Advanced Nitrogen-Phosphine Coordinated Trifluoromethoxylation Reagents for Commercial Scale-Up
Introduction to Next-Generation Trifluoromethoxylation Technology
The introduction of the trifluoromethoxy (-OCF3) group into organic molecules represents a critical strategy in modern medicinal chemistry and agrochemical design, significantly enhancing metabolic stability and lipophilicity. As highlighted in patent CN111217841B, filed in 2021, a breakthrough has been achieved with the development of a nitrogen-phosphine ligand coordination type trifluoromethoxylation reagent. This innovation addresses long-standing challenges in the field, such as the instability of traditional silver trifluoromethoxide sources and the difficulty in achieving high stereospecificity on aliphatic chains. The technology enables the direct transformation of primary or secondary p-nitrobenzenesulfonates into valuable trifluoromethoxy compounds with exceptional optical purity. For global procurement teams seeking a reliable pharmaceutical intermediate supplier, understanding this technological leap is essential for securing supply chains for next-generation APIs like Sonidegib Phosphate.
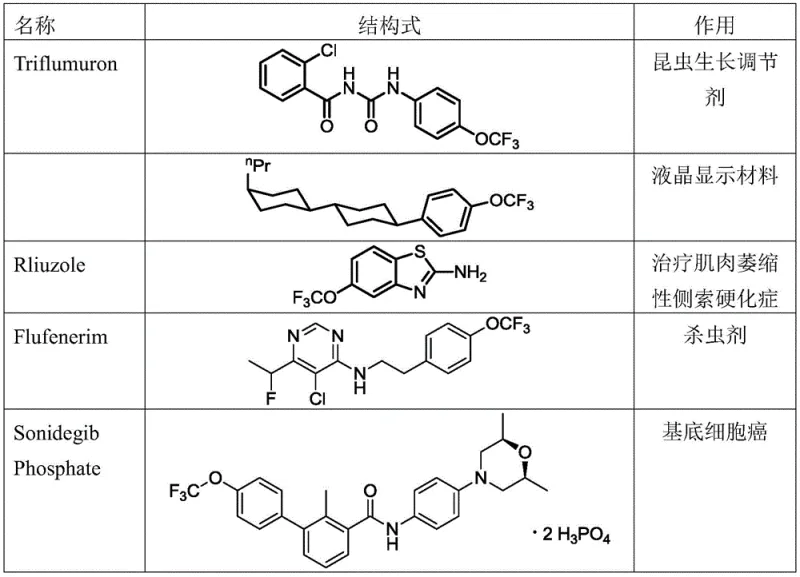
The strategic importance of this chemistry cannot be overstated, as the -OCF3 moiety is a privileged structure found in numerous FDA-approved drugs and advanced crop protection agents. However, historical methods for installing this group have been plagued by safety hazards, including the use of toxic gases like CF3OF, or poor reaction outcomes due to reagent decomposition. The new nitrogen-phosphine coordinated system offers a robust alternative, utilizing a stable silver complex that maintains reactivity while minimizing side reactions. This patent data provides a clear roadmap for the cost reduction in pharmaceutical intermediate manufacturing by streamlining synthetic routes that previously required multiple protection-deprotection steps or hazardous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
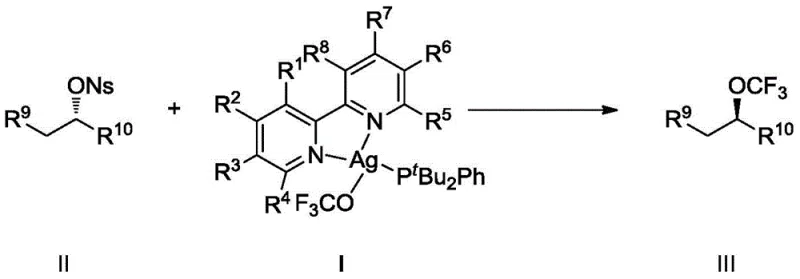
Traditional approaches to trifluoromethoxylation have faced significant hurdles that impede commercial scale-up of complex pharmaceutical intermediates. Historically, reagents such as AgOCF3 in acetonitrile have been thermodynamically unstable at room temperature, requiring low-temperature storage and immediate use, which complicates logistics and increases operational costs. Furthermore, alternative reagents like TASOCF3 suffer from similar stability issues and are prone to decomposition into fluorophosgene and fluoride anions. In nucleophilic substitution reactions, especially on secondary alkyl substrates, conventional methods often fail to control stereochemistry, leading to racemization or the formation of unwanted monofluoro by-products. Comparative examples in the patent data demonstrate that using simple phosphine ligands without nitrogen coordination (as seen in Comparative Example 1) results in complete failure to form the target fluorine signal, highlighting the inadequacy of prior art ligand systems.
The Novel Approach
The novel approach described in CN111217841B overcomes these deficiencies through the precise engineering of a nitrogen-phosphine ligand coordination environment around the silver center. By coordinating di-tert-butylphenylphosphine and specific bipyridine derivatives with AgOCF3, the resulting complex (Formula I) exhibits enhanced stability and reactivity. This system allows for the direct use of p-nitrobenzenesulfonates as substrates, which are superior leaving groups compared to halides or tosylates in this context. The method effectively suppresses the generation of monofluoro-substituted by-products, a common impurity that is difficult to remove and reduces overall yield. Consequently, this technology facilitates the production of high-purity OLED material precursors and drug intermediates with yields reaching up to 91% and enantiomeric excess (ee) values exceeding 90%, representing a substantial improvement over existing methodologies.
Mechanistic Insights into Nitrogen-Phosphine Coordinated SN2 Substitution
The core mechanism driving this transformation is a stereospecific SN2 nucleophilic substitution, facilitated by the unique electronic properties of the nitrogen-phosphine silver complex. The coordination of the bipyridine ligand to the silver atom modulates the electron density on the trifluoromethoxy group, making it a more effective nucleophile while preventing premature decomposition. In the reaction cycle, the silver center likely acts as a Lewis acid to activate the nosylate leaving group, while the stabilized -OCF3 moiety attacks the electrophilic carbon from the backside. This concerted mechanism ensures the inversion of configuration, which is critical for synthesizing chiral drugs where the wrong enantiomer can be inactive or toxic. The patent data explicitly notes that the reagent inhibits the formation of monofluoro by-products, suggesting that the coordination sphere prevents beta-fluorine elimination pathways that typically plague aliphatic fluorination reactions.
Impurity control is another critical aspect of this mechanism, particularly for R&D directors focused on regulatory compliance. The use of p-nitrobenzenesulfonate substrates provides a clean reaction profile, as the nosylate leaving group is stable under the reaction conditions but highly reactive towards the silver-activated nucleophile. Optimization studies within the patent reveal that reaction parameters such as temperature and additive loading (e.g., KI) play a pivotal role in maximizing stereospecificity. For instance, lowering the reaction temperature from 60°C to 40°C was shown to increase the ee value from 85.2% to 90%, demonstrating the fine-tunability of the process. This level of control allows manufacturers to reduce lead time for high-purity pharmaceutical intermediates by minimizing the need for extensive chiral chromatography downstream.
How to Synthesize Chiral Trifluoromethoxy Compounds Efficiently
The synthesis protocol outlined in the patent provides a reproducible method for generating these valuable building blocks. The process begins with the in-situ or pre-formed preparation of the silver reagent, followed by its reaction with the chiral substrate in an inert solvent like acetonitrile. The addition of a catalytic amount of potassium iodide serves to enhance the nucleophilicity, likely through a halogen exchange mechanism that generates a more reactive alkyl iodide intermediate in situ. Detailed experimental procedures in the patent examples describe working within a glove box to exclude moisture, which is essential for maintaining the integrity of the silver complex. The reaction mixture is typically heated in an oil bath, and progress is monitored via TLC or HPLC until the starting material is consumed. Following the reaction, standard workup procedures involving concentration and column chromatography yield the pure product.
- Preparation of the Reagent: React di-tert-butylphenylphosphine with AgOCF3 in toluene under inert atmosphere to form the silver dimer intermediate, then coordinate with a bipyridine ligand in acetonitrile at 40°C.
- Substrate Activation: Dissolve the chiral p-nitrobenzenesulfonate (nosylate) substrate and the prepared silver reagent in acetonitrile within a glove box to ensure moisture exclusion.
- Nucleophilic Substitution: Add catalytic potassium iodide and heat the mixture to 40-60°C for 8-48 hours to effect SN2 displacement, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nitrogen-phosphine coordinated reagent technology offers distinct strategic advantages beyond mere chemical efficacy. The primary benefit lies in the enhanced stability of the reagent compared to traditional silver trifluoromethoxide, which translates to reduced waste and lower inventory costs. Because the reagent can be isolated as a stable white solid and stored, it eliminates the need for just-in-time synthesis of unstable precursors, thereby de-risking the supply chain. Furthermore, the ability to use readily available p-nitrobenzenesulfonates as substrates simplifies the sourcing of starting materials, avoiding the need for specialized or hazardous alkylating agents. This streamlined workflow contributes to substantial cost savings in fine chemical manufacturing by reducing the number of unit operations and safety protocols required.
- Cost Reduction in Manufacturing: The high yields (up to 91%) and excellent stereoselectivity minimize the loss of expensive chiral starting materials. By suppressing by-product formation, the need for costly purification steps such as preparative HPLC is significantly reduced. Additionally, the mild reaction conditions (40-60°C) lower energy consumption compared to processes requiring cryogenic temperatures or high-pressure equipment. The elimination of toxic gases like CF3OF also removes the need for specialized scrubbing systems and hazardous waste disposal contracts, further optimizing the operational expenditure profile for large-scale production facilities.
- Enhanced Supply Chain Reliability: The robustness of the nitrogen-phosphine ligand system ensures consistent batch-to-batch quality, which is paramount for GMP manufacturing. The reagent's stability allows for bulk purchasing and warehousing, mitigating the risk of supply disruptions caused by the short shelf-life of conventional reagents. Moreover, the compatibility with standard solvents like acetonitrile and toluene means that existing reactor infrastructure can be utilized without major retrofitting. This flexibility enables suppliers to respond more rapidly to market demand fluctuations, ensuring a continuous flow of critical intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns with green chemistry principles by avoiding volatile and toxic reagents. The solid nature of the reagent reduces the risk of spills and exposure, improving workplace safety. The high atom economy of the SN2 reaction, combined with the recyclability of the nosylate by-product in some contexts, reduces the overall environmental footprint. Scalability is further supported by the straightforward workup procedure, which avoids complex aqueous quenches that generate large volumes of saline wastewater. These factors collectively make the process highly attractive for commercial scale-up of complex polymer additives and pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this trifluoromethoxylation technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation. Understanding these nuances is vital for process chemists evaluating the feasibility of integrating this method into their existing pipelines. The answers reflect the specific advantages of the nitrogen-phosphine coordination strategy over uncoordinated silver salts.
Q: How does this reagent improve stereoselectivity compared to traditional methods?
A: Unlike unstable silver trifluoromethoxide salts that decompose or lack selectivity, the nitrogen-phosphine coordination stabilizes the Ag-OCF3 bond, facilitating a clean SN2 inversion mechanism with ee values up to 91.6%.
Q: What types of substrates are compatible with this trifluoromethoxylation technology?
A: The method is specifically optimized for primary and secondary p-nitrobenzenesulfonates (nosylates), effectively suppressing monofluoro by-product formation common in aliphatic substitutions.
Q: Is the reagent stable enough for commercial storage and transport?
A: Yes, the coordination complex exhibits significantly improved thermal and thermodynamic stability compared to free AgOCF3, allowing for isolation as a white solid and handling under standard inert conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethoxylation Reagent Supplier
As the demand for fluorinated building blocks continues to surge across the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO is crucial for successful project execution. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our rigorous QC labs and stringent purity specifications guarantee that every batch of trifluoromethoxy intermediates meets the highest international standards, supporting your regulatory filings and clinical trials with confidence. We are committed to leveraging advanced technologies like the nitrogen-phosphine coordinated reagents to deliver superior value to our global partners.
We invite you to engage with our technical procurement team to discuss how this innovative chemistry can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this stable reagent system. We encourage potential clients to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your supply chain with reliable, high-quality fluorinated intermediates. Let us collaborate to accelerate your drug development timeline with cutting-edge synthetic solutions.
