Advanced Synthesis of 2-Bromo-3-Arylmercaptoindoles for Scalable Antibacterial Drug Manufacturing
Advanced Synthesis of 2-Bromo-3-Arylmercaptoindoles for Scalable Antibacterial Drug Manufacturing
The pharmaceutical industry constantly seeks robust, scalable pathways for constructing complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN102584675B introduces a significant breakthrough in the synthesis of 2-bromo-3-arylmercaptoindole compounds, represented by Formula (I). These molecules are not merely academic curiosities; they are pivotal intermediates for generating 2-aryl-3-arylmercaptoindoles and 2-aryl-3-arylsulfonylindoles, both of which exhibit potent antibacterial pharmacological activity. The structural versatility of Formula (I), where R1 and R2 can vary from hydrogen to alkyl, alkoxy, or halogen groups, allows medicinal chemists to rapidly explore structure-activity relationships (SAR) for optimizing drug candidates. 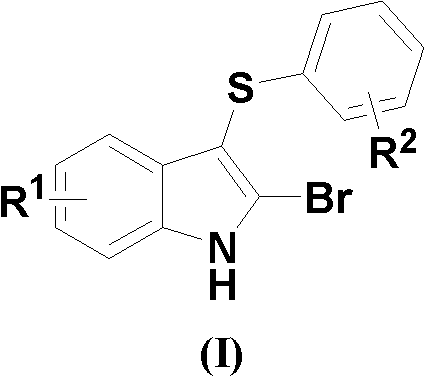 As a reliable pharmaceutical intermediate supplier, understanding the nuances of this patent is essential for securing a competitive edge in the supply of high-purity antibacterial precursors.
As a reliable pharmaceutical intermediate supplier, understanding the nuances of this patent is essential for securing a competitive edge in the supply of high-purity antibacterial precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-arylthio indole skeletons has been plagued by significant synthetic hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional methodologies often necessitate harsh reaction environments, specifically strong acidic conditions, which severely compromise the compatibility of sensitive functional groups on the aromatic rings. This lack of chemoselectivity frequently leads to the formation of numerous difficult-to-remove by-products, drastically reducing the overall isolated yield and complicating downstream purification processes. Furthermore, many prior art routes rely on specialized, expensive starting materials that are not readily available in the global bulk chemical market, creating supply chain bottlenecks and inflating the cost of goods sold (COGS). The difficulty in controlling reaction parameters under these aggressive conditions often results in batch-to-batch variability, a critical risk factor for regulatory compliance in GMP manufacturing environments.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102584675B offers a streamlined, operationally simple pathway that operates under remarkably mild conditions. The core innovation involves the direct reaction of readily available indole derivatives with diaryldisulfides in the presence of N-bromosuccinimide (NBS) using N,N-dimethylformamide (DMF) as the solvent. 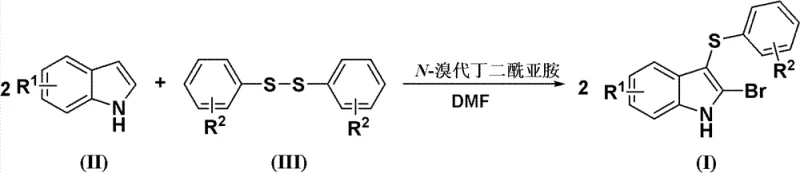 This novel approach eliminates the need for strong acids, thereby preserving the integrity of diverse functional groups and significantly minimizing the generation of impurities. The reaction proceeds efficiently at temperatures ranging from 10°C to 30°C over a period of 3.5 to 6 hours, demonstrating excellent energy efficiency. Experimental data from the patent indicates that this method achieves impressive yields, such as 81% with high purity (98.6%), proving its viability for cost reduction in pharmaceutical intermediate manufacturing without sacrificing quality.
This novel approach eliminates the need for strong acids, thereby preserving the integrity of diverse functional groups and significantly minimizing the generation of impurities. The reaction proceeds efficiently at temperatures ranging from 10°C to 30°C over a period of 3.5 to 6 hours, demonstrating excellent energy efficiency. Experimental data from the patent indicates that this method achieves impressive yields, such as 81% with high purity (98.6%), proving its viability for cost reduction in pharmaceutical intermediate manufacturing without sacrificing quality.
Mechanistic Insights into NBS-Mediated Bromination and Thiolation
The mechanistic elegance of this transformation lies in the dual role of N-bromosuccinimide (NBS) as both a brominating agent and an oxidant to activate the disulfide bond. In the proposed catalytic cycle, NBS likely generates an electrophilic bromine species that attacks the electron-rich C3 position of the indole ring, forming a reactive 3-bromoindolenine intermediate. Simultaneously, the disulfide bond of the diaryldisulfide is activated, possibly through interaction with the succinimide anion or the electrophilic bromine species, facilitating nucleophilic attack or radical substitution at the C3 position. This concerted or sequential process effectively installs both the bromine atom at the C2 position and the arylthio group at the C3 position in a single pot. The mild thermal conditions (10-30°C) are crucial here, as they prevent the decomposition of the reactive intermediates and suppress side reactions such as poly-bromination or oxidative degradation of the sulfur moiety, which are common pitfalls in harsher protocols.
From an impurity control perspective, the choice of DMF as a polar aprotic solvent enhances the solubility of both the organic substrates and the ionic intermediates, ensuring a homogeneous reaction mixture that promotes consistent kinetics. The subsequent workup procedure described in the patent—quenching with saturated brine followed by simple extraction and evaporation—is highly effective at removing succinimide by-products and unreacted starting materials. This simplicity in purification is a major advantage for R&D teams aiming for rapid iteration, as it avoids the need for complex chromatography columns typically required for crude reaction mixtures from acid-catalyzed routes. Furthermore, the downstream utility of the bromine handle at the C2 position is exemplified by its ability to undergo palladium-catalyzed cross-coupling reactions. 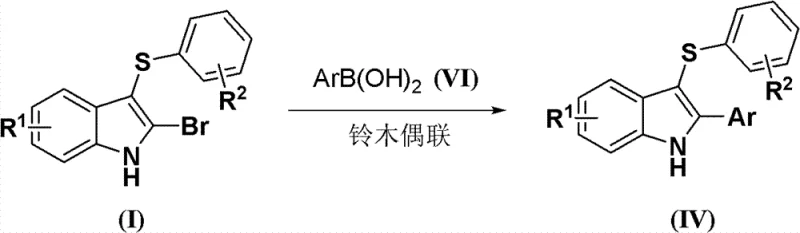 As shown in the conversion to Formula (IV), the bromine atom serves as an excellent leaving group for Suzuki coupling with aryl boronic acids, enabling the rapid diversification of the C2 position to create a library of biologically active analogs.
As shown in the conversion to Formula (IV), the bromine atom serves as an excellent leaving group for Suzuki coupling with aryl boronic acids, enabling the rapid diversification of the C2 position to create a library of biologically active analogs.
How to Synthesize 2-Bromo-3-Arylmercaptoindole Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing precise stoichiometric control and temperature management to maximize yield. The process begins with the dissolution of the indole substrate and N-bromosuccinimide in DMF, followed by the controlled addition of the diaryldisulfide solution to manage exotherms and ensure selectivity. Maintaining the reaction temperature strictly within the 10-30°C window is critical for preventing side reactions, while the molar ratios of diaryldisulfide, indole, and NBS (optimized around 1.0:2.5:5.0) drive the reaction to completion. For detailed operational parameters, safety data, and specific equipment requirements necessary for GMP compliance, please refer to the standardized synthesis steps provided below.
- Dissolve indole derivatives and N-bromosuccinimide (NBS) in N,N-dimethylformamide (DMF) solvent.
- Slowly add a solution of diaryldisulfide in DMF to the reaction mixture while maintaining temperature between 10-30°C.
- Stir for 3.5 to 6 hours, then quench with saturated brine, extract the organic layer, and evaporate to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The shift away from exotic, hard-to-source reagents towards commodity chemicals like indole, diphenyl disulfide, and NBS significantly de-risks the supply chain, ensuring continuity of supply even during global market fluctuations. The elimination of strong acids and the simplification of the workup procedure translate directly into reduced waste treatment costs and lower capital expenditure on corrosion-resistant reactor infrastructure. This process intensification allows manufacturers to achieve substantial cost savings by shortening the production cycle time and reducing the consumption of solvents and energy compared to multi-step, harsh-condition alternatives.
- Cost Reduction in Manufacturing: The economic model of this synthesis is driven by the use of low-cost, commercially abundant raw materials that do not require custom synthesis or long lead times for procurement. By avoiding expensive catalysts or specialized reagents often needed in traditional thiolation methods, the direct material costs are significantly lowered. Additionally, the high atom economy and minimal by-product formation reduce the burden on downstream purification units, leading to lower operational expenditures (OPEX) associated with solvent recovery and waste disposal. The ability to achieve high purity (over 98%) directly from crystallization or simple evaporation further reduces the need for resource-intensive chromatographic purification steps.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical metric for any long-term manufacturing partnership, and this route excels by utilizing reagents that are standard stock items in the global fine chemical market. Unlike proprietary intermediates that tie a buyer to a single vendor, the starting materials for Formula (I) are produced by multiple suppliers worldwide, fostering a competitive pricing environment and mitigating the risk of supply disruption. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain volatility and ensuring consistent output quality for the final API manufacturer.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often exposes hidden safety and environmental hazards, but this methodology is inherently designed for scalability. The mild operating temperatures eliminate the need for extreme heating or cooling utilities, simplifying the engineering requirements for large-scale reactors. Furthermore, the absence of heavy metal catalysts in the initial step and the generation of benign succinimide as a by-product align well with modern green chemistry principles and stringent environmental regulations. This facilitates easier permitting and reduces the environmental footprint of the manufacturing site, a key consideration for sustainable supply chain strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indole derivatives, based on the specific data and claims found within the patent literature. Understanding these details helps stakeholders make informed decisions regarding process adoption and material sourcing. The answers provided reflect the verified experimental outcomes and scope defined in the intellectual property documentation.
Q: What are the key advantages of this synthesis method over traditional routes?
A: Unlike conventional methods requiring strong acidic conditions which limit functional group tolerance, this patent utilizes mild temperatures (10-30°C) and neutral conditions, resulting in higher yields (up to 81%) and easier purification.
Q: Can this intermediate be used for synthesizing sulfonyl derivatives?
A: Yes, the 2-bromo-3-arylmercaptoindole serves as a versatile precursor. It can undergo Suzuki coupling to form 2-aryl-3-arylmercaptoindoles, which can subsequently be oxidized to biologically active 2-aryl-3-arylsulfonylindoles.
Q: Is this process suitable for large-scale commercial production?
A: Absolutely. The use of readily available raw materials like indole and diaryldisulfides, combined with simple workup procedures (brine wash and evaporation), makes this route highly scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-3-Arylmercaptoindole Supplier
The technological advancements detailed in CN102584675B represent a significant opportunity for the pharmaceutical industry to access high-quality antibacterial intermediates more efficiently. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the identity and purity of every batch, guaranteeing that the 2-bromo-3-arylmercaptoindole supplied meets the exacting standards required for subsequent Suzuki coupling and oxidation steps. We understand that consistency is key to successful drug development, and our manufacturing processes are designed to deliver that reliability.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this method for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a seamless transition from R&D to commercial manufacturing.
