Advanced Manufacturing of Etoricoxib Intermediates: A Novel Grignard-Based Synthetic Route for High-Purity API Production
Advanced Manufacturing of Etoricoxib Intermediates: A Novel Grignard-Based Synthetic Route for High-Purity API Production
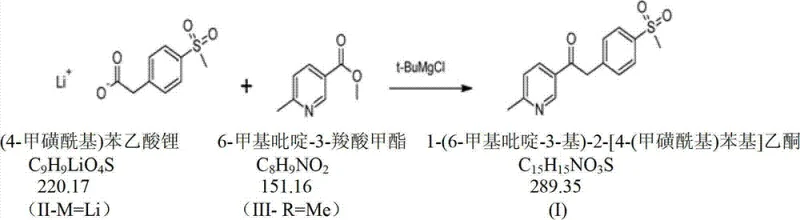
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for Critical Starting Materials (CSMs) and Key Intermediates, particularly for high-volume anti-inflammatory drugs like Etoricoxib (Arcoxia). Patent CN102898357A introduces a transformative methodology for synthesizing 1-(6-methylpyridin-3-yl)-2-[4-(methylsulfonyl)phenyl]ethanone, a pivotal building block in the COX-2 inhibitor value chain. This innovation addresses long-standing challenges in yield optimization and impurity profiling, specifically targeting the notorious 'Impurity 408'. By leveraging a sophisticated Grignard-mediated coupling strategy, the disclosed process achieves molar yields ranging from 78% to 88%, significantly outperforming historical benchmarks. For R&D directors and procurement specialists, this represents a viable pathway to secure a reliable API intermediate supplier capable of delivering high-purity materials with reduced environmental footprint.
![Chemical structure of 1-(6-methylpyridin-3-yl)-2-[4-(methylsulfonyl)phenyl]ethanone, the key Etoricoxib intermediate](/insights/img/etoricoxib-intermediate-grignard-synthesis-pharma-supplier-20260307040731-01.png)
The structural integrity of the final active pharmaceutical ingredient relies heavily on the quality of this ketone intermediate. Conventional syntheses often struggle with the introduction of the sulfone moiety late in the sequence or suffer from poor regioselectivity during the carbon-carbon bond formation. The technology described in CN102898357A circumvents these issues by utilizing a pre-functionalized (4-methylsulfonyl)phenylacetic acid derivative. This strategic choice not only simplifies the synthetic tree but also enhances the overall atom economy. As we delve deeper into the mechanistic nuances and commercial implications, it becomes evident that this process offers a compelling value proposition for cost reduction in pharmaceutical manufacturing, aligning perfectly with the rigorous demands of modern GMP production environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific ethanone scaffold has been plagued by inefficient oxidation steps and suboptimal coupling reactions. Prior art, such as the methods described in WO 2001/007410 by Lonza and Merck, typically involves a multi-step sequence culminating in a hazardous oxidation using hydrogen peroxide catalyzed by sodium tungstate. This approach presents significant safety liabilities due to the potential explosivity of organic peroxides and the generation of N-oxide byproducts on the pyridine ring, which are notoriously difficult to purge. Furthermore, alternative literature methods, such as those by Davies et al. (Journal of Organic Chemistry, 2000), rely on the formation of dianionic magnesium species but often require excessive amounts of Grignard reagents—typically three equivalents—to drive the reaction to completion. Even with this excess, these legacy processes frequently result in unacceptable levels of side products, necessitating complex purification protocols that erode overall yield and increase production costs.
The Novel Approach
The breakthrough detailed in the patent lies in the precise manipulation of reaction kinetics through simultaneous addition. Instead of sequential dosing or pre-mixing, the novel protocol dictates the concurrent and continuous introduction of both the Grignard reagent (preferably tert-butylmagnesium chloride) and the 6-methylpyridine-3-carboxylic acid ester into the reaction vessel containing the phenylacetic acid salt. This synchronized addition maintains a low steady-state concentration of reactive intermediates, effectively suppressing the bimolecular side reactions that lead to dimerization or over-alkylation. By optimizing the stoichiometry, particularly when using the lithium salt of the starting acid, the process reduces the Grignard requirement to merely 1.6 to 1.8 equivalents. This refinement not only lowers raw material consumption but also simplifies the workup procedure, as there is less excess organometallic species to quench, thereby enhancing the safety profile and operational efficiency of the manufacturing campaign.
Mechanistic Insights into Dianion-Mediated Acylation
At the heart of this chemical transformation is the generation of a highly reactive dianionic species derived from (4-methylsulfonyl)phenylacetic acid. Upon treatment with a strong base or directly with the Grignard reagent, the carboxylic acid proton and the benzylic proton are abstracted to form a resonance-stabilized nucleophile. When the lithium salt is employed, the resulting dianion exhibits superior solubility and reactivity characteristics in tetrahydrofuran (THF) compared to its sodium or potassium counterparts. The mechanism proceeds through a nucleophilic attack of this dianion on the carbonyl carbon of the pyridine ester. Crucially, the presence of the alkali metal cation (M+) appears to coordinate with the carbonyl oxygen of the incoming ester, activating it towards attack while simultaneously stabilizing the tetrahedral intermediate. This coordination sphere is vital for directing the reaction pathway towards the desired ketone rather than allowing competitive decomposition or side reactions.
Following the acylation event, the intermediate undergoes a spontaneous decarboxylation step, releasing carbon dioxide and yielding the target ketone structure. The elegance of this mechanism is further highlighted by its ability to suppress the formation of 'Impurity 408', a structurally related bis-ketone byproduct. Structural analysis reveals that Impurity 408 arises from the reaction of the product ketone with another equivalent of the dianion or Grignard species. By maintaining strict control over the addition rates and temperature (optimally between 65°C and 70°C), the process ensures that the concentration of the highly nucleophilic dianion never exceeds the threshold required to attack the newly formed ketone product. This kinetic discrimination is the key to achieving the reported purity levels of greater than 96% directly from the crude reaction mixture, minimizing the need for resource-intensive recrystallization steps.
Furthermore, the choice of the lithium cation plays a subtle yet profound role in the transition state geometry. The smaller ionic radius of lithium allows for tighter chelation with the oxygen atoms of the substrate and the solvent, creating a more organized and selective reaction environment. This 'template effect' likely contributes to the observed reduction in side products when compared to reactions utilizing sodium or magnesium salts alone. The decarboxylation step itself is facilitated by the electron-withdrawing nature of the sulfone group on the aromatic ring, which stabilizes the developing negative charge on the benzylic carbon during the loss of CO2. Understanding these electronic and steric factors allows process chemists to fine-tune reaction parameters for maximum efficiency, ensuring that the commercial scale-up of complex pharmaceutical intermediates remains robust and reproducible across different batch sizes.
How to Synthesize 1-(6-methylpyridin-3-yl)-2-[4-(methylsulfonyl)phenyl]ethanone Efficiently
The practical execution of this synthesis requires careful attention to reagent quality and addition dynamics to replicate the high yields reported in the patent literature. The process begins with the preparation of the lithium salt of (4-methylsulfonyl)phenylacetic acid, which serves as the foundational nucleophile for the subsequent coupling. Once the salt is suspended in anhydrous THF and heated to reflux, the critical phase of simultaneous addition commences. Operators must ensure that the flow rates of the Grignard solution and the pyridine ester solution are synchronized to maintain the optimal stoichiometric balance throughout the reaction duration. Detailed standardized operating procedures regarding temperature control, quenching protocols, and isolation techniques are essential for transferring this laboratory-scale success to pilot and production scales.
- Preparation of the alkali metal salt of (4-methylsulfonyl)phenylacetic acid, preferably the lithium salt, using lithium hydroxide in methanol.
- Simultaneous and continuous addition of tert-butylmagnesium chloride and methyl 6-methylpyridine-3-carboxylate to the salt solution in THF at 65-70°C.
- Quenching the reaction mixture with water and acid, followed by extraction and crystallization to isolate the high-purity ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the significant simplification of the supply chain for raw materials. By utilizing (4-methylsulfonyl)phenylacetic acid, a commercially available commodity chemical, the process eliminates the need for custom synthesis of sulfone-containing precursors or the handling of hazardous oxidizing agents like hydrogen peroxide on a large scale. This shift not only reduces the regulatory burden associated with storing and transporting dangerous goods but also mitigates the risk of supply disruptions caused by the limited availability of specialized catalysts such as tungsten salts. Consequently, manufacturers can achieve substantial cost savings through the consolidation of their vendor base and the utilization of bulk commodity chemicals.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic reduction in reagent consumption. Traditional methods often demand a three-fold molar excess of expensive Grignard reagents to overcome sluggish kinetics and side reactions. In contrast, this optimized protocol operates efficiently with less than two equivalents of the organometallic species when the lithium salt pathway is selected. This reduction directly translates to lower raw material costs and decreased waste disposal expenses, as there is less magnesium sludge to treat post-reaction. Additionally, the avoidance of heavy metal catalysts removes the costly and time-consuming step of metal scavenging, which is often required to meet stringent residual metal specifications for API intermediates.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical sector, where any delay in intermediate supply can halt entire production lines. This method enhances reliability by relying on robust, non-cryogenic conditions (65-70°C) that are easier to maintain in standard glass-lined steel reactors compared to low-temperature cryogenic processes. The tolerance of the reaction to slight variations in addition rates provides a wider operating window, reducing the likelihood of batch failures due to operator error or equipment fluctuation. Furthermore, the ability to recover and recycle unreacted starting materials, as demonstrated in the patent examples, adds an extra layer of security against yield losses, ensuring consistent output volumes even if minor deviations occur during the campaign.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. The elimination of peroxide-based oxidations removes the risk of thermal runaways and explosions, a critical consideration for scaling up exothermic reactions. The solvent system, primarily based on THF and methanol, is well-established in the industry with efficient recovery infrastructure already in place at most CDMO facilities. The reduction in waste generation, characterized by a higher E-factor due to better atom economy and fewer purification steps, aligns with green chemistry principles. This compliance facilitates smoother regulatory approvals and supports the sustainability goals of major pharmaceutical clients who are increasingly scrutinizing the environmental footprint of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on critical process parameters. Understanding these nuances is essential for process development teams aiming to integrate this technology into their existing manufacturing portfolios.
Q: How does this novel process control the formation of Impurity '408'?
A: The process utilizes a simultaneous addition strategy of the Grignard reagent and the pyridine ester to the dianionic species. This kinetic control prevents the over-reaction that leads to the bis-ketone impurity (Impurity '408'), keeping levels below 0.6% without extensive recrystallization.
Q: What are the stoichiometric advantages of using the lithium salt method?
A: By employing the lithium salt of the starting acid, the reaction requires significantly fewer equivalents of the Grignard reagent (1.6 to 1.8 equivalents) compared to conventional methods that often require 3 equivalents, resulting in substantial raw material cost savings.
Q: Why is this method safer than the traditional oxidation routes?
A: Traditional routes often rely on hazardous oxidation steps using hydrogen peroxide and tungsten catalysts to install the sulfone group. This novel route starts with the sulfone already present in the commercially available phenylacetic acid, eliminating the risks associated with explosive oxidants and heavy metal catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(6-methylpyridin-3-yl)-2-[4-(methylsulfonyl)phenyl]ethanone Supplier
As the global demand for COX-2 inhibitors continues to rise, securing a dependable source of high-quality intermediates is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced process chemistry expertise to deliver solutions that bridge the gap between innovative patent technologies and commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation, guaranteeing that every batch of 1-(6-methylpyridin-3-yl)-2-[4-(methylsulfonyl)phenyl]ethanone meets the exacting standards required for API synthesis.
We invite you to collaborate with us to explore how this novel Grignard-based process can optimize your supply chain and reduce your overall cost of goods. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to quality and innovation can support your long-term strategic goals in the pharmaceutical market.
