Advanced Manufacturing of 6-Benzothiazole Sulfonyl Chloride for High-Purity HIV Inhibitor Intermediates
Advanced Manufacturing of 6-Benzothiazole Sulfonyl Chloride for High-Purity HIV Inhibitor Intermediates
The pharmaceutical industry's relentless pursuit of effective antiretroviral therapies has placed significant demand on the supply chain for high-quality benzothiazole derivatives. Specifically, 6-benzothiazole sulfonyl chloride serves as a pivotal building block in the synthesis of compounds exhibiting potent activity against the human immunodeficiency virus (HIV). A groundbreaking technical disclosure, detailed in patent CN102108069A, introduces a revolutionary preparation method that addresses long-standing inefficiencies in traditional manufacturing. This novel approach leverages a strategic sequence of catalytic hydrogenation and diazotization-chlorosulfonylation to deliver a product with exceptional purity and structural integrity. By shifting away from corrosive halogenation cycles toward a more controlled, gas-phase functionalization strategy, this technology offers a robust pathway for producing reliable pharmaceutical intermediates. For global procurement teams and R&D directors, understanding the mechanistic nuances of this patent is critical for securing a stable supply of these vital antiviral precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-benzothiazole sulfonyl chloride has been plagued by significant operational hazards and environmental burdens, as exemplified by earlier methodologies such as those disclosed in U.S. Patent 6,140,505. The traditional route typically initiates with sulfanilamide and ammonium thiocyanate, proceeding through a thiourea intermediate that requires cyclization using elemental bromine. This reliance on bromine introduces severe challenges, including intense corrosion of reaction vessels and the generation of hazardous waste streams that complicate disposal and increase operational costs. Furthermore, the subsequent steps often involve diazotization with unstable reagents like isopentyl nitrite, leading to poor batch-to-batch repeatability and inconsistent yields. The cumulative effect of these harsh conditions is a manufacturing process that is not only environmentally unfriendly but also economically inefficient due to high maintenance requirements for corrosion-resistant equipment and extensive waste treatment protocols.
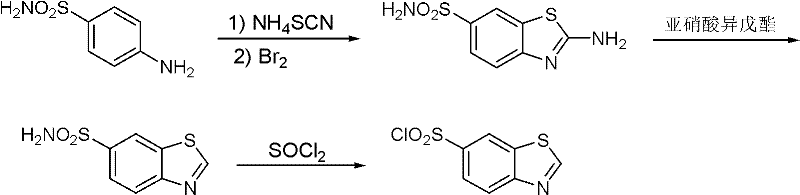
The Novel Approach
In stark contrast, the methodology outlined in patent CN102108069A presents a streamlined, three-step synthetic strategy that fundamentally redesigns the construction of the benzothiazole scaffold. Instead of building the ring under oxidative stress, this new approach begins with a pre-formed nitro-substituted benzothiazole skeleton (Compound III). The process employs a mild catalytic hydrogenation to reduce the nitro group to an amine, followed by a precise diazotization and a copper-catalyzed reaction with sulfur dioxide and chlorine. This shift eliminates the need for elemental bromine entirely, thereby removing the primary source of equipment corrosion and toxic byproduct formation. The reaction conditions are notably gentle, with the reduction step occurring at moderate temperatures around 50°C and pressures of 0.5MPa, ensuring safety and scalability. This modernization of the synthetic route represents a significant leap forward in cost reduction in pharmaceutical intermediates manufacturing, offering a cleaner, safer, and more reproducible alternative for industrial applications.
Mechanistic Insights into Catalytic Hydrogenation and Chlorosulfonylation
The core innovation of this process lies in the efficient transformation of the nitro group and the subsequent installation of the sulfonyl chloride moiety. The first critical stage involves the reduction of 6-nitrobenzothiazole (Compound III) to 6-aminobenzothiazole (Compound II). This transformation is achieved using a heterogeneous catalyst, specifically palladium on carbon (Pd/C), suspended in a polar protic solvent such as methanol. The use of molecular hydrogen at a controlled pressure of 0.1 to 1MPa ensures a highly selective reduction that preserves the sensitive benzothiazole ring structure. Unlike chemical reductions that might generate stoichiometric amounts of metal waste, this catalytic cycle is atom-economical and generates water as the only byproduct. The mild thermal conditions, typically maintained between 20°C and the reflux temperature of the solvent, prevent thermal degradation of the heterocyclic core, resulting in a crude product with high HPLC purity prior to any extensive workup.

Following the reduction, the generated amine undergoes diazotization to form the reactive diazonium salt (Compound V), which serves as the precursor for the final functionalization. The subsequent step is a sophisticated variation of the Meerwein reaction, where the diazonium salt reacts with sulfur dioxide and chlorine gas in the presence of a copper sulfate catalyst. This step effectively replaces the diazo group with a sulfonyl chloride functionality. The presence of copper ions facilitates the single-electron transfer processes necessary for the radical mechanism, allowing the insertion of the sulfur and chlorine atoms with high regioselectivity. Conducting this reaction in an organic solvent like toluene at temperatures between 45°C and 50°C optimizes the solubility of the gaseous reagents while maintaining control over the exothermic nature of the chlorosulfonylation. This mechanistic precision ensures that the final high-purity pharmaceutical intermediate is obtained with minimal formation of desulfonylated or polysulfonylated impurities, a common issue in less controlled sulfonation processes.
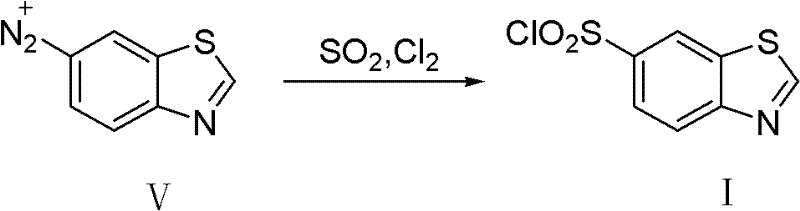
How to Synthesize 6-Benzothiazole Sulfonyl Chloride Efficiently
The practical implementation of this synthesis requires careful attention to reaction parameters to maximize yield and safety. The process begins with the dissolution of the nitro-starting material in a lower alcohol, followed by the introduction of hydrogen gas over the palladium catalyst until uptake ceases, indicating complete reduction. The resulting amine is then isolated and immediately subjected to diazotization conditions using sodium nitrite and a mineral acid at low temperatures to stabilize the diazonium intermediate. Finally, the introduction of sulfur dioxide and chlorine gases into the reaction mixture containing the copper catalyst must be managed with appropriate gas scrubbing systems to ensure operator safety and environmental compliance. For a detailed breakdown of the specific molar ratios, solvent volumes, and temperature profiles required for each stage, please refer to the standardized protocol below.
- Perform catalytic hydrogenation of 6-nitrobenzothiazole (Compound III) using Pd/C in methanol at 50°C and 0.5MPa to obtain 6-aminobenzothiazole.
- Conduct diazotization of the resulting amine with sodium nitrite and mineral acid at low temperatures (e.g., 10°C) to form the diazonium salt.
- React the diazonium salt with sulfur dioxide and chlorine gas in the presence of copper sulfate catalyst in toluene to yield the final sulfonyl chloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN102108069A offers substantial strategic benefits beyond mere technical feasibility. The elimination of corrosive reagents like bromine directly translates to extended equipment lifespan and reduced maintenance downtime, which are critical factors in maintaining consistent production schedules. Furthermore, the use of commodity chemicals such as hydrogen, sulfur dioxide, and chlorine, coupled with widely available catalysts like Pd/C, mitigates the risk of raw material shortages that often plague specialized synthetic routes. This robustness in the supply chain ensures that manufacturers can meet the rigorous delivery timelines demanded by the global pharmaceutical market without compromising on quality or regulatory standards.
- Cost Reduction in Manufacturing: The new process significantly lowers operational expenditures by removing the need for expensive corrosion-resistant reactors and complex waste neutralization systems associated with bromine handling. The catalytic nature of the hydrogenation step reduces reagent consumption compared to stoichiometric reductions, while the high selectivity of the final chlorosulfonylation step minimizes the loss of valuable intermediates to side reactions. These efficiencies collectively drive down the cost of goods sold (COGS), making the final API intermediate more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: By relying on a synthetic route that utilizes stable, commercially abundant starting materials, manufacturers can insulate their production lines from the volatility of niche reagent markets. The mild reaction conditions also reduce the energy intensity of the process, lowering utility costs and decreasing the carbon footprint of the manufacturing facility. This alignment with green chemistry principles not only improves corporate sustainability metrics but also future-proofs the supply chain against increasingly stringent environmental regulations.
- Scalability and Environmental Compliance: The simplicity of the workup procedures, which involve standard filtration, extraction, and crystallization techniques, facilitates seamless scale-up from pilot plant to commercial tonnage. The absence of heavy metal contaminants and toxic halogenated byproducts simplifies the effluent treatment process, ensuring compliance with international environmental discharge standards. This ease of scalability allows for rapid response to surges in market demand for HIV therapeutics, ensuring a continuous and reliable flow of critical medical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 6-benzothiazole sulfonyl chloride using this advanced methodology. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on the process's viability and performance characteristics.
Q: What are the primary advantages of this new synthesis method over conventional routes?
A: The new method eliminates the use of corrosive bromine and harsh cyclization conditions found in older patents. It utilizes mild catalytic hydrogenation and readily available gases (SO2, Cl2), significantly reducing equipment corrosion and environmental pollution while improving repeatability.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights suitability for industrial scale-up. The reaction conditions are gentle (e.g., 50°C for reduction), and the raw materials are cheap and commercially available, avoiding complex purification steps associated with heavy metal byproducts.
Q: What is the purity profile of the final 6-benzothiazole sulfonyl chloride?
A: Experimental embodiments in the patent demonstrate HPLC purity levels reaching 98%. The use of activated carbon decolorization and standard organic extraction ensures a high-purity profile suitable for downstream pharmaceutical synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Benzothiazole Sulfonyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antiviral medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative methods described in patent CN102108069A can be seamlessly translated into industrial reality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 6-benzothiazole sulfonyl chloride meets the exacting standards required for pharmaceutical synthesis. Our commitment to quality assurance ensures that our clients receive materials with consistent impurity profiles, facilitating smoother regulatory filings and faster time-to-market for their final drug products.
We invite global partners to collaborate with us to leverage this superior synthetic technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our optimized manufacturing processes can enhance your production efficiency and reduce overall project costs. Let us be your trusted partner in delivering the next generation of high-performance pharmaceutical intermediates.
