Scalable Synthesis of 2-Amino-5-Bromoisonicotinic Acid for Pharmaceutical Manufacturing
Scalable Synthesis of 2-Amino-5-Bromoisonicotinic Acid for Pharmaceutical Manufacturing
The development of efficient synthetic routes for heterocyclic building blocks remains a critical priority for the global pharmaceutical industry, particularly for intermediates used in kinase inhibitor development. Patent CN101863830A introduces a robust and economically viable methodology for the production of 2-amino-5-bromoisonicotinic acid, a pivotal scaffold in modern medicinal chemistry. This technical disclosure addresses the longstanding challenges associated with low yields and complex purification protocols found in traditional synthesis pathways. By leveraging a strategic sequence of bromination, protection, oxidation, and hydrolysis, the disclosed method achieves an overall yield exceeding 60 percent, representing a substantial improvement over historical benchmarks. The structural integrity and functional versatility of this pyridine derivative make it an essential component for constructing complex drug molecules, necessitating a supply chain capable of delivering high-purity material consistently.
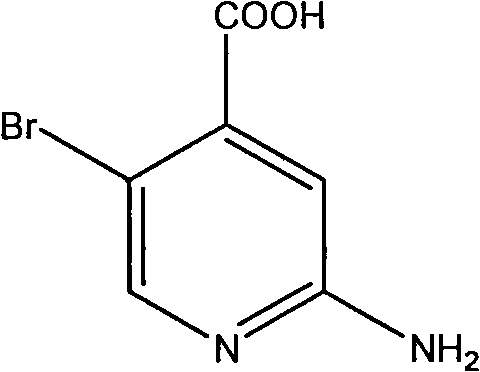
For procurement specialists and R&D leaders seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this route is paramount for long-term sourcing strategies. The ability to bypass inefficient esterification steps not only reduces raw material costs but also minimizes waste generation, aligning with modern green chemistry principles. As we analyze the technical specifics, it becomes evident that this process offers a scalable solution for cost reduction in pharmaceutical intermediates manufacturing. The transition from laboratory-scale discovery to commercial production requires methods that are both chemically elegant and operationally simple, criteria that this patent satisfies through its use of readily available reagents and straightforward isolation techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-5-bromoisonicotinic acid has been plagued by inefficiencies that drive up costs and limit supply reliability. Prior art, such as the methodologies described in international patent applications like WO2007/113226 and US2006/84802, typically relies on the bromination of pre-formed esters using reagents like N-bromosuccinimide (NBS) or elemental bromine. These conventional pathways suffer from critically low yields, often reported in the range of 20 to 35 percent for the bromination step alone. Furthermore, these routes necessitate an additional saponification step to convert the ester intermediate into the final carboxylic acid, which inevitably leads to further yield losses and increased processing time. The reliance on expensive brominating agents and the generation of stoichiometric amounts of succinimide byproducts create significant economic and environmental burdens for manufacturers.

The cumulative effect of these inefficiencies results in a total recovery rate that is commercially unattractive for large-scale API production. When the initial bromination yield is capped at roughly 35 percent, and subsequent hydrolysis steps incur further losses, the final throughput of the active pharmaceutical ingredient precursor becomes a bottleneck. For supply chain managers, this translates to higher unit costs and potential volatility in availability. The complexity of purifying the reaction mixture from succinimide residues and unreacted starting materials further complicates the manufacturing process, requiring extensive chromatography or recrystallization efforts that are difficult to scale. Consequently, the industry has urgently needed a more direct and high-yielding alternative to overcome these structural limitations in the supply chain.
The Novel Approach
The methodology disclosed in CN101863830A represents a paradigm shift by reversing the traditional order of operations and utilizing a more robust starting material, 2-amino-4-picoline. Instead of starting with an ester, this novel approach initiates the synthesis with a direct bromination of the picoline derivative in an acetic acid medium. This strategic change allows for the introduction of the bromine atom at the 5-position with significantly higher efficiency before the methyl group is oxidized. By avoiding the ester functionality during the critical bromination phase, the process eliminates the need for expensive esterification reagents and the associated yield penalties. The result is a streamlined workflow where the core pyridine skeleton is functionalized early, setting the stage for high-efficiency downstream transformations.
Furthermore, the integration of an acetylation step prior to oxidation serves a dual purpose: it protects the sensitive amino group from oxidative degradation and enhances the solubility characteristics of the intermediate. This protection strategy ensures that the subsequent oxidation with potassium permanganate proceeds cleanly to form the carboxylic acid without compromising the amine functionality. The final hydrolysis step is performed under mild alkaline conditions, which effectively removes the acetyl protecting group while precipitating the final product upon acidification. This sequence not only boosts the overall yield to greater than 60 percent but also simplifies the isolation process, as the final product can be obtained through straightforward filtration and washing, thereby drastically reducing solvent consumption and processing time.
Mechanistic Insights into Oxidative Functionalization and Protection Strategies
The core chemical innovation lies in the precise orchestration of protection and oxidation steps to maximize regioselectivity and yield. In the second step of the synthesis, the reaction of 2-amino-4-methyl-5-bromopyridine with acetic anhydride in the presence of 4-dimethylaminopyridine (DMAP) is critical. DMAP acts as a potent nucleophilic catalyst, accelerating the acetylation of the exocyclic amine to form the acetamido derivative. This protection is mechanistically essential because free amines on pyridine rings are susceptible to oxidation by strong oxidants like potassium permanganate, which could lead to N-oxide formation or ring degradation. By converting the amine to an acetamide, the electron density on the nitrogen is reduced, rendering it inert to the oxidative conditions required for the next transformation. This ensures that the oxidation is directed exclusively toward the benzylic methyl group at the 4-position.
Following protection, the oxidation step utilizes potassium permanganate in an aqueous medium, a classic yet highly effective method for converting methyl groups to carboxylic acids. The mechanism involves the sequential abstraction of hydrogen atoms from the methyl group, forming a radical intermediate that is further oxidized to an aldehyde and finally to the carboxylate salt. The use of water as a solvent in this step is particularly advantageous from a safety and cost perspective, eliminating the need for hazardous organic solvents during the most exothermic part of the reaction. The reaction temperature is carefully controlled between 60 and 100 degrees Celsius to ensure complete conversion while preventing thermal decomposition of the sensitive pyridine ring. The resulting manganese dioxide byproduct is easily removed by hot filtration, leaving a clear filtrate containing the desired acid salt, ready for the final deprotection and isolation.
How to Synthesize 2-Amino-5-Bromoisonicotinic Acid Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly temperature control during the initial bromination and the stoichiometry of the oxidant. The process begins with the dissolution of 2-amino-4-picoline in a mixture of acetic acid and dichloromethane, cooled to below 5 degrees Celsius to manage the exotherm of bromine addition. Maintaining this low temperature is crucial to prevent poly-bromination and ensure mono-substitution at the desired 5-position. Following the bromination and isolation of the solid intermediate, the acetylation is performed at elevated temperatures, preferably around 140 degrees Celsius, to drive the reaction to completion rapidly. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below for technical teams evaluating process feasibility.
- Brominate 2-amino-4-picoline in acetic acid at low temperature (-10 to 5°C) to form 2-amino-4-methyl-5-bromopyridine.
- Protect the amine group by reacting with acetic anhydride and DMAP at elevated temperatures (80-140°C) to yield 2-acetamido-4-methyl-5-bromopyridine.
- Oxidize the methyl group using potassium permanganate in aqueous solution at 60-100°C to form the carboxylic acid derivative.
- Hydrolyze the acetamido protecting group under alkaline conditions (pH 8-12) followed by acidification to isolate the final 2-amino-5-bromoisonicotinic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound benefits for organizations focused on cost reduction in pharmaceutical intermediates manufacturing. The most immediate impact is the dramatic increase in overall yield, which directly correlates to a lower cost of goods sold (COGS). By improving the yield from the typical 20-30 percent range of legacy methods to over 60 percent, manufacturers can effectively double the output from the same amount of raw materials. This efficiency gain is compounded by the elimination of expensive reagents like N-bromosuccinimide and the reduction in solvent usage due to simplified purification steps. For procurement managers, this translates into a more stable pricing structure and the ability to negotiate better terms with suppliers who utilize this superior technology.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of commodity chemicals such as acetic acid, bromine, and potassium permanganate, which are significantly cheaper than specialized brominating agents. The removal of the esterification and subsequent saponification steps reduces the number of unit operations, thereby lowering labor and energy costs. Additionally, the high purity of the crude product obtained after filtration minimizes the need for resource-intensive recrystallization or chromatography, further driving down operational expenses. These factors combine to create a lean manufacturing process that is highly resilient to fluctuations in raw material markets.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions and the availability of starting materials. 2-Amino-4-picoline is a widely produced bulk chemical, ensuring that supply bottlenecks are unlikely to occur. The process tolerance, demonstrated by the broad acceptable ranges for temperature and pH in the patent, indicates a forgiving manufacturing window that reduces the risk of batch failures. For supply chain heads, this reliability means shorter lead times and the confidence to commit to long-term supply agreements without the fear of production delays caused by finicky chemistry.
- Scalability and Environmental Compliance: The scalability of this route is evidenced by its reliance on heterogeneous reactions and simple filtration workups, which are easily translated from pilot plant to multi-ton production scales. The use of water as a primary solvent for the oxidation step significantly reduces the volume of volatile organic compounds (VOCs) emitted, aiding in compliance with stringent environmental regulations. The solid byproducts, such as manganese dioxide, can be managed and recycled more easily than liquid organic waste streams. This environmental profile makes the process attractive for production in regions with strict ecological standards, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this key intermediate. Understanding these details helps stakeholders assess the fit of this material within their specific development pipelines. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for decision-makers.
Q: What is the primary advantage of this synthesis method over prior art?
A: The primary advantage is the significantly improved overall yield (>60%) compared to conventional ester-based routes which typically suffer from yields below 35%. Additionally, it avoids the use of expensive esterification reagents and simplifies the purification process.
Q: How is the amino group protected during the oxidation step?
A: The amino group is protected via acetylation using acetic anhydride and DMAP prior to oxidation. This prevents unwanted side reactions and ensures the integrity of the pyridine ring during the harsh oxidative conditions required to convert the methyl group to a carboxylic acid.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common, inexpensive raw materials like 2-amino-4-picoline and standard oxidants like potassium permanganate. The reaction conditions are safe and reliable, making it highly suitable for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-5-Bromoisonicotinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of new drugs depends on the availability of high-quality intermediates produced via efficient routes. Our technical team has extensively analyzed the methodology described in CN101863830A and possesses the expertise to implement this high-yield synthesis at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-amino-5-bromoisonicotinic acid meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall development costs. By leveraging our advanced manufacturing capabilities, you can secure a stable source of this critical building block while benefiting from the economic advantages of the novel synthesis route. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project volume. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your transition from research to commercial manufacturing.
