Advanced One-Step Pd-Catalyzed Synthesis of 2-Cyano-4'-Methyl Biphenyl for Commercial Scale-Up
Advanced One-Step Pd-Catalyzed Synthesis of 2-Cyano-4'-Methyl Biphenyl for Commercial Scale-Up
The pharmaceutical industry's relentless demand for sartan-class antihypertensive agents, such as Losartan and Valsartan, has placed immense pressure on the supply chains of their critical intermediates. Among these, 2-cyano-4'-methyl biphenyl (CAS: 114772-53-1) stands out as a pivotal building block, yet its traditional manufacturing processes have long been plagued by inefficiencies and safety concerns. Patent CN103012201A introduces a transformative synthetic methodology that addresses these historical bottlenecks through a highly selective, one-step palladium-catalyzed coupling reaction. This innovation not only streamlines the production workflow but also establishes a new benchmark for purity and operational safety in the synthesis of biaryl nitriles. By leveraging a specialized Pd-complex catalyst and a optimized solvent system, this technology offers a robust pathway for the reliable pharmaceutical intermediate supplier seeking to modernize their production capabilities.
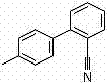
The molecular architecture of 2-cyano-4'-methyl biphenyl requires precise construction to ensure downstream efficacy in drug synthesis. The structural integrity of this intermediate is paramount, as impurities can propagate through the synthesis of the final API, leading to costly regulatory setbacks. The patented approach focuses on the direct coupling of readily available halides, bypassing the multi-step sequences that characterize older methodologies. This directness is not merely a matter of convenience; it is a strategic imperative for reducing the environmental footprint and enhancing the economic viability of large-scale manufacturing. As we delve deeper into the technical specifics, it becomes evident that this method represents a significant leap forward in process chemistry, offering a solution that balances high yield with rigorous quality control standards essential for global pharmaceutical markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-cyano-4'-methyl biphenyl has been fraught with technical challenges that hinder industrial scalability and cost-effectiveness. As illustrated in the comprehensive reaction scheme below, prior art encompasses seven distinct routes, each suffering from critical deficiencies that limit their commercial utility. For instance, early methods relying on the Meyer reaction from o-anisic acid involve excessively long synthetic sequences, resulting in cumulative yield losses that drive up the final cost of goods. Furthermore, routes utilizing Grignard reagents (Routes 2 and 4) introduce severe safety hazards associated with pyrophoric materials and require stringent anhydrous conditions, making them difficult to manage in large-scale reactors. Other approaches employing nickel, zinc, or manganese catalysts often struggle with poor reaction selectivity, leading to complex impurity profiles that are arduous and expensive to separate. Additionally, methods requiring cryogenic temperatures, such as the synthesis of 4-methylphenylboronic acid at -70°C, impose prohibitive energy costs and equipment requirements that are unsustainable for continuous manufacturing.
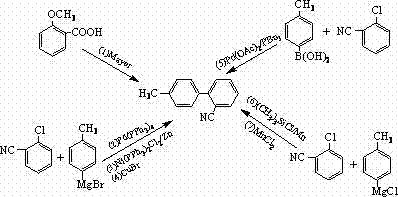
Beyond the technical hurdles, the environmental and operational burdens of these conventional methods cannot be overstated. The use of stoichiometric amounts of metals like manganese in Route 6 generates substantial quantities of heavy metal waste, complicating disposal and increasing the environmental compliance burden for manufacturers. Similarly, the industrialized method (Route 7), while using cheap raw materials, suffers from unstable yields and difficult product separation, leading to inconsistent batch quality. These inconsistencies are unacceptable in the highly regulated pharmaceutical sector, where batch-to-batch reproducibility is a non-negotiable requirement. The accumulation of byproducts in these traditional routes necessitates extensive purification steps, such as multiple recrystallizations or chromatographic separations, which further erode profit margins and extend production lead times. Consequently, there is a pressing need for a unified, efficient, and safe synthetic strategy that can overcome these multifaceted limitations.
The Novel Approach
The patented methodology presented in CN103012201A offers a decisive break from these inefficient traditions by employing a direct, one-step coupling of 2-bromobenzonitrile and p-bromotoluene. This streamlined approach eliminates the need for pre-functionalized reagents like boronic acids or Grignard reagents, thereby simplifying the raw material supply chain and reducing the number of unit operations. Central to this innovation is the use of a specialized palladium complex catalyst that facilitates the cross-coupling reaction under remarkably mild conditions, typically between 30°C and 60°C. This low-temperature operation not only enhances safety by minimizing thermal risks but also significantly reduces energy consumption compared to high-temperature reflux methods. The reaction is conducted in a carefully tuned solvent system, preferably a mixture of tetrahydrofuran and toluene, which has been empirically determined to maximize both conversion rates and selectivity. By optimizing the molar ratios of reactants and the sequence of addition—specifically adding the catalyst to the nitrile before introducing the toluene derivative—the process achieves superior control over the reaction trajectory, minimizing the formation of homocoupling byproducts.
Mechanistic Insights into Pd-Complex Catalyzed Coupling
The success of this novel synthetic route hinges on the sophisticated design of the palladium catalyst and its interaction with the reaction medium. The catalyst, a Pd-complex featuring a bulky phosphine or carbene-based ligand system (as depicted in the structural formulas), plays a critical role in facilitating the oxidative addition of the aryl bromide bonds. The steric bulk of the ligand environment surrounding the palladium center is engineered to promote the desired cross-coupling while sterically hindering unwanted side reactions, such as the homocoupling of the starting materials. This precise tuning of the electronic and steric properties of the catalyst ensures that the catalytic cycle proceeds efficiently through the transmetallation and reductive elimination steps, ultimately delivering the target biphenyl structure with high fidelity. The stability of this catalyst under the reaction conditions is another key factor, as it allows for sustained activity throughout the reaction duration without rapid degradation, which is a common failure mode in less robust catalytic systems.
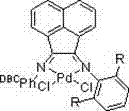
Furthermore, the solvent system exerts a profound influence on the mechanistic pathway and the overall outcome of the reaction. The patent highlights a surprising synergistic effect when using a mixture of tetrahydrofuran (THF) and toluene, particularly at a volume ratio of 1:1 or 1:2. This specific solvent blend appears to optimize the solvation of the polar transition states involved in the catalytic cycle while maintaining sufficient solubility for the organic substrates. The polarity of THF aids in stabilizing the ionic intermediates formed during the transmetallation phase, while the aromatic nature of toluene provides a compatible environment for the hydrophobic aryl rings. This dual-solvent strategy effectively suppresses the formation of insoluble palladium black, a common deactivation pathway, thereby extending the catalyst's lifespan. Additionally, the controlled addition of p-bromotoluene to the pre-mixed catalyst and o-bromobenzonitrile solution ensures that the concentration of the electrophile remains low relative to the nucleophile-catalyst complex, further driving the selectivity towards the cross-coupled product and minimizing impurity generation.
How to Synthesize 2-Cyano-4'-Methyl Biphenyl Efficiently
Implementing this advanced synthesis requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and purity. The process begins with the preparation of the reaction vessel under an inert atmosphere, followed by the dissolution of o-bromobenzonitrile in the THF/toluene solvent mixture. The specialized Pd catalyst is then introduced, and the mixture is heated gently to allow for the formation of the active catalytic species. Once this pre-activation step is complete, p-bromotoluene is added slowly via dropping funnel to maintain control over the reaction exotherm and kinetics. Following the reaction period, the workup involves a straightforward acid wash to remove basic impurities and residual catalyst, followed by extraction and distillation. The final purification is achieved through recrystallization using a benzene and tetrahydrofuran mixture, which yields the product as high-purity light yellow needle crystals. For the detailed standardized operating procedures and specific safety protocols, please refer to the guide below.
- Dissolve o-bromobenzonitrile in a mixed organic solvent of tetrahydrofuran and toluene under nitrogen protection.
- Add the specialized Pd complex catalyst and heat to 30-60°C, ensuring full complexation before adding the second reactant.
- Slowly drop p-bromotoluene into the mixture, maintain temperature for 2-4 hours, then purify via acid wash and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic benefits that extend far beyond simple chemical yield. The shift from multi-step, hazardous processes to a streamlined one-step coupling fundamentally alters the cost structure of producing this critical intermediate. By eliminating the need for cryogenic reagents and dangerous Grignard preparations, the process significantly reduces the capital expenditure required for specialized equipment and safety infrastructure. Moreover, the use of commodity chemicals like bromotoluenes and bromobenzonitriles as starting materials ensures a stable and diversified supply base, mitigating the risk of raw material shortages that often plague specialty chemical markets. The simplified workflow also means fewer unit operations, which directly correlates to reduced labor costs, lower utility consumption, and a smaller physical footprint for production facilities. These factors combine to create a more resilient and cost-effective supply chain capable of meeting the fluctuating demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the drastic simplification of the synthetic route. By condensing the synthesis into a single catalytic step, manufacturers can eliminate the costs associated with isolating and purifying intermediates, which traditionally account for a significant portion of production expenses. The ability to operate at near-ambient temperatures (30-60°C) further slashes energy bills compared to processes requiring deep cooling or high-temperature reflux. Additionally, the high selectivity of the catalyst minimizes the generation of waste byproducts, reducing the costs associated with waste treatment and disposal. The potential for catalyst recovery and reuse adds another layer of cost efficiency, lowering the effective cost per kilogram of the precious metal catalyst. Collectively, these efficiencies result in a substantially lower cost of goods sold, providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: From a logistics perspective, this method offers superior reliability due to the stability and availability of its inputs. Unlike routes dependent on unstable organometallic reagents that must be generated in situ or shipped under hazardous conditions, this process utilizes stable liquid and solid bromides that are easily sourced from multiple global suppliers. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by minor variations in utility supply or environmental conditions. Furthermore, the high purity of the crude product reduces the dependency on complex downstream purification services, shortening the overall lead time from raw material intake to finished goods. This predictability allows supply chain planners to maintain leaner inventories while still ensuring continuity of supply for downstream API manufacturers, thereby optimizing working capital.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is facilitated by its inherent safety and simplicity. The absence of pyrophoric reagents and the use of mild temperatures make the reaction inherently safer to scale up, reducing the engineering challenges associated with heat transfer and mixing in large reactors. Environmentally, the process aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product) through higher atom economy and reduced solvent usage. The minimized heavy metal waste, thanks to the efficient catalyst system, simplifies compliance with increasingly stringent environmental regulations regarding heavy metal discharge. This environmental stewardship not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is becoming a critical criterion for selection by major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The clarity provided here aims to bridge the gap between academic innovation and industrial application, ensuring that all stakeholders have a accurate assessment of the technology's value proposition.
Q: What are the primary advantages of this Pd-catalyzed method over traditional Grignard routes?
A: Unlike traditional Grignard routes which pose significant safety hazards due to pyrophoric reagents and require cryogenic conditions, this novel method operates at mild temperatures (30-60°C) using stable bromide starting materials, drastically improving operational safety and reducing energy consumption.
Q: How does the specific solvent system impact product purity?
A: The patent identifies a synergistic effect in using a mixture of tetrahydrofuran and toluene (specifically at a 1:1 or 1:2 volume ratio). This specific polarity balance optimizes the solubility of reactants while facilitating the precipitation of impurities, leading to significantly higher selectivity and purity compared to single-solvent systems.
Q: Is the catalyst reusable in this process?
A: Yes, the specialized Pd complex catalyst described in the patent is designed for stability and potential recovery. The process minimizes catalyst decomposition, allowing for more efficient metal usage and reduced heavy metal waste compared to non-recyclable homogeneous catalyst systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyano-4'-Methyl Biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methods requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in the laboratory can be reliably replicated on an industrial scale. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex catalytic reactions safely and efficiently, adhering to the highest international standards for quality and safety. By partnering with us, you gain access to a supply chain that is not only robust and compliant but also optimized for cost and performance.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific project requirements. We are prepared to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this Pd-catalyzed method for your operations. Please contact us to request specific COA data for our current batches and to receive detailed route feasibility assessments tailored to your production volumes. Let us help you secure a competitive advantage in the sartan intermediate market through superior technology and reliable supply.
