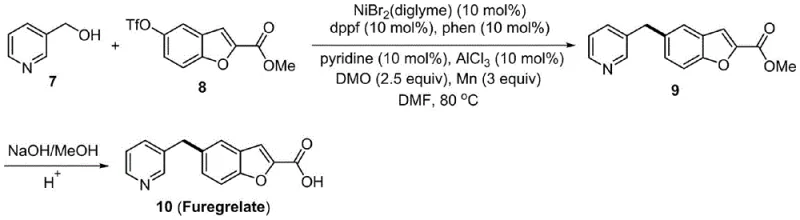
In stark contrast, the methodology disclosed in patent CN111646889B introduces a robust and atom-economical solution that fundamentally redesigns the synthetic landscape for these molecules. For GC-24, the new protocol condenses the synthesis from 10 steps down to merely 4 steps, while for Furegrelate, the route is shortened by half. The core innovation lies in the use of a nickel-catalyzed reductive coupling strategy, where dimethyl oxalate serves as an effective activator to enable the direct coupling of benzyl alcohols with aryl electrophiles. This approach completely bypasses the need for hazardous halogenating agents and strong acids, replacing them with milder conditions and earth-abundant nickel catalysts. The operational simplicity is remarkable; reactions proceed smoothly in polar aprotic solvents like DMF at moderate temperatures (80°C to 100°C), yielding the target diarylmethane skeletons with high efficiency. This transition not only enhances the overall yield but also aligns perfectly with modern green chemistry principles, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming for sustainability.
Mechanistic Insights into Nickel-Catalyzed Reductive Coupling
The success of this green synthesis hinges on the intricate mechanistic interplay between the nickel catalyst, the activator, and the reductant. The reaction initiates with the activation of the benzyl alcohol C-O bond, a traditionally difficult transformation, facilitated by the presence of dimethyl oxalate and the nickel complex. Under the reaction conditions, typically employing a ligand system such as 4,5-diazafuoren-9-one or dppf, the nickel center undergoes oxidative addition into the activated C-O bond of the benzyl alcohol derivative. Simultaneously, the aryl electrophile (such as 4-chloro-2-benzylphenol for GC-24 or a benzofuran triflate for Furegrelate) interacts with the catalytic cycle. Manganese powder acts as the terminal reductant, regenerating the active low-valent nickel species necessary to sustain the catalytic turnover. This reductive cross-coupling mechanism effectively merges two distinct fragments into the desired diarylmethane structure without the need for pre-functionalization into halides or triflates for the alcohol component, which is a major departure from classical cross-coupling paradigms. The choice of ligands and the specific oxidation state management of the nickel center are critical for suppressing homocoupling side reactions, ensuring that the heterocoupled product is formed with high selectivity.
From an impurity control perspective, this mechanism offers distinct advantages over acid-mediated Friedel-Crafts type alkylations or harsh halogenation sequences. The mild reaction environment (80°C in DMF) prevents the degradation of sensitive functional groups that might occur under the strongly acidic conditions of the old routes (e.g., H2SO4/HNO3 mixtures). Furthermore, the absence of free radical halogenation steps minimizes the formation of poly-halogenated byproducts, which are notoriously difficult to separate and can pose genotoxicity risks. The use of dimethyl oxalate as an activator generates benign byproducts upon workup, simplifying the downstream purification process. For quality control teams, this translates to a cleaner crude reaction profile, reducing the burden on chromatographic purification and crystallization steps. The ability to achieve high purity specifications directly from the reactor output is a key factor in reducing the overall manufacturing lead time and ensuring batch-to-batch consistency, which is paramount for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize GC-24 and Furegrelate Efficiently
The practical implementation of this synthesis involves a straightforward three-stage protocol that is amenable to both laboratory scale and pilot plant operations. The process begins with the preparation of the benzyl alcohol intermediate via simple alkylation and reduction, followed by the pivotal nickel-catalyzed coupling step, and concludes with a standard ester hydrolysis. Detailed standardized operating procedures for each stage, including precise stoichiometric ratios, temperature ramps, and workup protocols, are essential for reproducibility. The following guide outlines the critical operational parameters derived from the patent examples to ensure optimal yield and purity.
- Prepare the benzyl alcohol intermediate by alkylating 2,6-dimethyl-4-hydroxybenzaldehyde with methyl bromoacetate followed by reduction with sodium borohydride.
- Perform the key nickel-catalyzed reductive coupling between the benzyl alcohol intermediate and the aryl electrophile (4-chloro-2-benzylphenol or benzofuran triflate) using dimethyl oxalate and manganese powder in DMF at 80-100°C.
- Hydrolyze the resulting ester intermediate using sodium hydroxide in methanol, followed by acidification to isolate the final carboxylic acid product GC-24 or Furegrelate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed technology presents a multitude of strategic benefits that extend far beyond simple chemical curiosity. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the dependency on specialized, high-cost reagents like palladium catalysts, carbene ligands, and corrosive gases (HBr, BCl3), manufacturers can source inputs from a broader, more stable vendor base. This diversification mitigates the risk of supply disruptions caused by the limited availability of precious metals or regulated hazardous chemicals. Moreover, the reduction in synthetic steps from 10 to 4 for GC-24 inherently reduces the cumulative loss of material at each stage, leading to a substantial increase in overall throughput without the need for additional capital investment in reactor volume. This step economy directly correlates to lower operational expenditures (OPEX) and a smaller physical footprint for production facilities.
- Cost Reduction in Manufacturing: The economic impact of switching to this green method is profound, driven primarily by the replacement of expensive noble metals with earth-abundant nickel. Palladium catalysts and their associated ligands represent a significant portion of the material cost in traditional cross-coupling reactions; substituting these with nickel complexes and inexpensive activators like dimethyl oxalate results in significant cost savings. Additionally, the avoidance of harsh reagents reduces the costs associated with specialized corrosion-resistant equipment (such as Hastelloy reactors) and the extensive neutralization and disposal of hazardous acidic waste streams. The higher overall yield achieved through fewer steps means less raw material is wasted, further driving down the cost per kilogram of the final API intermediate. These factors combine to create a much more competitive cost structure for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commodity chemicals rather than bespoke or regulated substances. Reagents like manganese powder, dimethyl oxalate, and standard nickel salts are widely available globally, reducing lead times for high-purity pharmaceutical intermediates. In contrast, the old routes relied on reagents like boron trichloride and concentrated nitric acid, which are subject to strict transportation regulations and storage requirements due to their corrosive and toxic nature. By removing these logistical bottlenecks, manufacturers can maintain more consistent inventory levels and respond more agilely to fluctuations in market demand. The simplified process also reduces the complexity of the manufacturing schedule, allowing for faster turnaround times between batches and improving the reliability of delivery commitments to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the mild reaction conditions and the absence of exothermic hazards associated with nitration or diazotization. The nickel-catalyzed coupling proceeds at moderate temperatures (80-100°C) in standard solvents, making it compatible with existing stainless steel infrastructure without the need for exotic materials. From an environmental standpoint, the process aligns with increasingly stringent global regulations regarding waste discharge and worker safety. Eliminating the generation of heavy metal waste (Pd) and acidic effluents simplifies the permitting process for new production lines and reduces the liability associated with environmental compliance. This green credential is becoming a decisive factor for multinational corporations seeking sustainable partners, thereby enhancing the marketability of the produced intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the feasibility and advantages of the new route. Understanding these details is crucial for technical teams evaluating the transfer of this technology to commercial production scales.
Q: How does the new nickel-catalyzed method improve upon traditional GC-24 synthesis?
A: The new method reduces the synthetic route from 10 steps to just 4 steps, eliminating the need for highly corrosive reagents like HBr and BCl3, as well as expensive palladium catalysts and carbene ligands used in conventional processes.
Q: What safety advantages does the new Furegrelate synthesis offer?
A: The novel route avoids dangerous nitration reactions involving concentrated nitric and sulfuric acids, and eliminates explosive diazotization and catalytic hydrogenation steps, significantly enhancing operational safety and environmental compliance.
Q: What is the role of dimethyl oxalate in this green synthesis protocol?
A: Dimethyl oxalate acts as a crucial activator in the nickel-catalyzed reductive coupling, facilitating the cleavage of the C-O bond in cheap and readily available benzyl alcohol starting materials to construct the key diarylmethane skeleton efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable GC-24 and Furegrelate Supplier
The technological advancements described in patent CN111646889B underscore the immense potential for optimizing the production of critical pharmaceutical intermediates like GC-24 and Furegrelate. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of such chemical innovations, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex nickel-catalyzed reactions with precision, ensuring that every batch meets stringent purity specifications required by global regulatory bodies. With our rigorous QC labs and commitment to green chemistry, we are uniquely positioned to deliver high-quality intermediates that support your drug development timelines while adhering to the highest standards of safety and sustainability.
We invite you to explore how our advanced manufacturing capabilities can enhance your supply chain efficiency and reduce your overall project costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for GC-24 and Furegrelate. Let us partner with you to bring these vital therapeutic agents to market faster, safer, and more economically.
