Advanced Synthetic Route for Clarithromycin Intermediate via Lactam Salt Catalysis
The pharmaceutical industry is constantly seeking more sustainable and efficient pathways for the production of critical antibiotic intermediates. A significant breakthrough in this domain is detailed in patent CN102633851A, which discloses a novel synthetic method for a clarithromycin intermediate. This technology represents a paradigm shift from traditional hazardous processes to a greener, safer catalytic system. By replacing the conventional pyridine hydrochloride catalyst with an environmentally benign lactam salt, the process addresses long-standing issues regarding equipment corrosion, operator safety, and waste management. For R&D directors and procurement specialists alike, this innovation offers a compelling value proposition: a robust, scalable route that delivers high-purity intermediates while drastically simplifying the environmental compliance burden. As a leading manufacturer, we recognize the strategic importance of adopting such forward-thinking chemistries to secure supply chains for second-generation macrolide antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of clarithromycin intermediates has relied heavily on the use of pyridine hydrochloride as a catalyst during the critical etherification step. While effective in driving the reaction, pyridine hydrochloride presents severe operational challenges that impact both the bottom line and workplace safety. It is highly corrosive to standard reactor materials, necessitating expensive specialized equipment and frequent maintenance schedules to prevent leaks and failures. Furthermore, pyridine itself is volatile, explosive, and possesses strong irritant and neurotoxic properties, creating a hazardous environment for plant personnel. From a supply chain perspective, the disposal of waste streams containing pyridine derivatives is costly and complex, requiring rigorous treatment protocols to meet environmental regulations. These factors collectively inflate the cost of goods sold (COGS) and introduce significant risk into the manufacturing timeline, making the conventional route increasingly untenable for modern, compliant production facilities.
The Novel Approach
The innovative methodology described in the patent data introduces a lactam salt (Formula IV) as a superior catalytic alternative, fundamentally altering the reaction landscape for producing clarithromycin precursors. This approach utilizes readily available lactams reacted with simple acids to generate the active catalyst in situ or as a pre-formed salt, eliminating the need for hazardous pyridine derivatives. The process involves dissolving erythromycin A-9-oxime (Formula II) in a safe organic solvent, followed by the addition of the lactam salt and an etherifying agent like 2-ethoxypropene. This modification not only preserves the integrity of the sensitive macrolide ring but also operates under mild thermal conditions, typically between 10-60°C. The result is a cleaner reaction profile with significantly reduced byproduct formation. By integrating this greener catalyst system, manufacturers can achieve substantial cost reduction in pharmaceutical intermediates manufacturing through simplified waste treatment and enhanced equipment longevity.
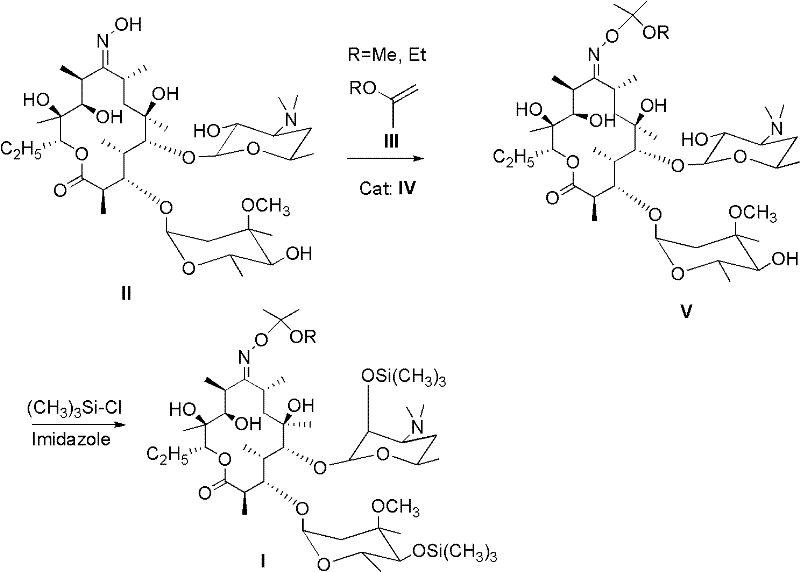
Mechanistic Insights into Lactam Salt-Catalyzed Etherification
The core of this technological advancement lies in the mechanistic role of the lactam salt during the etherification and subsequent silylation steps. Unlike strong mineral acids or corrosive pyridine salts, the lactam salt acts as a mild, buffered proton source that facilitates the activation of the etherifying agent without promoting the acid-catalyzed degradation of the glycosidic bonds in the erythromycin scaffold. In the first stage, the oxime group at the C-9 position is protected via etherification using agents such as 2-methoxypropene or 2-ethoxypropene. The lactam salt stabilizes the transition state, ensuring high regioselectivity and preventing unwanted side reactions that often plague macrolide chemistry. Following this, the process employs a silylation step using trimethylchlorosilane and imidazole to protect the hydroxyl groups at the 2' and 4'' positions. This dual-protection strategy is crucial for the subsequent methylation steps required to produce the final API, and the mild nature of the lactam catalyst ensures that the stereochemistry of the molecule remains intact throughout the sequence.
Impurity control is another critical aspect where this novel mechanism excels. Traditional methods often suffer from the formation of difficult-to-remove impurities caused by the harsh acidic environment of pyridine hydrochloride, which can lead to dehydration or rearrangement of the macrolide ring. The new process mitigates these risks by maintaining a neutral to slightly acidic environment controlled by the lactam salt. This results in a crude product with a much cleaner impurity profile, reducing the burden on downstream purification units such as chromatography or recrystallization. For quality assurance teams, this means a more consistent product with fewer out-of-specification batches. The ability to strictly control the reaction parameters—specifically temperature ranges of 20-60°C during silylation and precise molar ratios of reagents—further enhances the reproducibility of the synthesis, making it an ideal candidate for technology transfer and commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Clarithromycin Intermediate Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and thermal control to maximize yield and purity. The process begins with the dissolution of the starting material, erythromycin A-9-oxime, in a suitable solvent such as dichloromethane or 2-methyltetrahydrofuran. The lactam salt is then introduced alongside the etherifying agent, initiating the protection of the oxime moiety. Once this step is complete, the reaction mixture is treated with silylating agents to cap the remaining hydroxyl groups. The detailed standardized synthesis steps, including specific molar ratios, temperature ramps, and workup procedures, are outlined in the guide below to ensure successful replication in your pilot or production plant.
- Dissolve Erythromycin A-9-oxime (Formula II) in an organic solvent such as dichloromethane or DMF.
- Add the environmentally friendly lactam salt (Formula IV) and the etherifying agent (Formula III), reacting at 10-60°C for 1-4 hours.
- Introduce imidazole and trimethylchlorosilane for silylation at 20-60°C, followed by aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this lactam salt-catalyzed process offers tangible strategic benefits beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Pyridine and its salts are subject to strict regulatory controls and price volatility due to their hazardous nature and widespread industrial use. In contrast, the lactams and simple mineral acids required for this new method are commodity chemicals with stable pricing and abundant global availability. This shift reduces the risk of supply disruptions and allows for more predictable budgeting. Furthermore, the elimination of corrosive reagents extends the lifespan of reactor vessels and piping, deferring capital expenditure on equipment replacement. These factors combine to create a more resilient and cost-effective manufacturing operation that is less susceptible to external market shocks.
- Cost Reduction in Manufacturing: The economic impact of switching to this green catalytic system is profound, driven primarily by the elimination of expensive waste treatment protocols. Disposing of pyridine-containing waste streams requires specialized incineration or chemical neutralization, which adds significant overhead to every batch produced. By replacing this with biodegradable lactam salts, the volume and toxicity of waste are significantly reduced, leading to lower disposal fees. Additionally, the milder reaction conditions reduce energy consumption for heating and cooling, further optimizing the utility costs associated with production. The high yields reported in the patent data, often exceeding 90%, mean that less starting material is wasted, directly improving the material cost efficiency of the entire process.
- Enhanced Supply Chain Reliability: Reliability is the cornerstone of a robust pharmaceutical supply chain, and this method enhances it by removing bottlenecks associated with hazardous material handling. Facilities no longer need to maintain specialized storage zones for explosive pyridine derivatives, freeing up warehouse space and reducing insurance premiums. The use of common solvents like dichloromethane or ethyl acetate, which are already staples in most chemical plants, ensures that logistics remain straightforward. Moreover, the safety profile of the new reagents minimizes the risk of unplanned shutdowns due to safety incidents or regulatory inspections, ensuring a continuous flow of high-purity intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this technology is inherently designed for scalability. The exothermic nature of the reaction is manageable within standard jacketed reactors, and the absence of toxic gases simplifies venting requirements. From an environmental compliance standpoint, the process aligns perfectly with modern green chemistry principles, reducing the carbon footprint of the manufacturing site. This makes it easier for companies to meet increasingly stringent ESG (Environmental, Social, and Governance) targets. The ability to run this process in standard equipment without major retrofits means that capacity can be increased rapidly to meet market demand, providing a competitive edge in the fast-paced antibiotic market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These answers are derived directly from the experimental data and claims found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source.
Q: Why is the lactam salt catalyst preferred over pyridine hydrochloride?
A: Pyridine hydrochloride is corrosive to equipment and poses significant safety risks due to explosivity and neurotoxicity. The lactam salt offers a non-corrosive, environmentally friendly alternative that eliminates these hazards while maintaining high reaction yields.
Q: What are the typical reaction conditions for this synthesis?
A: The etherification step typically occurs between 10-60°C for 1-4 hours, followed by a silylation step at 20-60°C for 1-3 hours. Common solvents include dichloromethane, cyclohexanone, or DMF.
Q: How does this method impact product purity?
A: By utilizing milder catalytic conditions and avoiding harsh acidic environments associated with traditional methods, this process minimizes degradation of the macrolide structure, resulting in high-purity intermediates suitable for downstream API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clarithromycin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this lactam salt-catalyzed process can be realized at an industrial level. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of clarithromycin intermediate meets the highest global standards. Our commitment to quality ensures that the impurity profiles remain consistent, facilitating smoother regulatory filings for our clients' final drug products.
We invite you to collaborate with us to optimize your supply chain for macrolide antibiotics. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and regional regulatory requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a secure, sustainable, and economically superior source of critical pharmaceutical intermediates, positioning your organization for success in the evolving global healthcare market.
