Revolutionizing Benzothiazole Synthesis: A Green Copper-Catalyzed Alkylation Strategy for Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and sustainable methodologies for constructing complex heterocyclic scaffolds, particularly those serving as critical building blocks for bioactive molecules. Patent CN102120734A introduces a groundbreaking advancement in this domain by detailing a novel method for preparing 2-(N-alkyl)aminobenzothiazole derivatives using activated alcohols as alkylating reagents. This technology represents a significant paradigm shift from traditional halogen-dependent syntheses, leveraging a copper-catalyzed system that operates with exceptional atom economy. For R&D directors and process chemists, this patent offers a compelling solution to long-standing challenges in heterocycle functionalization, specifically addressing the need for greener reaction pathways that minimize hazardous waste generation while maintaining high purity standards essential for drug development.
The strategic value of this invention extends beyond mere academic interest; it provides a tangible pathway for cost reduction in pharmaceutical intermediate manufacturing. By replacing expensive and hazardous halogenated precursors with readily available alcohols, the process simplifies the supply chain and reduces the environmental footprint associated with chemical production. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative synthetic routes is crucial for maintaining competitiveness in a market that increasingly demands sustainability and efficiency. The following analysis delves into the technical nuances of this copper-catalyzed alkylation, exploring its mechanistic underpinnings and commercial viability for large-scale applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(N-alkyl)aminobenzothiazole derivatives has relied heavily on palladium or copper-catalyzed cyclization reactions involving o-halophenylthiourea precursors. This conventional approach suffers from several inherent drawbacks that hinder its efficiency and environmental compatibility. Primarily, the preparation of the starting material, o-halophenylthiourea, requires multi-step synthetic sequences, which inherently lowers the overall yield and increases production costs due to additional purification and handling requirements. Furthermore, these cyclization reactions typically generate hydrohalic acid as a stoichiometric by-product, posing serious environmental hazards and necessitating complex waste treatment protocols to neutralize corrosive acidic streams before disposal.
In addition to the environmental concerns, the reliance on halogenated starting materials introduces significant supply chain vulnerabilities. Halogenated aromatics are often subject to price volatility and regulatory scrutiny due to their toxicity and persistence in the environment. The low atomic efficiency of these traditional methods means that a substantial portion of the reactant mass ends up as waste rather than incorporated into the final product, leading to higher raw material consumption and increased operational expenses. For procurement managers, these factors translate into higher costs of goods sold and greater logistical complexity in managing hazardous chemical inventories, making the search for alternative synthetic routes a high priority for sustainable manufacturing strategies.
The Novel Approach
In stark contrast to the legacy methods, the technology disclosed in CN102120734A utilizes a direct N-alkylation strategy where commercially available or easily synthesized 2-aminobenzothiazole compounds react directly with alcohols. This innovative route eliminates the need for pre-functionalized halogenated precursors, thereby streamlining the synthetic sequence into a single, efficient step. The reaction is facilitated by a catalytic amount of copper salt and a base, proceeding under relatively mild thermal conditions to yield the target 2-(N-alkyl)aminobenzothiazole derivatives with high selectivity. Crucially, the only by-product formed in this transformation is water, which aligns perfectly with the principles of green chemistry and significantly simplifies downstream processing and waste management.
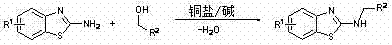
The atom economy of this novel approach is reported to be greater than 93%, a figure that underscores its superior efficiency compared to traditional cyclization methods. By employing alcohols as alkylating agents, the process leverages inexpensive and widely available feedstocks, reducing dependency on specialized halogenated intermediates. This shift not only lowers the direct material costs but also enhances the safety profile of the manufacturing process by avoiding the handling of corrosive hydrohalic acids. For supply chain heads, this translates to a more resilient sourcing strategy, as alcohols are generally more stable and easier to transport than their halogenated counterparts, ensuring consistent production schedules and reduced risk of supply disruptions.
Mechanistic Insights into Copper-Catalyzed N-Alkylation
The core of this technological breakthrough lies in the activation of the alcohol hydroxyl group by the copper catalyst, enabling it to function as an electrophile in the presence of the nucleophilic 2-aminobenzothiazole. The proposed mechanism likely involves the formation of a copper-alkoxide intermediate, which facilitates the departure of the hydroxyl group, potentially through a borrowing hydrogen pathway or direct substitution depending on the specific reaction conditions. The presence of a base, such as sodium hydroxide or potassium hydroxide, is critical for deprotonating the amine nitrogen and regenerating the active catalytic species. This synergistic interaction between the copper salt and the base allows the reaction to proceed efficiently at temperatures ranging from 80°C to 160°C, accommodating a wide variety of substrate electronic properties.
From an impurity control perspective, this mechanism offers distinct advantages over halogen-based routes. Traditional methods often leave traces of unreacted halides or generate halogenated side products that are difficult to remove and can be genotoxic impurities of concern in pharmaceutical applications. The alcohol-based alkylation described here avoids the introduction of halogen atoms entirely, resulting in a cleaner crude reaction profile. This inherent purity simplifies the purification process, often requiring only standard column chromatography or crystallization to achieve high-purity specifications. For quality control teams, this means reduced analytical burden and a lower risk of failing stringent regulatory limits for residual solvents and heavy metals, ultimately accelerating the timeline for clinical material production.
How to Synthesize 2-(N-alkyl)aminobenzothiazole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting involves a straightforward protocol that balances reagent stoichiometry with thermal energy to drive the reaction to completion. The general procedure entails mixing the 2-aminobenzothiazole derivative with a catalytic loading of copper salt, typically between 0.5 mol% and 10 mol%, and a base in a molar ratio of 5 to 40 mol% relative to the substrate. The alcohol alkylating agent is added in slight excess, usually 1 to 4 equivalents, to ensure complete conversion of the amine. The reaction mixture is then heated in a suitable high-boiling solvent such as p-xylene, toluene, or DMF, although solvent-free conditions have also been demonstrated to be effective for certain substrates.
- Charge a reaction vessel with 2-aminobenzothiazole derivatives, a catalytic amount of copper salt (0.5-10 mol%), and a base (5-40 mol%) under nitrogen or air atmosphere.
- Add the activated alcohol alkylating reagent (1-4 equivalents) and a suitable high-boiling solvent such as p-xylene or DMF, or proceed under solvent-free conditions.
- Heat the reaction mixture to a temperature between 80-160°C for 4 to 48 hours, then cool, remove solvent, and purify the target compound via column chromatography.
Following the reaction period, which typically spans from 4 to 48 hours depending on the specific reactivity of the alcohol and substituents on the benzothiazole ring, the mixture is cooled to room temperature. Workup involves the removal of the solvent via rotary evaporation, followed by purification of the residue. In the patent examples, column chromatography using ethyl acetate and petroleum ether as the eluent system successfully isolated the target compounds as white or yellow solids with high purity. This operational simplicity makes the method highly attractive for scale-up, as it avoids the need for exotic reagents or complex equipment, allowing for seamless transition from gram-scale optimization to kilogram-scale production.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed alkylation technology presents a multitude of strategic benefits that directly impact the bottom line. The primary advantage stems from the drastic simplification of the raw material portfolio. By shifting from multi-step halogenated precursors to single-step alcohol alkylation, companies can reduce the number of SKUs they need to manage and qualify. Alcohols are commodity chemicals with stable pricing and abundant global supply, whereas specialized halogenated thioureas often require custom synthesis, leading to longer lead times and higher costs. This transition effectively decouples production from the volatility of the fine chemical halogen market, providing a more predictable cost structure for long-term manufacturing contracts.
- Cost Reduction in Manufacturing: The elimination of multi-step precursor synthesis and the use of catalytic rather than stoichiometric metal loads significantly lowers the cost of goods. Since the reaction generates water as the sole by-product, there is no need for expensive waste neutralization facilities required for hydrohalic acid disposal. This reduction in waste treatment overhead, combined with the high atom economy, results in substantial cost savings per kilogram of product. Furthermore, the ability to run the reaction under solvent-free conditions in certain embodiments removes the cost of solvent purchase, recovery, and disposal entirely, enhancing the overall economic viability of the process.
- Enhanced Supply Chain Reliability: Utilizing broadly available alcohols and simple inorganic bases like sodium hydroxide mitigates the risk of supply chain disruptions. Unlike proprietary or hard-to-source halogenated intermediates, these reagents are produced by numerous vendors worldwide, ensuring competitive pricing and consistent availability. This diversification of the supply base strengthens negotiation leverage and reduces the risk of production stoppages due to raw material shortages. Additionally, the robustness of the reaction conditions, which tolerate both air and nitrogen atmospheres, simplifies logistics by reducing the need for stringent inert gas handling infrastructure.
- Scalability and Environmental Compliance: The green chemistry credentials of this method facilitate easier regulatory approval and environmental compliance. With water as the only by-product, the environmental impact is minimal, aligning with increasingly strict global regulations on industrial emissions and waste. The process is inherently scalable, as demonstrated by the wide range of successful examples in the patent covering various substituted benzothiazoles and alcohols. This scalability ensures that the method can support commercial volumes from 100 kgs to 100 MT annual production without requiring fundamental changes to the reaction engineering, making it a future-proof solution for growing market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims within CN102120734A, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing workflows and assess its alignment with their specific product development goals.
Q: What are the primary environmental advantages of this new alkylation method compared to traditional cyclization routes?
A: Unlike conventional methods that generate hydrohalic acid by-products and require multi-step synthesis of o-halophenylthiourea, this novel approach uses environmentally friendly alcohols as alkylating agents. The only by-product generated is water, resulting in high atom economy (>93%) and eliminating the need for complex waste neutralization processes associated with halogenated waste streams.
Q: Which copper salts and bases are most effective for this transformation according to the patent data?
A: The patent data indicates that cuprous chloride (CuCl) is highly effective, often yielding over 90% conversion. Other viable copper sources include cuprous bromide and cuprous iodide. For the base component, strong inorganic bases such as sodium hydroxide and potassium hydroxide demonstrate superior performance compared to weaker carbonate bases, with sodium hydroxide frequently providing optimal yields in the range of 90-98%.
Q: Is this synthetic route scalable for commercial production of pharmaceutical intermediates?
A: Yes, the process is designed for scalability. It utilizes commercially available starting materials like 2-aminobenzothiazoles and common alcohols. The reaction can be performed under air or nitrogen protection and tolerates a wide range of solvents including toluene and xylene, which are standard in industrial settings. Furthermore, the ability to operate under solvent-free conditions in some embodiments further enhances its potential for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(N-alkyl)aminobenzothiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed alkylation technology described in CN102120734A for the production of high-value pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are fully realized in practical manufacturing settings. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the exacting standards required for drug substance synthesis, thereby minimizing risk for our partners.
We invite you to collaborate with us to leverage this efficient synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this method can optimize your supply chain economics. Please contact us to request specific COA data for our benzothiazole derivatives and to discuss route feasibility assessments that can accelerate your development timelines. Partnering with NINGBO INNO PHARMCHEM ensures access to cutting-edge chemistry backed by reliable supply and unwavering quality commitment.
