Advanced Continuous Flow Synthesis of Alpha-Ketoleucine Calcium for Renal Care Applications
Introduction to Next-Generation Renal Care Intermediates
The pharmaceutical landscape for chronic kidney disease management relies heavily on the availability of high-purity alpha-keto acid analogues, specifically alpha-ketoleucine calcium, which serves as the active nitrogen-scavenging component in compound preparations for uremia treatment. Patent CN102030631A discloses a transformative manufacturing methodology that addresses the longstanding safety and efficiency bottlenecks associated with producing this critical medicinal raw material. Unlike legacy processes that depend on hazardous oxidants and toxic nitrile precursors, this innovation leverages a glycine-based foundation to establish a cleaner, more robust synthetic pathway. The structural integrity of the final product is paramount for its biological efficacy in reducing urea synthesis and toxic product accumulation in nephrotic patients.
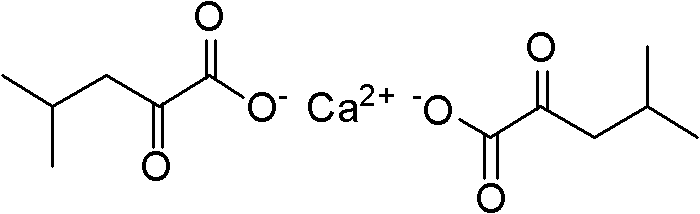
By shifting the synthetic strategy towards a one-pot cyclization followed by continuous flow hydrolysis, the technology enables a level of process control that is unattainable with traditional batch reactors. This approach not only mitigates the environmental burden of wastewater generation but also ensures a consistent impurity profile essential for regulatory compliance in global markets. For R&D directors and supply chain leaders, understanding this shift from batch to continuous processing is critical for evaluating future sourcing strategies for renal care intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of alpha-ketoleucine calcium has been plagued by significant operational hazards and inefficiencies that compromise both cost and safety. One prominent legacy route, documented in early chemical literature, utilizes ethyl cyanoacetate and isobutyraldehyde via a Knoevenagel-Cope condensation, necessitating a subsequent oxidation step with hydrogen peroxide. This pathway is inherently dangerous due to the toxicity of the cyanide-containing starting material and the explosion risks associated with large-scale peroxide handling, while also suffering from a cumbersome six-step sequence that erodes overall yield. Another common method involves the condensation of glycolylurea with isobutyraldehyde, which presents a different set of challenges related to the extreme difficulty of hydrolyzing the resulting intermediate.
The hydrolysis of 5-benzylidene glycolylurea typically requires harsh alkaline conditions and prolonged reaction times, leading to significant decomposition of the sensitive keto-acid framework and consequently low isolation yields. Furthermore, the necessity for massive quantities of alkali lye generates substantial saline wastewater, creating a heavy environmental compliance burden for manufacturers. These conventional batch processes are characterized by poor heat transfer during exothermic steps and inconsistent mixing, which often results in broad impurity distributions that require expensive downstream purification to meet pharmaceutical standards.
The Novel Approach
The methodology outlined in the patent represents a decisive break from these inefficient traditions by employing a streamlined two-step sequence initiated by a one-pot catalytic cyclization. By reacting pharmaceutical-grade glycine with isobutyraldehyde and acetic anhydride in the presence of an organic base, the process directly constructs the 4-isobutylidene-2-methyl dihydro oxazolone intermediate with high atom economy. This cyclization step avoids the use of toxic nitriles entirely and operates under mild conditions where the organic catalyst and excess reagents can be easily distilled and recycled, drastically reducing raw material consumption.
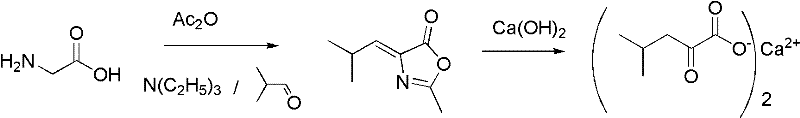
Following cyclization, the process innovates further by utilizing a continuous pipeline reactor for the hydrolysis stage, where the oxazolone intermediate reacts directly with calcium hydroxide. This continuous flow setup allows for precise regulation of residence time and temperature, ensuring that the ring-opening reaction proceeds rapidly without exposing the product to degrading conditions. The direct use of calcium hydroxide eliminates the need for a separate neutralization step with sodium hydroxide followed by salt metathesis, thereby simplifying the workflow and minimizing the generation of inorganic salt waste.
Mechanistic Insights into Organic Base-Catalyzed Cyclization and Flow Hydrolysis
The core chemical transformation in the first stage is an Erlenmeyer-Plöchl type azlactone formation, driven by the nucleophilic attack of the glycine amino group on the activated carbonyl species generated in situ. The organic base catalyst, which can range from aliphatic tertiary amines like tri-n-propylamine to heterocyclic amines like N-methylmorpholine, plays a dual role in scavenging the acetic acid byproduct and activating the acetic anhydride for N-acylation. This activation facilitates the subsequent dehydration and cyclization to form the oxazolone ring, a highly reactive electrophile that is primed for nucleophilic attack in the subsequent step. The choice of catalyst is critical, as it must be sufficiently basic to drive the condensation yet volatile enough to be separated from the product mixture via distillation for reuse.
In the second stage, the mechanism shifts to a nucleophilic acyl substitution where the hydroxide ion from calcium hydroxide attacks the carbonyl carbon of the oxazolone ring. The unique advantage of this specific intermediate lies in the relative lability of the amido linkage within the oxazolone system compared to the more stable urea linkages found in glycolylurea derivatives. This inherent reactivity allows the hydrolysis to proceed efficiently at moderate temperatures (120-130°C) within a short residence time of 5 to 10 minutes. The continuous flow environment ensures that every molecule experiences identical thermal history, effectively suppressing side reactions such as decarboxylation or polymerization that typically plague batch hydrolysis of alpha-keto acid precursors.
How to Synthesize Alpha-Ketoleucine Calcium Efficiently
The operational protocol for this synthesis is designed to maximize throughput while maintaining stringent quality control over the intermediate and final product specifications. The process begins with the careful metering of glycine and the organic catalyst into a reactor, followed by the controlled addition of acetic anhydride to manage the exotherm, ensuring the temperature remains within the optimal 40-50°C range during the initial acylation. Once the acylation is complete, isobutyraldehyde is introduced to drive the condensation, and the mixture is heated to promote cyclization, after which volatile components are stripped under reduced pressure to isolate the crude oxazolone.
- Perform a one-pot catalytic cyclization using glycine, isobutyraldehyde, and acetic anhydride with an organic base catalyst to form 4-isobutylidene-2-methyl dihydro oxazolone.
- Prepare a mixture of the oxazolone intermediate and calcium hydroxide in water for continuous feeding.
- Pump the mixture through a heated pipeline reactor at 120-130°C and 0.2-0.3 MPa for 5-10 minutes to achieve ring-opening hydrolysis and salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented methodology offers profound implications for cost structure and supply reliability. The elimination of hazardous raw materials like ethyl cyanoacetate removes the need for specialized storage infrastructure and expensive safety protocols, directly lowering the overhead costs associated with facility maintenance and insurance. Furthermore, the ability to recover and recycle the organic amine catalyst and acetic anhydride creates a closed-loop system for key reagents, insulating the production cost from volatility in the petrochemical market and reducing the total volume of purchased inputs required per kilogram of product.
- Cost Reduction in Manufacturing: The integration of a continuous pipeline reactor for the hydrolysis step significantly enhances energy efficiency compared to traditional batch heating and cooling cycles. By reducing the reaction time from hours to mere minutes and eliminating the need for extensive neutralization and filtration steps associated with sodium hydroxide usage, the process lowers utility consumption and labor hours. The direct formation of the calcium salt avoids the yield losses inherent in multi-step salt metathesis procedures, ensuring that a higher percentage of raw material mass is converted into saleable product.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as glycine, isobutyraldehyde, and acetic anhydride ensures a robust supply base that is not subject to the geopolitical or logistical constraints often faced by specialty fine chemical precursors. The continuous nature of the hydrolysis step allows for scalable production rates that can be adjusted dynamically to meet demand fluctuations without the long turnaround times typical of batch campaign manufacturing. This flexibility provides a strategic buffer against market shortages, ensuring consistent delivery schedules for downstream pharmaceutical formulators.
- Scalability and Environmental Compliance: The reduction in wastewater volume and the absence of heavy metal or cyanide contaminants simplify the effluent treatment process, reducing the capital expenditure required for environmental compliance infrastructure. The modular nature of pipeline reactors facilitates linear scale-up, allowing manufacturers to increase capacity by adding parallel reactor lines rather than building massively larger vessels, which minimizes the technical risk associated with scaling novel chemistries. This approach aligns with modern green chemistry principles, enhancing the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this continuous flow synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on how this method compares to established industry practices.
Q: How does this method improve safety compared to traditional ethyl cyanoacetate routes?
A: This method replaces toxic ethyl cyanoacetate and hazardous hydrogen peroxide oxidation steps with pharmaceutical-grade glycine and acetic anhydride, significantly reducing operator exposure to carcinogens and explosion risks.
Q: What are the advantages of using a pipeline reactor for hydrolysis?
A: The pipeline reactor allows for precise control of residence time (5-10 minutes) and temperature, preventing the degradation of the sensitive alpha-keto acid structure that often occurs during prolonged batch heating.
Q: Can the organic catalysts be recovered in this process?
A: Yes, the organic amine catalysts used in the cyclization step, such as triethylamine, can be distilled and recovered along with excess acetic anhydride and acetic acid for reuse in subsequent batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Ketoleucine Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced continuous flow technologies is essential for meeting the rigorous demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102030631A are fully realized in practical manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-ketoleucine calcium meets the exacting standards required for renal care formulations.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages specific to your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our commitment to delivering high-quality pharmaceutical intermediates.
