Advanced Manufacturing of Chlorantraniliprole Key Intermediate via Green Chemistry and Process Optimization
Advanced Manufacturing of Chlorantraniliprole Key Intermediate via Green Chemistry and Process Optimization
The global demand for high-efficiency, low-toxicity insecticides has driven significant innovation in the synthesis of diamide class pesticides, particularly chlorantraniliprole. Patent CN102311424A presents a breakthrough methodology for synthesizing the critical intermediate 3-bromo-1-(3-chloropyridin-2-yl)-1H-pyrazole-5-carbonyl chloride, addressing long-standing challenges in yield, environmental impact, and operational safety. This technical insight report analyzes the proprietary six-step synthetic route which utilizes 2,3-dichloropyridine as a starting material, undergoing hydrazination, cyclization, bromination, oxidation, hydrolysis, and acyl chloride formation. The strategic replacement of traditional reagents with phosphorus pentabromide and solid phosgene marks a pivotal shift towards greener chemistry in agrochemical manufacturing. For R&D directors and procurement specialists, understanding this pathway is essential for securing a reliable agrochemical intermediate supplier capable of delivering high-purity materials with consistent quality.
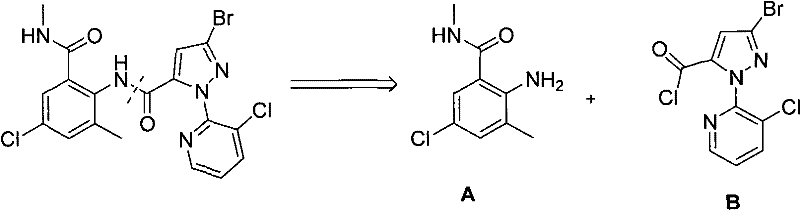
The retrosynthetic logic illustrated above underscores the strategic importance of Intermediate B in the overall value chain of chlorantraniliprole production. By optimizing the synthesis of this specific pyrazole derivative, manufacturers can significantly influence the cost structure and supply stability of the final active pharmaceutical ingredient (API). The patent details a comprehensive approach that not only improves chemical efficiency but also aligns with modern regulatory standards regarding waste reduction and hazard minimization. This makes the technology particularly attractive for companies seeking cost reduction in agrochemical intermediates manufacturing without compromising on the stringent purity specifications required for crop protection products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing this key intermediate have historically suffered from severe operational constraints that hinder large-scale commercial viability. For instance, earlier patents such as WO03106427 and WO2004033468 describe routes relying on N,N-dimethylsulfamoylpyrazole as a starting material, which necessitates the use of specialized and expensive reagents like LDA (lithium diisopropylamide) and TFA (trifluoroacetic acid). These processes often require cryogenic conditions as low as -78°C, imposing heavy burdens on refrigeration infrastructure and energy consumption. Furthermore, alternative routes utilizing maleic anhydride, as reported in WO2004087689, involve excessive synthetic steps and demonstrate low overall yields, making them economically unfeasible for mass production. The reliance on such harsh conditions and costly inputs creates significant bottlenecks in the supply chain, leading to extended lead times and increased vulnerability to raw material price fluctuations.
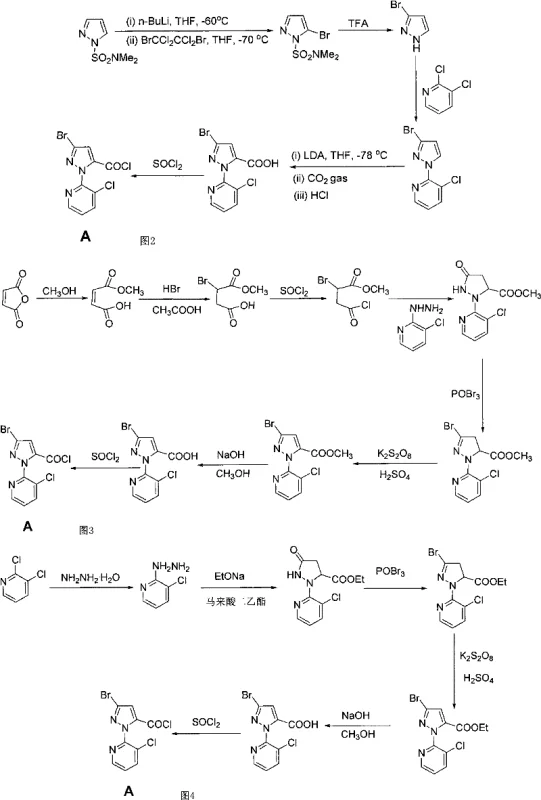
The Novel Approach
In stark contrast, the novel approach detailed in CN102311424A leverages a more accessible starting material, 2,3-dichloropyridine, combined with diethyl maleate, to establish a robust and scalable foundation. This method eliminates the need for extreme cryogenic temperatures and expensive organometallic bases, thereby drastically simplifying the equipment requirements and operational complexity. The innovation lies not just in the starting materials but in the specific reagent choices for critical transformation steps, specifically the bromination and acylation stages. By substituting phosphorus oxybromide with phosphorus pentabromide and replacing thionyl chloride with solid phosgene, the process achieves superior reaction control and product quality. This strategic pivot enables the commercial scale-up of complex pyrazole derivatives with a significantly reduced environmental footprint, offering a compelling value proposition for supply chain heads focused on continuity and sustainability.
Mechanistic Insights into PBr5 Bromination and Solid Phosgene Acylation
The core chemical innovation of this patent resides in the optimization of the bromination and acylation steps, which are traditionally prone to side reactions and waste generation. In the bromination stage, the use of phosphorus pentabromide (PBr5) instead of the conventional phosphorus oxybromide (POBr3) facilitates a more efficient electrophilic substitution at the pyrazole ring. This reagent swap enhances the conversion rate and minimizes the formation of phosphorus-containing byproducts that are difficult to separate, thus streamlining the downstream purification process. The mechanistic advantage of PBr5 ensures that the bromine atom is introduced with high regioselectivity, preserving the integrity of the sensitive pyridine moiety attached to the nitrogen atom. This precision is critical for maintaining the biological activity of the final insecticide and ensuring that the impurity profile meets rigorous international standards.
Furthermore, the acylation step employs solid phosgene (triphosgene) as a safer and more manageable alternative to gaseous phosgene or thionyl chloride. Solid phosgene decomposes in situ to generate phosgene under controlled conditions, allowing for a homogeneous reaction environment that promotes high yields of the acid chloride. This method avoids the generation of sulfur dioxide and hydrogen chloride gases associated with thionyl chloride, which are corrosive and require extensive scrubbing systems. The result is a cleaner reaction mixture that simplifies isolation and reduces the load on wastewater treatment facilities. For technical teams, this translates to a process that is not only chemically elegant but also operationally superior, reducing the risk of equipment corrosion and enhancing the overall safety profile of the manufacturing plant.
How to Synthesize 3-Bromo-1-(3-chloropyridin-2-yl)-1H-pyrazole-5-carbonyl Chloride Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for reproducing this high-yield pathway in a pilot or production setting. The process begins with the hydrazination of 2,3-dichloropyridine in ethylene glycol, followed by cyclization with diethyl maleate in the presence of sodium ethoxide. Subsequent steps involve the critical bromination with PBr5, oxidative aromatization using potassium persulfate, ester hydrolysis, and final activation with solid phosgene. Each step has been optimized to maximize recovery and minimize solvent usage, reflecting a design philosophy centered on efficiency. The detailed standardized synthetic steps see the guide below for specific reaction conditions and stoichiometry required to achieve the reported yields of up to 98.6% in the final step.
- Hydrazination of 2,3-dichloropyridine with hydrazine hydrate in ethylene glycol to form 3-chloro-2-hydrazinopyridine.
- Cyclization with diethyl maleate in sodium ethoxide to form the pyrazolidinone ester intermediate.
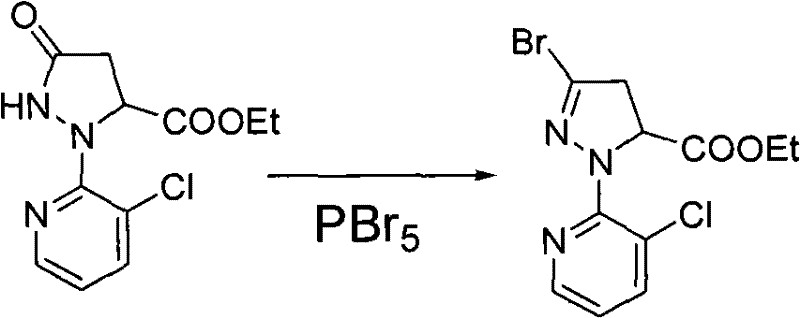
- Bromination using phosphorus pentabromide (PBr5) in acetonitrile to introduce the bromine atom at the 3-position.
- Oxidative aromatization using potassium persulfate and sulfuric acid to form the pyrazole ring.
- Hydrolysis of the ester group using sodium hydroxide followed by acidification to obtain the carboxylic acid.
- Acyl chloride formation using solid phosgene (triphosgene) in dichloromethane with DMF catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical curiosity. The elimination of cryogenic requirements and special reagents like LDA directly translates to lower capital expenditure on specialized equipment and reduced energy costs for cooling. By utilizing commodity chemicals such as 2,3-dichloropyridine and diethyl maleate, the process mitigates the risk of supply disruptions associated with niche starting materials. This stability is crucial for maintaining consistent production schedules and meeting the demanding delivery timelines of the global agrochemical market. Additionally, the improved yield and purity reduce the need for extensive reprocessing, further driving down the cost of goods sold (COGS) and enhancing overall margin potential.
- Cost Reduction in Manufacturing: The substitution of expensive and hazardous reagents with cost-effective alternatives like solid phosgene and PBr5 leads to substantial cost savings in raw material procurement. The avoidance of low-temperature operations removes the energy burden of maintaining -78°C environments, which is a significant operational expense in traditional syntheses. Moreover, the higher yields achieved at each step mean less raw material is wasted, optimizing the atom economy of the entire process. These factors combine to create a leaner manufacturing model that is highly competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified by the reliance on widely available bulk chemicals rather than specialized intermediates that may have limited suppliers. The robustness of the reaction conditions ensures that production is less susceptible to minor variations in temperature or reagent quality, leading to more predictable output volumes. This reliability allows supply chain planners to maintain lower safety stock levels while still guaranteeing on-time delivery to customers. Consequently, the working capital tied up in inventory can be reduced, improving the overall financial health of the operation.
- Scalability and Environmental Compliance: The process is inherently designed for scale, with simple workup procedures such as filtration and extraction that are easily adapted to large reactors. The reduction in hazardous waste, particularly the avoidance of sulfur-containing byproducts and phosphorus sludge, simplifies compliance with increasingly strict environmental regulations. This "green" credential is becoming a key differentiator in the industry, allowing companies to market their products as sustainably produced. The ease of waste treatment also lowers the overhead costs associated with environmental management and disposal services.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific innovations and data points presented in the patent documentation, providing clarity on yield expectations, safety protocols, and scalability factors. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this technology into their existing production lines or sourcing strategies.
Q: What are the primary advantages of using solid phosgene over thionyl chloride in this synthesis?
A: The use of solid phosgene (triphosgene) significantly improves operational safety and environmental compliance compared to gaseous phosgene or thionyl chloride. It generates less corrosive waste, simplifies the workup procedure, and results in higher product purity by minimizing side reactions associated with sulfur-containing byproducts.
Q: How does the PBr5 bromination method compare to traditional POBr3 methods?
A: Replacing phosphorus oxybromide (POBr3) with phosphorus pentabromide (PBr5) enhances the reaction efficiency and yield. This substitution allows for milder reaction conditions and reduces the generation of difficult-to-treat phosphorus waste, contributing to a more sustainable manufacturing process suitable for large-scale production.
Q: Is this synthetic route scalable for industrial production of agrochemical intermediates?
A: Yes, the route is specifically designed for industrial scalability. By avoiding cryogenic conditions (such as -78°C required in prior art) and utilizing readily available raw materials like 2,3-dichloropyridine, the process offers robust commercial scale-up potential with reduced equipment constraints and lower operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Bromo-1-(3-chloropyridin-2-yl)-1H-pyrazole-5-carbonyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern agrochemical formulations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-bromo-1-(3-chloropyridin-2-yl)-1H-pyrazole-5-carbonyl chloride meets the highest industry standards. Our commitment to green chemistry aligns perfectly with the innovations described in CN102311424A, allowing us to offer a product that is both economically and environmentally superior.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific application. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential ROI of switching to this optimized manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless transition to a more efficient and reliable supply chain partner.
