Advanced Four-Step Synthesis of 3,7-Diethyl Nonane-4,6-Diketone for Electronic Coatings
Advanced Four-Step Synthesis of 3,7-Diethyl Nonane-4,6-Diketone for Electronic Coatings
The landscape of electronic component coating manufacturing is constantly evolving, driven by the demand for higher purity intermediates and more sustainable production methodologies. A significant breakthrough in this domain is detailed in patent CN108383705B, which discloses a robust preparation method for 3,7-diethyl nonane-4,6-diketone. This specific compound serves as a critical precursor in the formulation of advanced electronic coatings, where structural integrity and chemical consistency are paramount. The patented technology shifts the synthetic paradigm from legacy multi-step processes to a streamlined four-step sequence originating from low-cost dimethyl isophthalate. This strategic pivot not only addresses the economic inefficiencies of prior art but also resolves persistent purification challenges that have long plagued R&D teams. By leveraging a combination of Grignard addition, catalytic hydrogenation, Birch reduction, and controlled ozonolysis, the process delivers a high-purity product suitable for sensitive electronic applications. For procurement specialists and supply chain managers, this innovation represents a tangible opportunity for cost reduction in electronic chemical manufacturing without compromising on quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing 3,7-diethylnonane-4,6-dione have been fraught with operational complexities and yield limitations that hinder commercial viability. As early as 1951, Eugene H. Man et al. reported a six-step reaction pathway utilizing 2-ethylbutyric acid as the starting material. While conceptually straightforward, this legacy method suffers from severe downstream processing issues, particularly regarding the purification of intermediate 6, which possesses a low boiling point that complicates isolation. Furthermore, the final step of this conventional route generates phenol as a byproduct, a contaminant that is notoriously difficult to remove completely from the final matrix. The presence of such impurities can be detrimental to the performance of electronic coatings, necessitating expensive and time-consuming purification protocols. Similarly, a contemporaneous method by Braendstroem et al. attempted to shorten the sequence to three steps but introduced harsh reaction conditions that resulted in substantially lower yields. These archaic methodologies rely on starting materials and conditions that are no longer aligned with modern green chemistry principles or cost-effective manufacturing goals.
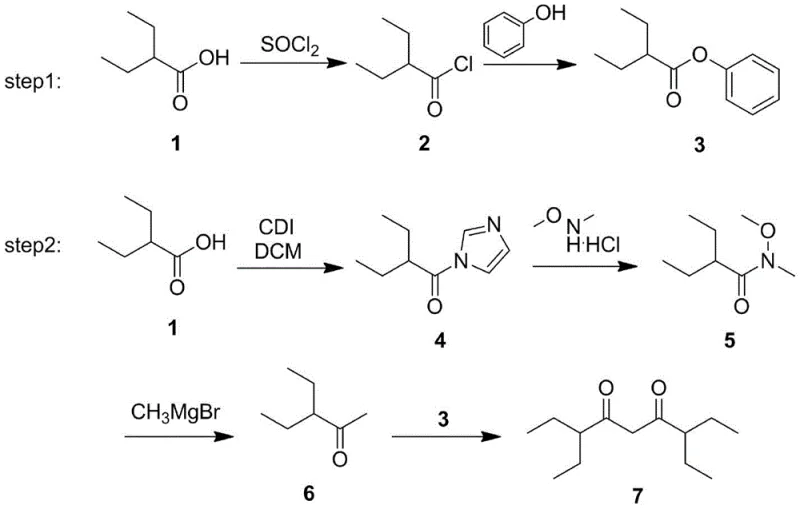
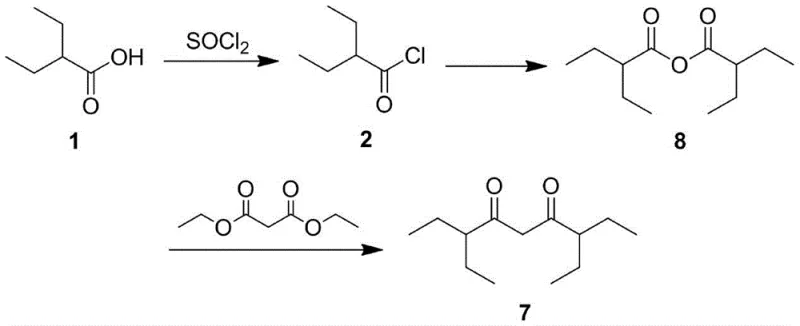
The Novel Approach
In stark contrast to these historical precedents, the novel approach disclosed in the patent utilizes dimethyl isophthalate, a readily available and inexpensive aromatic diester, as the foundational building block. This new strategy condenses the synthesis into just four distinct chemical transformations, drastically simplifying the operational workflow. The process begins with a controlled Grignard reaction, followed by a catalytic hydrogenation step that efficiently removes hydroxyl functionalities. Subsequent Birch reduction and ozonolysis steps are carefully tuned to maximize the formation of the target diketone structure while minimizing side reactions. This modernization of the synthetic route eliminates the generation of difficult-to-remove phenolic byproducts and avoids the handling of low-boiling intermediates that characterized the 1951 methods. For a reliable electronic chemical supplier, adopting this methodology translates to a more robust supply chain with fewer bottlenecks and a significantly reduced environmental footprint due to the elimination of complex waste streams associated with phenol removal.
Mechanistic Insights into the Four-Step Cascade Synthesis
The core of this technological advancement lies in the precise orchestration of four distinct reaction mechanisms, each optimized to ensure high conversion and selectivity. The initial step involves the nucleophilic addition of a Grignard reagent, specifically ethyl magnesium bromide, to dimethyl isophthalate at a controlled temperature range of 0-5°C. This exothermic reaction is critical; maintaining the temperature prevents runaway conditions and ensures the formation of the tertiary alcohol intermediate, 3,3'-(1,3-phenylene)bis(3-pentanol), with high fidelity. Following this, the process employs catalytic hydrogenation using palladium on carbon in an acidic medium (acetic acid) at elevated temperatures around 100°C. This step is mechanistically fascinating as it facilitates the deoxygenation of the benzylic alcohols to yield 1,3-di(3-pentyl)benzene, effectively setting the carbon skeleton for the final cleavage. The subsequent Birch reduction utilizes lithium metal in liquid ammonia at cryogenic temperatures (-40°C to -35°C) to partially reduce the aromatic ring to a 1,4-cyclohexadiene system. This specific reduction pattern is crucial as it positions the double bonds perfectly for the final oxidative cleavage.
The final transformation involves ozonolysis, a powerful oxidative cleavage reaction performed on a silica gel support at temperatures as low as -78°C. This heterogeneous approach to ozonolysis is a key innovation, as it allows for better heat dissipation and control over the highly reactive ozone species. The cleavage of the double bonds in the cyclohexadiene intermediate generates the desired 1,4-diketone functionality directly. The use of dimethyl sulfide as a quenching agent ensures the safe reduction of ozonides to the final ketone products without over-oxidation to carboxylic acids. From an impurity control perspective, this mechanism is superior because the silica gel support helps adsorb polar byproducts, and the specific reaction conditions minimize the formation of polymeric tars often seen in solution-phase ozonolysis. This level of mechanistic control is essential for producing high-purity OLED material or electronic coating precursors where trace metallic or organic impurities can degrade device performance.
How to Synthesize 3,7-Diethyl Nonane-4,6-Diketone Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding stoichiometry, temperature, and solvent systems to achieve the reported yields. The process is designed to be scalable, moving from laboratory glassware to industrial reactors with minimal modification to the fundamental chemistry. Operators must pay particular attention to the moisture sensitivity of the Grignard reagent and the cryogenic requirements of the Birch reduction to ensure safety and reproducibility. The following guide outlines the standardized operational framework derived from the patent examples, serving as a blueprint for process engineers aiming to replicate this high-efficiency route. Detailed standard operating procedures for each unit operation are provided below to facilitate immediate technology transfer.
- Perform Grignard addition of ethyl magnesium bromide to dimethyl isophthalate at 0-5°C to form 3,3'-(1,3-phenylene)bis(3-pentanol).
- Execute catalytic hydrogenation using Pd/C in acetic acid at 100°C to convert the bis-alcohol to 1,3-di(3-pentyl)benzene.
- Conduct Birch reduction with lithium in liquid ammonia at -40°C to generate the cyclohexadiene mixture.
- Finalize with ozonolysis on silica gel at -78°C followed by reductive workup to yield the target diketone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of sourcing complex fine chemical intermediates. The shift to dimethyl isophthalate as a starting material leverages a commodity chemical with a stable global supply chain, mitigating the risks associated with sourcing specialized or scarce reagents. This foundational change drives significant cost optimization throughout the manufacturing value chain. Furthermore, the reduction in synthetic steps from six or more down to four inherently lowers the cumulative loss of material, improving the overall mass balance and reducing the cost of goods sold. The elimination of difficult purification steps, such as the removal of phenol, further reduces the consumption of solvents and energy, contributing to a leaner and more sustainable production model. These factors combined make the technology highly attractive for long-term supply agreements.
- Cost Reduction in Manufacturing: The utilization of low-cost dimethyl isophthalate replaces expensive or inefficient starting materials like 2-ethylbutyric acid derivatives, leading to substantial raw material savings. Additionally, the streamlined four-step sequence reduces the number of isolation and purification stages, which significantly lowers labor costs and solvent consumption. The avoidance of complex distillation setups for low-boiling intermediates further decreases capital expenditure and operational overhead. By eliminating the need for extensive post-reaction cleanup to remove phenolic byproducts, the process achieves a more efficient use of resources, translating to a lower price point for the final high-purity electronic chemical.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit greatly from the use of widely available bulk chemicals, ensuring that production schedules are not disrupted by raw material shortages. The robustness of the reaction conditions, particularly the tolerance achieved in the hydrogenation and ozonolysis steps, ensures consistent batch-to-batch quality, which is critical for maintaining trust with downstream electronics manufacturers. The simplified workflow reduces the lead time for high-purity electronic chemical intermediates, allowing for faster response to market demands. Moreover, the scalability of the process means that suppliers can rapidly ramp up production volumes to meet surges in demand for electronic component coatings without requiring entirely new infrastructure.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor types for hydrogenation and extraction that are common in fine chemical plants. The use of silica gel-supported ozonolysis enhances safety by mitigating the risks associated with handling large quantities of explosive ozonides in solution. From an environmental standpoint, the reduction in waste generation, particularly the absence of phenolic waste streams, simplifies effluent treatment and ensures compliance with stringent environmental regulations. This green chemistry profile aligns with the sustainability goals of major multinational corporations, making the supply chain more resilient against regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation. Understanding these nuances is vital for R&D directors evaluating the feasibility of in-house production versus outsourcing. The answers highlight the specific improvements in yield and purity that distinguish this method from historical alternatives.
Q: What are the primary advantages of using dimethyl isophthalate over 2-ethylbutyric acid?
A: Dimethyl isophthalate is a low-cost commodity chemical that avoids the complex purification issues associated with the low-boiling intermediates and phenol byproducts found in traditional 2-ethylbutyric acid routes.
Q: How does the new process improve yield compared to historical methods?
A: By utilizing a streamlined four-step sequence with optimized conditions such as controlled ozonolysis on silica gel, the process achieves significantly higher overall yields compared to the multi-step, low-yield protocols reported in 1951.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method employs standard unit operations like hydrogenation and extraction with common solvents, making it highly scalable and safe for commercial manufacturing of electronic chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,7-Diethyl Nonane-4,6-Diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the advancement of electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify every batch. Our expertise in managing complex chemistries, such as the Birch reduction and ozonolysis steps described in this patent, allows us to offer a stable and high-quality supply of 3,7-diethyl nonane-4,6-diketone. We understand that consistency is key in the electronics industry, and our manufacturing protocols are designed to minimize variability.
We invite you to collaborate with us to optimize your supply chain for electronic component coatings. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage potential partners to contact us to request specific COA data and route feasibility assessments for your projects. By leveraging our technical capabilities and commitment to excellence, we can help you accelerate your product development timelines while maintaining cost efficiency. Let us be your partner in navigating the complexities of fine chemical synthesis for the next generation of electronic devices.
