Advanced Aqueous Synthesis of (E)-2-Hexenoic Acid for High-Purity Flavor Manufacturing
Introduction to Green Synthesis of (E)-2-Hexenoic Acid
The global demand for high-purity flavor and fragrance intermediates is driving a significant shift towards sustainable manufacturing protocols, particularly for key compounds like (E)-2-hexenoic acid. This unsaturated fatty acid, known commercially as trans-2-Hexenoic acid (FEMA 3169), is a critical component in formulating apple, banana, and caramel notes for the food and beverage industry. 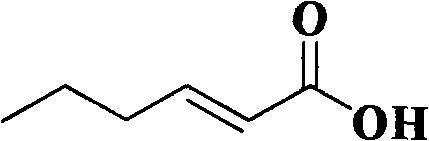 Recent intellectual property developments, specifically patent CN101602663A, have introduced a groundbreaking aqueous-phase synthesis route that addresses long-standing environmental and efficiency challenges. By leveraging a phase-transfer catalyzed Knoevenagel condensation followed by hydrolysis and decarboxylation, this technology enables the production of high-purity (E)-2-hexenoic acid without relying on hazardous organic solvents. For R&D directors and procurement specialists, understanding this technological pivot is essential for securing a reliable flavor intermediate supplier capable of meeting stringent regulatory and sustainability standards.
Recent intellectual property developments, specifically patent CN101602663A, have introduced a groundbreaking aqueous-phase synthesis route that addresses long-standing environmental and efficiency challenges. By leveraging a phase-transfer catalyzed Knoevenagel condensation followed by hydrolysis and decarboxylation, this technology enables the production of high-purity (E)-2-hexenoic acid without relying on hazardous organic solvents. For R&D directors and procurement specialists, understanding this technological pivot is essential for securing a reliable flavor intermediate supplier capable of meeting stringent regulatory and sustainability standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of (E)-2-hexenoic acid has been plagued by significant technical and environmental drawbacks associated with traditional synthetic pathways. The Perkin reaction, once a standard approach, suffers from excessively high reaction temperatures and prolonged processing times, which inevitably lead to thermal degradation of sensitive intermediates and suboptimal overall yields. Furthermore, alternative halogenation routes are often constrained by the scarcity and high cost of specific halogenated raw materials, creating supply chain bottlenecks that jeopardize production continuity. Perhaps most critically, the widely adopted Knovenagel-Dobner reaction typically necessitates the use of toxic organic solvents such as benzene or pyridine to drive the condensation equilibrium. These volatile organic compounds (VOCs) pose severe occupational health risks and impose heavy financial burdens related to solvent recovery, waste treatment, and environmental compliance, making them increasingly untenable in modern green chemistry frameworks.
The Novel Approach
In stark contrast to these legacy methods, the innovative protocol described in patent CN101602663A utilizes water as the exclusive reaction medium, fundamentally altering the safety and economic profile of the synthesis. This aqueous-phase strategy employs readily available and inexpensive starting materials, specifically diethyl malonate and n-butyraldehyde, which are commoditized chemicals with stable global supply chains. The integration of phase transfer catalysts allows the reaction to proceed under remarkably mild conditions, typically ranging from ambient temperature up to 90°C, thereby minimizing energy consumption and preventing the formation of polymeric byproducts common in harsher environments. By eliminating toxic solvents entirely, this novel approach not only simplifies the downstream purification process but also aligns perfectly with the rigorous environmental, social, and governance (ESG) goals of modern multinational corporations seeking cost reduction in synthetic flavors manufacturing.
Mechanistic Insights into Aqueous Phase Knoevenagel Condensation
The core of this technological breakthrough lies in the efficient execution of the Knoevenagel condensation within a biphasic or micellar aqueous environment facilitated by quaternary ammonium salts. In this system, the phase transfer catalyst, such as tetrabutylammonium bromide (TBAB) or triethylbenzyl ammonium chloride (TEBA), acts as a molecular shuttle, transporting the deprotonated active methylene species of diethyl malonate into the organic micro-environment where it reacts with n-butyraldehyde.  This interfacial catalysis significantly accelerates the reaction kinetics while maintaining high stereoselectivity for the desired (E)-isomer. Following the initial condensation, the reaction mixture undergoes a sequential one-pot transformation involving alkaline hydrolysis of the ester groups and subsequent thermal decarboxylation. This tandem process is meticulously controlled by adjusting the temperature profile from 25°C to 60°C and finally to 80-90°C, ensuring complete conversion while suppressing side reactions that could compromise the purity of the final carboxylic acid product.
This interfacial catalysis significantly accelerates the reaction kinetics while maintaining high stereoselectivity for the desired (E)-isomer. Following the initial condensation, the reaction mixture undergoes a sequential one-pot transformation involving alkaline hydrolysis of the ester groups and subsequent thermal decarboxylation. This tandem process is meticulously controlled by adjusting the temperature profile from 25°C to 60°C and finally to 80-90°C, ensuring complete conversion while suppressing side reactions that could compromise the purity of the final carboxylic acid product.
From an impurity control perspective, the use of water as a solvent provides a unique thermodynamic advantage by dissolving polar byproducts and inorganic salts, which remain in the aqueous phase during the final isolation step. The subsequent acidification with mineral acids like hydrochloric or sulfuric acid precipitates the target (E)-2-hexenoic acid, allowing for easy separation via filtration or extraction. This inherent self-purification mechanism drastically reduces the complexity of the work-up procedure compared to organic solvent-based systems where extensive washing and drying steps are required. For quality assurance teams, this translates to a cleaner crude product profile with fewer trace organic impurities, facilitating easier attainment of the stringent purity specifications required for food-grade applications and reducing the load on analytical QC labs.
How to Synthesize (E)-2-Hexenoic Acid Efficiently
Implementing this synthesis route requires precise control over stoichiometry and thermal gradients to maximize the efficiency of the phase transfer catalysis. The process begins with the charging of water, diethyl malonate, n-butyraldehyde, and the selected catalyst into a reactor equipped with efficient agitation and reflux capabilities. The detailed standardized operating procedure involves a multi-stage heating protocol designed to optimize the condensation equilibrium before triggering the hydrolysis and decarboxylation cascades. Operators must strictly adhere to the specified molar ratios and temperature ramps to ensure consistent batch-to-b reproducibility and high yield. For a comprehensive breakdown of the exact operational parameters and safety considerations, please refer to the technical guide below.
- Combine water, diethyl malonate, n-butyraldehyde, and a phase transfer catalyst (such as TBAB) in a reactor equipped with a reflux condenser.
- Stir the mixture at room temperature, then gradually increase the temperature to 60°C and subsequently to 80-90°C to facilitate the Knoevenagel condensation.
- Add an alkaline solution for hydrolysis followed by acidification to induce decarboxylation, then isolate the product via reduced pressure distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous-based synthesis methodology offers profound strategic advantages that extend far beyond simple chemical efficiency. The elimination of expensive and regulated organic solvents like benzene removes a major variable cost driver and mitigates the risk of supply disruptions associated with petrochemical-derived solvents. Furthermore, the simplified work-up procedure reduces the requirement for specialized solvent recovery infrastructure, leading to substantial capital expenditure savings and lower operational overheads. The use of commodity-grade raw materials ensures that the production cost remains stable even during fluctuations in the specialty chemical market, providing a predictable pricing model for long-term contracts.
- Cost Reduction in Manufacturing: The replacement of toxic organic solvents with water eliminates the significant costs associated with solvent purchase, storage, recycling, and hazardous waste disposal. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high selectivity minimizes the loss of valuable raw materials to side products, collectively driving down the cost of goods sold (COGS) significantly without compromising quality.
- Enhanced Supply Chain Reliability: By utilizing diethyl malonate and n-butyraldehyde, which are produced on a massive industrial scale globally, manufacturers can secure a resilient supply chain that is less susceptible to the volatility seen in niche reagent markets. The robustness of the phase transfer catalyst system also allows for flexibility in sourcing, as multiple chemically equivalent catalysts (such as TBAB, TEBA, or CTAB) can be substituted based on availability, ensuring uninterrupted production schedules.
- Scalability and Environmental Compliance: The aqueous nature of the reaction makes scale-up inherently safer and more straightforward, as issues related to solvent flammability and explosion limits are effectively removed. This facilitates the commercial scale-up of complex flavor intermediates from pilot plants to multi-ton production facilities with minimal engineering modifications. Moreover, the drastic reduction in VOC emissions and hazardous waste generation ensures full compliance with increasingly strict international environmental regulations, safeguarding the company's license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of (E)-2-hexenoic acid using this patented aqueous methodology. These insights are derived directly from the experimental data and beneficial effects reported in the underlying intellectual property, providing a transparent view of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their broader supply networks.
Q: What are the primary environmental advantages of this synthesis method?
A: This method utilizes water as the sole reaction medium, completely eliminating the need for toxic organic solvents like benzene or pyridine traditionally used in Knoevenagel-Dobner reactions, thereby significantly reducing hazardous waste generation.
Q: What yield efficiency can be expected from this aqueous process?
A: According to patent data, the optimized aqueous phase protocol consistently achieves yields between 75% and 85%, which represents a substantial improvement over older Perkin or halogenation methods.
Q: Which phase transfer catalysts are compatible with this reaction system?
A: The process demonstrates robust compatibility with various quaternary ammonium salts, including TBAB (tetrabutylammonium bromide), TEBA, and hexadecyltrimethylammonium bromide, allowing for flexible sourcing strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-2-Hexenoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of fine chemicals requires more than just a laboratory recipe; it demands a partner with deep process engineering expertise and a commitment to quality. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of aqueous-phase reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of (E)-2-hexenoic acid meets the highest industry standards for flavor and fragrance applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your product portfolio. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green chemistry approach can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this critical flavor intermediate.
