Industrial Scale Synthesis of 4-Furfurylthiopentanone-2 via Safe Wittig Protocol
The global demand for high-quality meat flavor enhancers continues to drive innovation in fine chemical synthesis, particularly for key intermediates like 4-furfurylthiopentanone-2. A significant technological breakthrough in this domain is detailed in patent CN110357840B, which introduces a robust and industrially viable preparation method that fundamentally alters the economic and safety profile of production. Unlike legacy processes that depend on perilous organometallic reagents, this novel approach leverages a Wittig reaction strategy starting from chloropropanone, a readily available and cost-effective feedstock. This shift not only mitigates the severe safety hazards associated with pyrophoric materials but also streamlines the purification workflow, resulting in superior product consistency. For R&D directors and procurement strategists, understanding this transition is critical, as it represents a move towards sustainable, high-yield manufacturing capable of meeting stringent food safety regulations while optimizing supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-furfurylthiopentanone-2 has been plagued by reliance on highly reactive and dangerous chemical pathways that pose significant barriers to safe industrial scale-up. The most prominent traditional route involves a Grignard reaction, where methyl magnesium bromide is reacted with an unsaturated aldehyde, followed by oxidation using Pyridinium Chlorochromate (PCC). As illustrated in the reaction scheme below, this pathway is fraught with complications, including the inherent instability of Grignard reagents which require strictly anhydrous conditions and inert atmospheres to prevent runaway reactions. Furthermore, the subsequent oxidation step using PCC is notorious for generating toxic chromium waste, creating substantial environmental compliance burdens and disposal costs that erode profit margins. Alternative historical methods utilizing methyllithium or trimethylaluminum suffer from similar drawbacks, characterized by extreme reactivity, low tolerance for functional groups, and prohibitively high raw material costs that render them economically unfeasible for bulk commodity production.
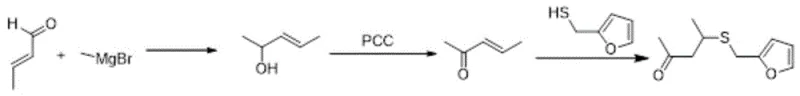
The Novel Approach
In stark contrast to these hazardous legacy methods, the invention disclosed in CN110357840B proposes a transformative synthetic strategy that prioritizes safety and economic efficiency without compromising on yield. The core of this innovation lies in the utilization of chloropropanone as the primary building block, which undergoes a quaternization reaction with triphenylphosphine or triethyl phosphite to form a stable phosphonium salt. This salt is then converted into a phosphorus ylide, which serves as the key nucleophile in a subsequent Wittig olefination with acetaldehyde. This sequence elegantly constructs the carbon skeleton of the target molecule, 3-penten-2-one, under mild conditions that are easily manageable in standard stainless steel reactors. The final step involves a base-catalyzed conjugate addition of furfuryl mercaptan, completing the synthesis with high atom economy. This modular approach eliminates the need for cryogenic temperatures and exotic reagents, thereby drastically simplifying the engineering requirements for commercial plants.
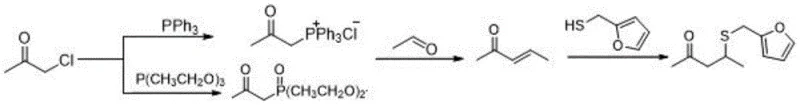
Mechanistic Insights into Wittig Olefination and Thiol Addition
The mechanistic elegance of this new process is rooted in the precise control of the Wittig reaction dynamics, which ensures high stereoselectivity and minimizes byproduct formation. In the initial phase, the phosphonium salt is generated through a nucleophilic substitution where the phosphorus atom attacks the electrophilic carbon of chloropropanone. Upon treatment with a strong base such as sodium hydroxide or sodium hydride, deprotonation occurs at the alpha-position relative to the phosphorus, generating the reactive ylide species. This ylide then attacks the carbonyl carbon of acetaldehyde to form a betaine intermediate, which cyclizes into an oxaphosphetane four-membered ring. The collapse of this ring releases triphenylphosphine oxide and yields the desired alpha,beta-unsaturated ketone, 3-penten-2-one. The choice of base and solvent system, such as dichloromethane with aqueous NaOH or THF with NaH, is critical for maintaining the stability of the ylide and preventing side reactions like self-condensation of the aldehyde, ensuring a clean reaction profile that facilitates downstream processing.
Following the formation of the enone intermediate, the final transformation relies on a Michael-type addition mechanism where the sulfur nucleophile attacks the beta-carbon of the unsaturated system. The presence of a mild organic base catalyst, specifically piperidine, activates the furfuryl mercaptan by increasing the nucleophilicity of the thiol group while simultaneously activating the enone electrophile. This step is conducted at a controlled temperature range of 35-40°C, which is sufficiently warm to drive the kinetics forward but cool enough to prevent polymerization or degradation of the sensitive furan ring. The result is a highly regioselective addition that places the sulfur moiety exactly at the 4-position of the pentanone chain. This mechanistic precision is what allows the process to achieve a remarkable product purity of up to 98.5% as verified by gas chromatography, effectively eliminating the complex impurity profiles often seen in radical-based or organometallic coupling reactions.
How to Synthesize 4-Furfurylthiopentanone-2 Efficiently
Implementing this synthesis requires careful attention to the sequential generation of the phosphorus ylide and the subsequent olefination steps to maximize throughput. The process begins with the reflux of chloropropanone and triphenylphosphine in toluene, followed by the careful generation of the ylide using a biphasic system or anhydrous conditions depending on the specific base selected. Once the 3-penten-2-one intermediate is secured, the final thiol addition must be managed with precise temperature control to ensure safety and yield. For laboratory technicians and process engineers looking to replicate or scale this methodology, the detailed standardized operating procedures regarding reagent ratios, addition rates, and workup protocols are essential for success. The following guide outlines the critical operational parameters derived directly from the patent examples to ensure reproducible high-quality outcomes.
- React chloropropanone with triphenylphosphine or triethyl phosphite under reflux to generate the phosphonium salt precursor.
- Treat the phosphonium salt with a strong base such as sodium hydroxide or sodium hydride to form the reactive phosphorus ylide.
- Perform a Wittig reaction with acetaldehyde to obtain 3-penten-2-one, followed by piperidine-catalyzed addition of furfuryl mercaptan to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this chloropropanone-based route offers profound advantages that directly address the pain points of cost volatility and supply discontinuity in the flavor and fragrance sector. By shifting away from specialized organometallic reagents like methyllithium, which require dedicated storage infrastructure and specialized handling teams, manufacturers can significantly reduce their operational overhead and insurance liabilities. The substitution of expensive, hazard-class 4.2 reagents with commodity chemicals like chloropropanone and triphenylphosphine creates a more resilient supply chain that is less susceptible to market shocks or transportation restrictions. Furthermore, the simplified post-treatment steps, which involve standard liquid-liquid extractions and crystallizations rather than complex chromatographic purifications, translate into faster batch cycle times and higher equipment utilization rates. This efficiency gain allows suppliers to offer more competitive pricing structures while maintaining healthy margins, making it an attractive option for large-scale buyers seeking long-term cost reduction in flavor intermediate manufacturing.
- Cost Reduction in Manufacturing: The elimination of costly and hazardous reagents such as trimethylaluminum and PCC removes a significant portion of the raw material expense and waste disposal burden. The new process utilizes chloropropanone, a bulk chemical with a stable and low market price, which drives down the overall cost of goods sold substantially. Additionally, the ability to recover and recycle solvents like toluene and dichloromethane further enhances the economic viability of the process, ensuring that the final product remains price-competitive even in fluctuating market conditions.
- Enhanced Supply Chain Reliability: Relying on widely available industrial feedstocks rather than niche organometallics ensures a consistent and uninterrupted supply of raw materials. The robustness of the reaction conditions, which do not require extreme cryogenic temperatures or ultra-dry environments, reduces the risk of batch failures due to minor deviations in utility quality. This reliability is crucial for procurement managers who need to guarantee delivery schedules to downstream flavor houses, as it minimizes the likelihood of production delays caused by reagent shortages or safety shutdowns.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing reaction types that are well-understood and easily managed in multi-ton reactors. The avoidance of heavy metal oxidants like chromium aligns with increasingly strict global environmental regulations, reducing the regulatory risk profile for manufacturing sites. This green chemistry advantage not only simplifies permitting and auditing processes but also enhances the brand reputation of the supplier as a responsible manufacturer committed to sustainable production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived from the specific experimental data and beneficial effects reported in the patent documentation, providing clarity on yield expectations, safety protocols, and scalability factors. Understanding these details is vital for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement specialists assessing supplier capabilities.
Q: Why is the new Wittig-based route safer than traditional Grignard methods?
A: Traditional methods rely on hazardous reagents like methyllithium or trimethylaluminum which are pyrophoric and difficult to handle on large scales. The new protocol uses stable chloropropanone and standard phosphine reagents, significantly reducing operational risk.
Q: What is the expected purity of 4-furfurylthiopentanone-2 using this method?
A: According to patent data, the optimized process achieves a product purity of up to 98.5% as determined by gas chromatography, with a total yield reaching 77.7%.
Q: Can this process be scaled for industrial production?
A: Yes, the use of inexpensive, commercially available raw materials like chloropropanone and the elimination of cryogenic conditions make this route highly suitable for multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Furfurylthiopentanone-2 Supplier
As the industry shifts towards safer and more efficient synthetic methodologies, NINGBO INNO PHARMCHEM stands at the forefront of translating these patented innovations into commercial reality. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and robust. We are committed to delivering high-purity 4-furfurylthiopentanone-2 that meets stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest standards of quality and consistency required by the global flavor and fragrance industry.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your flavor ingredient sourcing strategy.
