Scalable Synthesis of 4-Bromo-2-(Difluoromethyl)-5-Methylpyridine for Pharma Applications
Introduction to Advanced Pyridine Functionalization
The pharmaceutical and agrochemical industries increasingly rely on complex fluorinated heterocycles to enhance the metabolic stability and binding affinity of active ingredients. Patent CN113563258A discloses a robust and highly efficient preparation method for 4-bromo-2-(difluoromethyl)-5-methylpyridine, a critical building block in modern medicinal chemistry. This technical insight report analyzes the novel seven-step synthetic route detailed in the patent, which transforms the inexpensive starting material, 2,5-dimethylpyridine (Compound A), into the high-value target molecule through a sequence of oxidation, nitration, substitution, functionalization, hydrolysis, oxidation, and fluorination reactions. The methodology addresses significant challenges in regioselectivity and functional group tolerance, offering a viable pathway for the reliable supply of this specialized intermediate.
The strategic value of this synthesis lies in its linear progression and the use of well-understood chemical transformations that minimize the formation of difficult-to-remove impurities. By leveraging the activating effect of the N-oxide moiety, the process achieves precise substitution patterns that are often elusive in direct pyridine functionalization. For R&D directors and process chemists, this route represents a significant optimization over traditional methods that may require harsh conditions or multiple protection-deprotection cycles. The ability to install the difluoromethyl group late in the synthesis ensures that the sensitive fluorine functionality is preserved, resulting in a final product with exceptional purity profiles suitable for stringent pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing fluorinated pyridines often struggle with the direct introduction of fluorine atoms or difluoromethyl groups due to the electron-deficient nature of the pyridine ring. Conventional halogenation methods frequently result in mixtures of regioisomers, necessitating costly and yield-reducing purification steps such as preparative HPLC or repeated recrystallization. Furthermore, many existing routes rely on precious metal catalysts or exotic reagents that are not only expensive but also pose significant supply chain risks and environmental disposal challenges. The lack of regiocontrol in direct C-H functionalization of simple alkylpyridines often leads to complex impurity profiles that can compromise the safety and efficacy of the final drug substance.
The Novel Approach
The methodology presented in CN113563258A circumvents these issues by employing a stepwise functionalization strategy centered on the pyridine N-oxide intermediate. By first oxidizing the nitrogen atom, the electronic properties of the ring are altered to favor electrophilic attack at the 4-position, allowing for the clean installation of the nitro group which serves as a versatile leaving group. This is followed by a clever substitution with acetyl bromide to install the bromine atom with high fidelity. The subsequent transformation of the methyl group into a difluoromethyl moiety via an alcohol and aldehyde intermediate demonstrates a high level of chemoselectivity, ensuring that the bromine atom remains intact throughout the oxidation and fluorination steps. This logical disconnection of the synthesis problem allows for the use of commodity chemicals while achieving high-value structural complexity.
Mechanistic Insights into Regioselective Functionalization
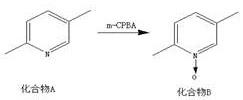
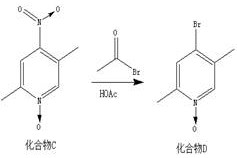
The core of this synthetic success lies in the mechanistic behavior of the pyridine N-oxide. In the initial oxidation step, m-chloroperoxybenzoic acid (m-CPBA) converts the pyridine nitrogen into an N-oxide, which significantly increases the electron density at the 4-position of the ring. This activation is crucial for the subsequent nitration step, where the nitronium ion attacks the 4-position exclusively, avoiding the formation of 2- or 3-substituted byproducts. The presence of the N-oxide also facilitates the subsequent displacement of the nitro group by bromide. Although the patent describes the reaction with acetyl bromide as a substitution, mechanistically it likely involves the activation of the N-oxide oxygen or the nitro group, leading to the expulsion of nitrogen oxides and the insertion of the bromine atom. This transformation is particularly valuable as it installs the halogen handle required for downstream cross-coupling reactions without requiring direct bromination conditions that could affect the methyl groups.
The final stage of the synthesis involves the conversion of the methyl group into a difluoromethyl group, a transformation that requires precise control over oxidation states. The process first functionalizes the methyl group to an acetoxymethyl ester using acetic anhydride, which is then hydrolyzed to the primary alcohol. The alcohol is selectively oxidized to the aldehyde using manganese dioxide, a mild oxidant that prevents over-oxidation to the carboxylic acid. Finally, the aldehyde is treated with DAST (diethylaminosulfur trifluoride), a powerful deoxofluorinating agent. The mechanism involves the formation of an aminooxysulfonium intermediate which collapses to release the difluoromethyl group. This sequence ensures that the fluorine atoms are introduced in the final step, minimizing exposure of the sensitive C-F bonds to the earlier acidic and oxidative conditions, thereby maximizing the overall yield and purity of the final 4-bromo-2-(difluoromethyl)-5-methylpyridine.
How to Synthesize 4-Bromo-2-(Difluoromethyl)-5-Methylpyridine Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for producing this complex intermediate with high reproducibility. The process begins with the careful control of temperature during the exothermic oxidation and nitration steps to prevent runaway reactions and ensure safety. Each intermediate is isolated and purified, typically via column chromatography or extraction, to maintain high purity standards before proceeding to the next step. The use of standard solvents like dichloromethane, chloroform, and acetic acid makes this process adaptable to existing manufacturing infrastructure without the need for specialized equipment. Detailed standardized synthesis steps follow below for technical reference.
- Oxidize Compound A (2,5-dimethylpyridine) with m-CPBA to form the N-oxide (Compound B).
- Perform nitration on Compound B using nitric acid and sulfuric acid to introduce the nitro group at the 4-position (Compound C).
- Execute substitution using acetyl bromide to replace the nitro group with bromine, yielding Compound D.
- React Compound D with acetic anhydride to functionalize the methyl group into an acetoxymethyl group (Compound E).
- Hydrolyze Compound E with hydrochloric acid to obtain the hydroxymethyl derivative (Compound F).
- Oxidize Compound F using manganese dioxide to generate the aldehyde intermediate (Compound G).
- Conduct fluorination using DAST in DCM to convert the aldehyde to the difluoromethyl group, yielding the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers substantial advantages over alternative methods that rely on scarce resources or complex catalytic systems. The reliance on bulk commodity chemicals such as nitric acid, sulfuric acid, acetic anhydride, and manganese dioxide ensures that raw material costs remain stable and predictable, shielding the supply chain from the volatility associated with precious metal catalysts. The elimination of transition metals like palladium or platinum not only reduces the direct cost of goods sold but also simplifies the downstream processing requirements, as there is no need for expensive metal scavenging resins or rigorous testing for residual heavy metals in the final API. This streamlined workflow translates directly into improved margin potential for the final drug product.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing a linear sequence of reactions that employ inexpensive, readily available reagents. The avoidance of cryogenic conditions for most steps (except the final fluorination which is standard) and the use of ambient pressure reactions reduce energy consumption and capital expenditure on specialized reactor vessels. Furthermore, the high selectivity of the N-oxide mediated steps minimizes the generation of isomeric impurities, which drastically reduces the volume of solvent and silica gel required for purification, leading to lower waste disposal costs and higher overall mass efficiency.
- Enhanced Supply Chain Reliability: By sourcing starting materials like 2,5-dimethylpyridine and reagents like m-CPBA and DAST from established global chemical suppliers, the risk of supply disruption is significantly mitigated. Unlike proprietary catalysts that may be sourced from a single vendor, the reagents used in this pathway are produced by multiple manufacturers worldwide, ensuring business continuity. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without catastrophic failure, further enhances the reliability of the manufacturing process, ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been designed with kilogram-to-ton production in mind. The reactions do not generate excessive amounts of hazardous byproducts, and the solvents used are common organic liquids that can be efficiently recovered and recycled through distillation. The use of manganese dioxide as a heterogeneous oxidant simplifies filtration and waste handling compared to homogeneous oxidants. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site, aligning with the sustainability goals of modern pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for process development teams. Understanding these nuances is critical for successful technology transfer and scale-up operations.
Q: What is the key advantage of using the N-oxide intermediate in this synthesis?
A: The formation of the pyridine N-oxide (Compound B) activates the ring towards electrophilic substitution, specifically facilitating the regioselective nitration at the 4-position, which is otherwise difficult to achieve directly on the pyridine ring.
Q: How is the bromine atom introduced in this specific pathway?
A: The bromine atom is introduced via a substitution reaction where the nitro group at the 4-position of Compound C is replaced by bromine using acetyl bromide, avoiding the need for direct halogenation which can suffer from poor selectivity.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common industrial reagents such as m-CPBA, nitric acid, and manganese dioxide, and avoids expensive transition metal catalysts, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Bromo-2-(Difluoromethyl)-5-Methylpyridine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract manufacturing, possessing the technical expertise to translate complex patent methodologies like CN113563258A into commercial reality. Our team of experienced process chemists is adept at optimizing reaction parameters to maximize yield and purity, ensuring that the final product meets the rigorous specifications required for pharmaceutical intermediates. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, utilizing our state-of-the-art facilities equipped with rigorous QC labs to guarantee batch-to-batch consistency. Our commitment to quality ensures that every shipment of 4-bromo-2-(difluoromethyl)-5-methylpyridine adheres to stringent purity specifications, providing our partners with the confidence needed to advance their drug development pipelines.
We invite global pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced synthetic technology for their specific needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements, helping you identify the most economical production strategy. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our manufacturing capabilities can support your long-term supply chain objectives for high-value fluorinated intermediates.
