Revolutionizing Deuterated Solvent Production: A Safe, Scalable Route to HMPA-D18
Revolutionizing Deuterated Solvent Production: A Safe, Scalable Route to HMPA-D18
The landscape of deuterated solvent manufacturing is undergoing a significant transformation driven by the need for safer, more scalable, and cost-effective synthetic routes. Patent CN111303206B introduces a groundbreaking methodology for the synthesis of Deuterated Hexamethylphosphoric Triamide-D18 (HMPA-D18), a critical high-boiling polar aprotic solvent widely used in NMR spectroscopy and specialized organic synthesis. This innovation addresses long-standing logistical and safety challenges associated with traditional gas-phase reactions by leveraging a novel solid-state substitution strategy. By shifting from hazardous gaseous reagents to stable solid salts, this technology not only enhances operational safety but also streamlines the supply chain for high-value deuterated intermediates. For R&D directors and procurement specialists, understanding this shift is vital for securing reliable sources of high-purity analytical reagents.
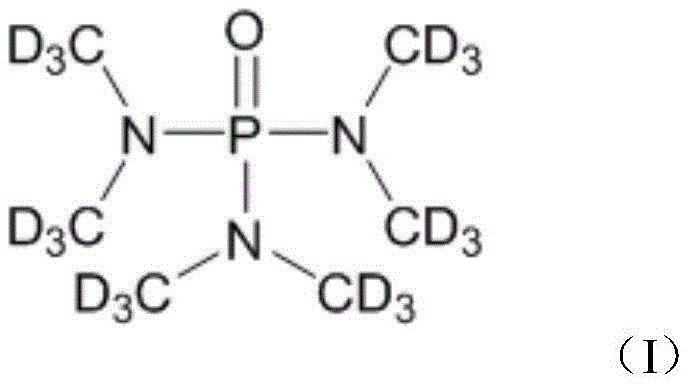
HMPA-D18 serves as a multifunctional solvent capable of dissolving a vast array of organic compounds, including biomass and metal complexes, which are often insoluble in common deuterated solvents like CDCl3. Its ability to form mixed solvent systems without precipitation makes it indispensable for complex structural analysis. However, the historical reliance on difficult-to-handle gaseous precursors has limited its widespread commercial adoption. The new synthesis method detailed in the patent overcomes these barriers by utilizing deuterated dimethylamine hydrochloride, a commercially accessible solid, reacting it with phosphorus oxychloride in the presence of triethylamine. This approach represents a paradigm shift in how fine chemical manufacturers approach the production of complex deuterated molecules, offering a robust pathway for industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of hexamethylphosphoric triamide and its deuterated analogues relied heavily on the reaction between dimethylamine gas and phosphorus oxychloride. This conventional approach presents severe logistical and safety hurdles that complicate large-scale manufacturing. Dimethylamine is a volatile gas at room temperature, requiring specialized pressurized storage cylinders and complex gas-handling infrastructure to ensure safe delivery into the reaction vessel. Furthermore, sourcing high-purity deuterated dimethylamine gas is exceptionally difficult and costly, often leading to supply chain bottlenecks for manufacturers of deuterated solvents. The handling of gaseous amines also poses significant occupational health risks, necessitating rigorous containment protocols and increasing the overall operational expenditure related to safety compliance. These factors collectively render the traditional gas-phase route inefficient and economically unviable for modern, agile chemical production facilities seeking to minimize risk while maximizing output.
The Novel Approach
The innovative method disclosed in patent CN111303206B fundamentally re-engineers the synthesis by replacing the problematic gaseous amine with deuterated dimethylamine hydrochloride, a stable solid salt. This substitution eliminates the need for complex gas handling equipment, allowing the reaction to proceed in a standard liquid-phase batch reactor. The process utilizes dichloromethane as the reaction medium and triethylamine as a base to scavenge the hydrochloric acid byproduct, facilitating a smooth nucleophilic substitution at the phosphorus center. By operating at mild conditions—specifically room temperature after an initial cooling phase—the method ensures high selectivity and minimizes side reactions that could compromise isotopic purity. This transition from gas to solid reagents not only simplifies the physical setup of the plant but also drastically reduces the barrier to entry for producing high-value deuterated solvents, making the supply chain more resilient and responsive to market demands.
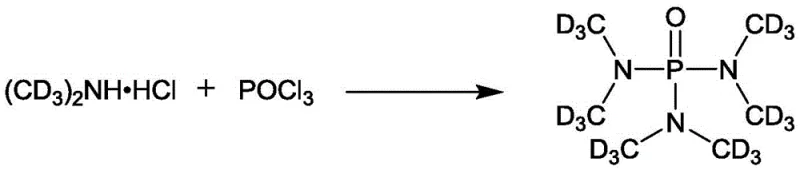
Mechanistic Insights into Phosphorylation via Nucleophilic Substitution
The core chemical transformation in this synthesis is a nucleophilic substitution reaction where the nitrogen atom of the deuterated amine attacks the electrophilic phosphorus atom of phosphorus oxychloride (POCl3). In this mechanism, the deuterated dimethylamine hydrochloride acts as the source of the nucleophile, while triethylamine plays a dual role: it neutralizes the hydrochloric acid bound to the amine salt to release the free amine in situ, and it subsequently scavenges the HCl generated during the substitution steps. The reaction proceeds through a series of displacements where chloride ions on the phosphorus are sequentially replaced by dimethylamino groups. The choice of dichloromethane as the solvent is mechanistically critical; it provides the necessary polarity to dissolve the ionic intermediates and the amine salt while remaining inert to the highly reactive phosphoryl chloride. Unlike non-polar solvents like toluene or coordinating solvents like THF, which were found to be ineffective in this specific system, DCM supports the formation of the transition state required for high-yield conversion to the final triamide product.
Controlling impurities in deuterated solvent synthesis is paramount, as residual protons or chlorinated byproducts can interfere with sensitive NMR applications. The patented process achieves exceptional purity levels, reported at 98.5% by GC analysis, through precise stoichiometric control and optimized workup procedures. The use of a slight excess of the amine salt (molar ratio of 4.50:1 relative to POCl3) ensures complete consumption of the phosphorus starting material, preventing the formation of partially substituted chlorophosphates which are difficult to separate. Furthermore, the reaction temperature profile, involving an initial ice bath addition followed by a prolonged 16-hour stir at room temperature, allows for the gradual evolution of the reaction without thermal runaway. This controlled kinetic environment minimizes the formation of degradation products and ensures that the deuterium label remains intact on the methyl groups, preserving the high isotopic enrichment required for premium analytical grade solvents.
How to Synthesize Deuterated Hexamethylphosphoric Triamide-D18 Efficiently
The synthesis protocol outlined in the patent offers a straightforward yet highly effective route for producing HMPA-D18, suitable for both laboratory optimization and commercial manufacturing. The process begins with the suspension of the solid amine salt in the solvent, followed by the careful addition of the base to generate the reactive species. Subsequent addition of the phosphorylating agent is conducted under cooling to manage the exotherm, ensuring safety and product integrity. The reaction mixture is then allowed to mature over an extended period to drive the conversion to completion. For detailed operational parameters, stoichiometry, and purification techniques, please refer to the standardized synthesis guide below.
- Suspend deuterated dimethylamine hydrochloride in dichloromethane and add triethylamine at room temperature with stirring.
- Add phosphorus oxychloride in portions under an ice bath to control the exothermic reaction.
- Warm the mixture to room temperature and react for 16 hours, then filter, concentrate, and distill to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this solid-state synthesis route offers profound strategic advantages that extend beyond simple chemical yield. The shift away from gaseous reagents fundamentally alters the cost structure and risk profile of manufacturing deuterated solvents. By utilizing commercially available solid salts, manufacturers can leverage existing bulk chemical logistics networks rather than relying on specialized gas suppliers, leading to significant reductions in raw material acquisition costs and lead times. Additionally, the simplified reactor requirements mean that production can be scaled up using standard glass-lined or stainless steel vessels without the need for expensive pressure-rated equipment, thereby lowering capital expenditure barriers for new production lines. This flexibility allows suppliers to respond more rapidly to fluctuations in demand from the pharmaceutical and analytical sectors, ensuring a more stable and continuous supply of critical reagents.
- Cost Reduction in Manufacturing: The elimination of gaseous dimethylamine removes the need for complex vaporization and dosing systems, which are capital-intensive and require frequent maintenance. Furthermore, solid reagents generally have lower transportation and storage costs compared to compressed gases, contributing to a leaner overall cost of goods sold. The high yield reported in the patent implies less waste generation and lower raw material consumption per unit of product, further enhancing the economic efficiency of the process. These cumulative savings allow suppliers to offer more competitive pricing structures for high-purity deuterated solvents without compromising on quality margins.
- Enhanced Supply Chain Reliability: Sourcing deuterated dimethylamine hydrochloride is significantly more reliable than sourcing the corresponding gas, as solid chemicals are less susceptible to shipping restrictions and logistical delays associated with hazardous pressurized containers. This stability in raw material supply translates directly into improved delivery performance for downstream customers, reducing the risk of production stoppages due to reagent shortages. The robustness of the reaction conditions also means that production schedules are less prone to disruption from equipment failures or safety incidents, fostering a more predictable and trustworthy supply partnership for global chemical buyers.
- Scalability and Environmental Compliance: The use of dichloromethane, a common industrial solvent, facilitates easy solvent recovery and recycling, aligning with modern green chemistry initiatives and reducing waste disposal costs. The absence of high-pressure operations simplifies regulatory compliance and safety auditing, accelerating the time-to-market for new production facilities. Moreover, the high selectivity of the reaction minimizes the formation of hazardous byproducts, simplifying effluent treatment processes and reducing the environmental footprint of the manufacturing site. This scalability ensures that the technology can meet growing global demand for deuterated materials while adhering to stringent environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of HMPA-D18, based on the specific innovations detailed in the patent literature. Understanding these nuances helps stakeholders make informed decisions about integrating this material into their analytical workflows or supply chains. The answers reflect the practical realities of implementing this novel synthesis route in a commercial setting.
Q: Why is the solid-state route for HMPA-D18 preferred over the gas-phase method?
A: The conventional method uses gaseous dimethylamine, which is difficult to purchase, store, and handle safely. The patented method utilizes commercially available deuterated dimethylamine hydrochloride, a solid salt that significantly simplifies operation and improves safety profiles.
Q: What is the critical solvent for this deuteration reaction?
A: Dichloromethane (DCM) is the critical solvent. Experiments showed that solvents like toluene or tetrahydrofuran failed to yield the target product effectively when used with triethylamine, whereas DCM provided high yields and purity.
Q: What purity levels can be achieved with this synthesis method?
A: According to the patent data, this method achieves a GC purity of 98.5% with a yield of approximately 90%, making it highly suitable for analytical and pharmaceutical applications requiring high isotopic enrichment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Hexamethylphosphoric Triamide-D18 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity deuterated solvents play in advancing pharmaceutical research and materials science. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of HMPA-D18 meets the exacting standards required for NMR spectroscopy and sensitive chemical synthesis. Our commitment to quality assurance means that you receive a product that is not only chemically pure but also isotopically enriched to the highest degree, minimizing background noise in your analytical data.
We invite you to collaborate with us to optimize your solvent supply chain and reduce your overall operational costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific consumption patterns. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your R&D objectives. Let us be your partner in delivering the high-performance chemical solutions necessary for your next breakthrough.
