Optimizing Rosuvastatin Calcium Production For Commercial Scale And Supply Chain Reliability
The pharmaceutical landscape for lipid-lowering agents continues to evolve, with Rosuvastatin Calcium remaining a cornerstone therapy for managing hypercholesterolemia and preventing cardiovascular events. The technical efficiency of its manufacturing process directly impacts global availability and cost structures for healthcare systems. A pivotal advancement in this domain is documented in patent CN102219749B, which outlines a robust preparation method designed to overcome the limitations of earlier synthetic routes. This patent details a sequence involving condensation, deprotection, stereoselective reduction, and final salt formation, emphasizing stable yields and the use of cost-effective reagents. For R&D directors and procurement specialists, understanding the nuances of this specific pathway is critical for evaluating potential supply partners and optimizing their own production pipelines. The method described offers a compelling alternative to traditional routes that often suffer from expensive reagents or harsh reaction conditions, thereby presenting a significant opportunity for cost reduction in API manufacturing and enhanced supply chain reliability for global pharmaceutical companies seeking a reliable Rosuvastatin Calcium supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rosuvastatin Calcium has been approached through various pathways, many of which present significant challenges for large-scale commercial production. Conventional methods often rely on Wittig reactions or similar olefination strategies that require expensive phosphorus ylides or specialized catalysts, driving up the raw material costs substantially. Furthermore, many prior art processes involve harsh reaction conditions, such as extreme temperatures or the use of hazardous solvents, which complicate safety protocols and increase the burden on environmental compliance teams. The impurity profiles generated by these older methods can be complex, necessitating extensive and costly purification steps to meet the stringent purity specifications required for active pharmaceutical ingredients. Additionally, the reliance on transition metal catalysts in some traditional routes introduces the risk of heavy metal contamination, requiring additional downstream processing to ensure patient safety. These factors collectively contribute to longer lead times for high-purity Rosuvastatin Calcium and reduce the overall economic viability of the manufacturing process, creating bottlenecks for procurement managers aiming to secure cost-effective supply chains.
The Novel Approach
In contrast, the methodology disclosed in CN102219749B introduces a streamlined approach that addresses many of these historical inefficiencies. This novel route utilizes a condensation reaction between 4-(4-fluorophenyl)-6-isopropyl-2-chloropyrimidine-5-carbaldehyde and a specific phosphorus ylide reagent, followed by a series of controlled transformations. The process is characterized by the use of inexpensive and readily available reagents, which significantly lowers the barrier to entry for production and reduces the overall cost of goods sold. The reaction conditions are milder and easier to operate, facilitating a smoother scale-up from laboratory bench to industrial reactor without the need for specialized equipment. By avoiding the use of expensive transition metal catalysts in key steps, the process simplifies the purification workflow and minimizes the risk of metal residues in the final product. This approach not only enhances the economic feasibility of production but also aligns with modern green chemistry principles by reducing waste and energy consumption. The stability of the yield across multiple steps ensures a predictable output, which is crucial for supply chain heads managing inventory and delivery schedules for complex polymer additives or pharmaceutical intermediates.
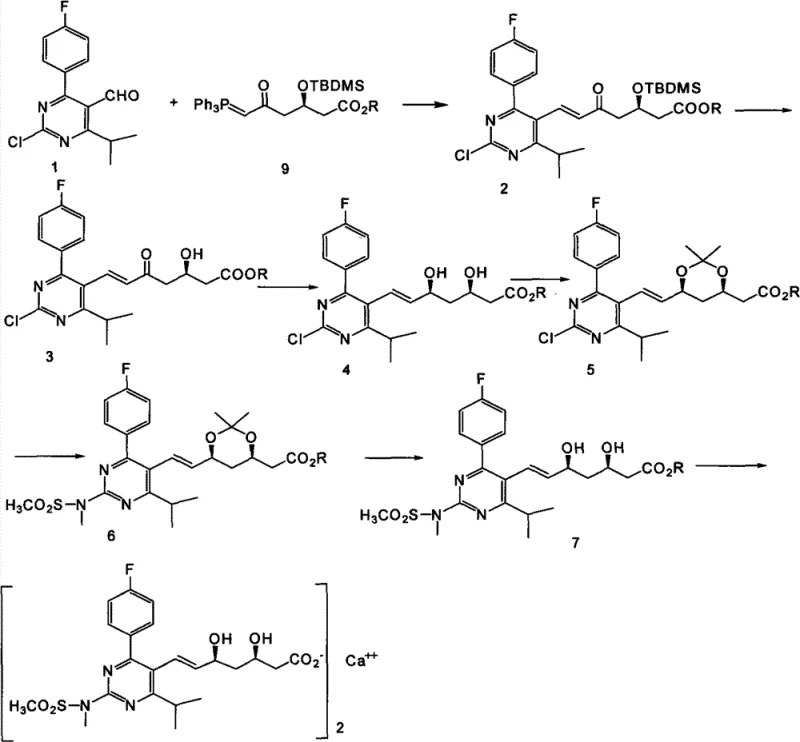
Mechanistic Insights into Stereoselective Reduction and Impurity Control
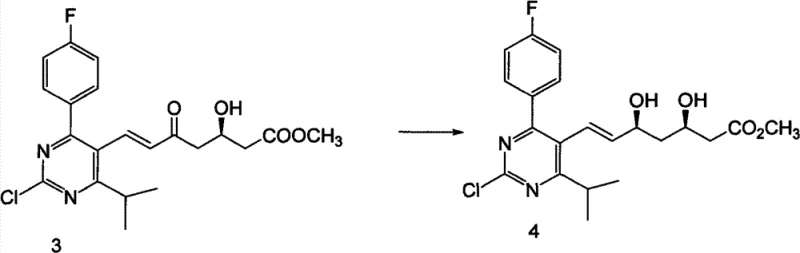
A critical aspect of the Rosuvastatin Calcium synthesis is the establishment of the correct stereochemistry at the 3R and 5S positions, which is essential for the drug's biological activity and safety profile. The patent describes a sophisticated stereoselective reduction step where a keto intermediate is reduced in the presence of organoboron reagents and sodium borohydride. This specific combination of reagents allows for precise control over the stereochemical outcome, ensuring high diastereoselectivity and minimizing the formation of unwanted isomers. The use of anhydrous solvents and controlled low temperatures during this reduction phase is paramount to maintaining the integrity of the chiral centers. From an R&D perspective, this mechanistic detail highlights the importance of process parameters in achieving consistent quality. The ability to control the reduction so precisely reduces the burden on downstream chromatography or crystallization steps, which are often the most costly parts of API manufacturing. This level of control is indicative of a mature process that has been optimized for both quality and efficiency, providing confidence to technical teams evaluating the feasibility of commercial scale-up of complex pharmaceutical intermediates.
Impurity control is another vital component of this synthesis, particularly given the regulatory scrutiny applied to statin medications. The pathway outlined in the patent incorporates specific protection and deprotection strategies, such as the use of silyl ether protecting groups and acetonide formation, to shield reactive functional groups during intermediate steps. This strategic masking prevents side reactions that could lead to difficult-to-remove impurities or degradation products. For instance, the protection of the diol system as an acetonide ensures that subsequent reactions, such as the amination with N-methylmethanesulfonamide, proceed cleanly without affecting the hydroxyl groups. The final deprotection and hydrolysis steps are carefully managed to ensure complete removal of protecting groups without inducing epimerization or decomposition. This rigorous approach to impurity management results in a cleaner crude product, which simplifies the final purification process and ensures that the Rosuvastatin Calcium meets the high-purity standards expected by regulatory bodies. For procurement managers, this translates to a lower risk of batch rejection and a more reliable supply of high-purity OLED material or pharmaceutical grade products.
How to Synthesize Rosuvastatin Calcium Efficiently
Implementing this synthesis route requires a clear understanding of the sequential chemical transformations and the specific operational parameters defined in the patent. The process begins with the condensation of the pyrimidine aldehyde and the phosphorus ylide, followed by deprotection and the critical stereoselective reduction. Subsequent steps involve protection of the diol, amination, deprotection, hydrolysis, and finally, salt formation with a calcium source. Each step must be monitored closely, typically using techniques like TLC or HPLC, to ensure complete conversion before proceeding to the next stage. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety considerations. Adhering to these protocols is essential for reproducing the stable yields and high purity reported in the patent documentation.
- Condensation of 4-(4-fluorophenyl)-6-isopropyl-2-chloropyrimidine-5-carbaldehyde with a phosphorus ylide reagent to form the key olefin intermediate.
- Stereoselective reduction of the keto group using organoboron reagents and sodium borohydride to establish the critical 3R,5S chiral centers.
- Final deprotection, hydrolysis, and calcium salt formation to yield the target Rosuvastatin Calcium API with stringent purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical advantages of this synthesis route translate directly into tangible commercial benefits. The use of inexpensive and readily available reagents significantly reduces the raw material costs associated with production, allowing for more competitive pricing structures in the global market. The simplicity of the operation and the mild reaction conditions lower the capital expenditure required for manufacturing facilities, as there is no need for specialized high-pressure or high-temperature equipment. This accessibility facilitates a broader base of potential suppliers, enhancing supply chain resilience and reducing the risk of disruptions. Furthermore, the stable yields reported in the patent ensure a predictable production volume, which is crucial for long-term supply planning and inventory management. The elimination of expensive transition metal catalysts not only reduces costs but also simplifies the regulatory filing process by minimizing the need for extensive heavy metal testing and clearance. These factors collectively contribute to a more robust and cost-effective supply chain for Rosuvastatin Calcium.
- Cost Reduction in Manufacturing: The process utilizes cheap reagents and avoids expensive catalysts, leading to substantial cost savings in raw materials and downstream processing. The simplified purification workflow further reduces operational expenses by minimizing solvent usage and energy consumption. This economic efficiency allows manufacturers to offer more competitive pricing without compromising on quality, providing significant value to procurement teams looking to optimize their budget allocations for API sourcing.
- Enhanced Supply Chain Reliability: The use of common organic solvents and readily available starting materials ensures that the supply chain is not vulnerable to shortages of specialized or exotic chemicals. The robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites, reducing the risk of batch failures. This reliability is essential for maintaining continuous supply to pharmaceutical customers and avoiding costly production delays that can impact patient access to medication.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward workup procedures make this process highly scalable from pilot plant to commercial production. The reduced use of hazardous reagents and the generation of less toxic waste align with increasingly strict environmental regulations, minimizing the environmental footprint of the manufacturing process. This compliance reduces the regulatory burden on manufacturers and ensures long-term sustainability of the production facility, which is a key consideration for supply chain heads evaluating potential partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the Rosuvastatin Calcium synthesis process described in CN102219749B. These answers are derived from the patent's technical disclosures and are intended to provide clarity on the process capabilities and advantages. Understanding these details is crucial for stakeholders evaluating the feasibility of this route for their specific manufacturing needs. The information provided here serves as a foundational guide for further technical discussions and feasibility assessments.
Q: What are the key advantages of the CN102219749B synthesis route?
A: The method described in patent CN102219749B offers stable yields and utilizes inexpensive reagents, making it highly suitable for industrialized production compared to conventional methods that rely on costly catalysts or harsh conditions.
Q: How is stereochemistry controlled in this Rosuvastatin process?
A: Stereochemistry is rigorously controlled through a specific stereoselective reduction step using organoboron reagents and sodium borohydride at low temperatures, ensuring the correct 3R,5S configuration essential for biological activity.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the process is designed for scalability with easy operation steps, mild reaction conditions, and the use of common organic solvents, facilitating a smooth transition from laboratory scale to commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Supplier
The synthesis route detailed in CN102219749B represents a significant advancement in the production of Rosuvastatin Calcium, offering a balance of efficiency, cost-effectiveness, and quality that is essential for modern pharmaceutical manufacturing. NINGBO INNO PHARMCHEM, as a leading CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this process to life on an industrial scale. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of Rosuvastatin Calcium meets the highest global standards. We understand the critical nature of API supply and are committed to providing a reliable partnership that supports your long-term commercial goals. Our technical team is ready to assist in optimizing this pathway for your specific production requirements, ensuring a seamless transition from development to commercialization.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of adopting this method for your production needs. We encourage you to reach out for specific COA data and route feasibility assessments to validate the compatibility of this process with your existing manufacturing infrastructure. Our team is dedicated to providing the technical support and commercial flexibility needed to secure a stable and cost-effective supply of high-quality Rosuvastatin Calcium for your global operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
