Revolutionizing Sartan Intermediate Production via Continuous Flow Bromination Technology
Revolutionizing Sartan Intermediate Production via Continuous Flow Bromination Technology
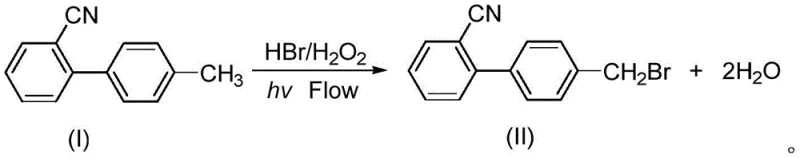
The global demand for angiotensin II receptor blockers (ARBs), commonly known as Sartans, continues to drive innovation in the synthesis of their key building blocks. Patent CN111960967A introduces a groundbreaking continuous flow method for synthesizing p-bromomethyl biphenyl carbonitrile, a critical intermediate for major antihypertensive drugs including Losartan, Irbesartan, Candesartan, and Valsartan. This technology represents a paradigm shift from traditional batch processing to a safer, greener, and more efficient continuous manufacturing protocol. By utilizing hydrobromic acid and hydrogen peroxide as bromine sources within a specialized pipeline reactor illuminated by LED lights, the process achieves superior atom economy and operational safety. For pharmaceutical manufacturers and procurement strategists, this patent offers a compelling roadmap for reducing production costs while adhering to stringent environmental regulations.
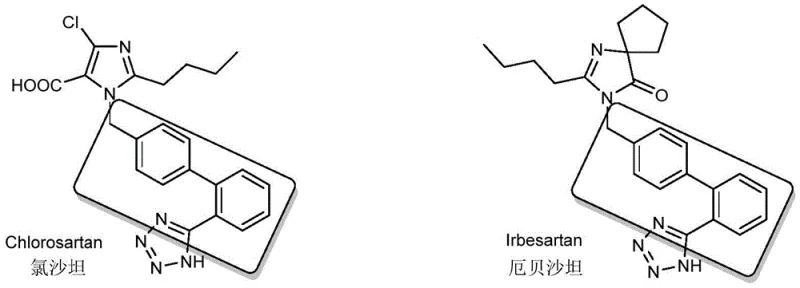
The significance of this intermediate cannot be overstated, as it serves as the foundational scaffold for a class of blockbuster medications used worldwide to treat essential hypertension and heart failure. The ability to produce this molecule with high purity and minimal waste directly impacts the supply chain stability of these life-saving drugs. The patented approach addresses long-standing challenges in bromination chemistry, specifically the issues of hazardous reagent handling and difficult waste disposal associated with conventional methods. As the industry moves towards Industry 4.0 and continuous manufacturing, adopting such flow chemistry protocols becomes not just an option but a strategic necessity for maintaining competitiveness in the pharmaceutical intermediates market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of p-bromomethyl biphenyl carbonitrile has relied on batch processes that suffer from significant inefficiencies and environmental drawbacks. Traditional methods often employ N-bromosuccinimide (NBS) or dibromo hydantoin (DBDMH) as brominating agents. While effective, these reagents generate stoichiometric amounts of solid byproducts, such as succinimide or hydantoin, which require complex and costly separation procedures to recycle or dispose of safely. Furthermore, alternative methods using elemental bromine directly pose severe safety risks due to the toxicity and corrosivity of bromine vapor, alongside poor atom economy where nearly half of the bromine atoms end up as waste hydrogen bromide. These legacy processes are increasingly untenable in a regulatory environment that demands greener chemistry and lower E-factors.
The Novel Approach
The patented continuous flow method fundamentally reimagines the bromination step by generating the active brominating species in situ. Instead of introducing pre-formed bromine or solid N-bromo compounds, the system mixes hydrobromic acid and hydrogen peroxide immediately before the reaction zone. This mixture generates elemental bromine instantaneously within the flow stream, which then reacts with the substrate under LED irradiation. This approach ensures that the bromine is consumed as soon as it is formed, minimizing exposure risks and maximizing atom utilization. The integration of flow chemistry allows for precise control over reaction temperature and residence time, eliminating the hot spots and runaway reaction risks common in exothermic batch brominations. Consequently, this novel route transforms a hazardous, waste-intensive operation into a streamlined, continuous process suitable for modern industrial application.
Mechanistic Insights into Photochemical Flow Bromination
The core of this innovation lies in the synergistic combination of chemical oxidation and photochemical activation within a confined flow environment. In the pipeline reactor, hydrobromic acid is oxidized by hydrogen peroxide to generate molecular bromine (Br2). Under the influence of specific LED wavelengths (ranging from 405nm to 610nm), the bromine molecules undergo homolytic cleavage to form highly reactive bromine radicals. These radicals selectively abstract a hydrogen atom from the methyl group of the p-methyl biphenyl carbonitrile substrate, initiating a free-radical chain substitution mechanism. The continuous flow setup ensures that the concentration of radicals remains optimal throughout the reactor length, preventing side reactions such as over-bromination or ring substitution which are common in uncontrolled batch conditions.
Impurity control is inherently superior in this system due to the plug-flow nature of the reactor. Unlike batch reactors where concentration gradients exist, every molecule of substrate experiences the exact same reaction history in terms of time, temperature, and light exposure. This uniformity drastically reduces the formation of poly-brominated byproducts and oxidation impurities. Furthermore, the immediate quenching of the reaction mixture downstream with a reducing agent (such as sodium bisulfite or carbonate) ensures that any residual active bromine is neutralized instantly. This rapid quenching prevents post-reaction degradation, resulting in a crude product profile that is exceptionally clean, thereby simplifying the subsequent purification steps and ensuring final purity specifications are met with minimal effort.
How to Synthesize p-Bromomethyl Biphenyl Carbonitrile Efficiently
Implementing this synthesis requires a specialized setup involving multiple feed streams and a tubular reactor system. The process begins with the preparation of three distinct liquid streams: a solution of the starting material in an organic solvent like dichloromethane or ethyl acetate, an aqueous hydrobromic acid solution, and an aqueous hydrogen peroxide solution. These streams are metered precisely into the system using high-precision pumps to maintain the stoichiometric balance required for optimal conversion. The mixing and reaction occur in transparent tubing (Teflon or quartz) that allows for maximum light penetration from the internal LED sources, ensuring efficient photon utilization for the radical initiation step.
- Prepare feed solutions: Dissolve p-methyl biphenyl carbonitrile in an organic solvent (e.g., dichloromethane), and prepare aqueous solutions of hydrobromic acid and hydrogen peroxide.
- Initiate flow reaction: Pump the organic substrate and aqueous oxidants through high-efficiency mixers into a temperature-controlled pipeline reactor (5-70°C) equipped with LED light sources.
- Quench and isolate: Mix the effluent with a quenching agent (e.g., sodium bisulfite), separate the organic phase, dry, concentrate, and recrystallize to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this continuous flow technology offers tangible strategic benefits beyond mere technical elegance. The elimination of solid brominating agents like NBS removes the logistical burden of handling and storing large quantities of hazardous solids, which often require special warehousing and incur higher transportation costs. Additionally, the byproduct of this reaction is primarily water, which dramatically simplifies wastewater treatment compared to the nitrogen-containing organic waste streams generated by NBS or DBDMH methods. This reduction in waste treatment complexity translates directly into lower operational expenditures and a reduced environmental footprint, aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic argument for this process is robust, driven primarily by the superior atom economy of the HBr/H2O2 system. Unlike traditional methods where expensive carrier molecules like succinimide are discarded or require energy-intensive recycling, the reagents here are commodity chemicals with low market prices. The continuous nature of the process also reduces labor costs associated with batch charging, monitoring, and cleaning. By minimizing solvent usage and eliminating the need for extensive purification to remove solid byproducts, the overall cost of goods sold (COGS) for this pharmaceutical intermediate can be significantly optimized, providing a competitive edge in pricing negotiations.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents and the safety restrictions on transporting hazardous materials. This method relies on hydrobromic acid and hydrogen peroxide, which are widely available bulk chemicals with stable supply chains. The inherent safety of the flow system, which holds only a small volume of reactive mixture at any given time, reduces the risk of plant shutdowns due to safety incidents. This reliability ensures that production schedules for downstream Sartan APIs remain uninterrupted, safeguarding against market shortages and price volatility that can arise from batch process failures or regulatory inspections.
- Scalability and Environmental Compliance: Scaling photochemical reactions in batch mode is notoriously difficult due to the Beer-Lambert law limiting light penetration in large vessels. Continuous flow reactors overcome this limitation by maintaining a small path length for light regardless of the total production volume, allowing for linear scale-up simply by running the reactor longer or numbering up the units. This scalability ensures that the process remains efficient from pilot plant to full commercial production. Moreover, the green chemistry profile of the process, characterized by water as the main byproduct and high atom utilization, facilitates easier compliance with increasingly strict environmental regulations, reducing the risk of fines or production halts related to waste discharge limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous flow bromination technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of adoption. Understanding these details is crucial for technical teams assessing the integration of flow chemistry into existing manufacturing lines.
Q: What are the primary advantages of the HBr/H2O2 flow method over traditional NBS bromination?
A: The HBr/H2O2 method eliminates solid succinimide waste, achieves higher atom economy by generating bromine in situ, and significantly improves safety by avoiding the handling of elemental bromine or unstable solid brominating agents.
Q: How does continuous flow technology impact the scalability of this intermediate?
A: Continuous flow allows for precise control of reaction parameters like residence time and light exposure, enabling linear scale-up from laboratory to industrial production without the heat transfer limitations typical of batch photochemical reactions.
Q: What purity levels can be achieved with this patented synthesis route?
A: According to patent examples, the process consistently yields products with HPLC purity exceeding 99%, with specific examples demonstrating purity up to 99.4% after simple recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Bromomethyl Biphenyl Carbonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our expertise in process development allows us to adapt advanced technologies, such as the continuous flow bromination described in CN111960967A, to meet the rigorous demands of international clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of p-bromomethyl biphenyl carbonitrile meets the highest standards required for API synthesis.
We invite you to collaborate with us to leverage these technological advancements for your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can reduce your overall manufacturing costs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term supply chain strategy.
