Advanced Manufacturing of Alpha-Ketophenylalanine Calcium for Uremia Treatment Applications
Advanced Manufacturing of Alpha-Ketophenylalanine Calcium for Uremia Treatment Applications
The pharmaceutical landscape for uremia treatment relies heavily on the availability of high-purity alpha-keto acid compounds, specifically Alpha-Ketophenylalanine Calcium, which serves as a critical nitrogen scavenger to reduce urea synthesis in patients with renal insufficiency. Patent CN102050725B introduces a transformative manufacturing methodology that addresses the longstanding inefficiencies and safety hazards associated with traditional synthesis routes. This innovative approach utilizes a streamlined two-step sequence involving a one-pot catalytic cyclization followed by a controlled pressurized hydrolysis, fundamentally altering the economic and environmental profile of producing this essential medical material. By shifting away from toxic cyanide-based precursors and harsh oxidative conditions, this technology offers a robust platform for the reliable Alpha-Ketophenylalanine Calcium supplier market, ensuring consistent quality and supply continuity for global pharmaceutical manufacturers.
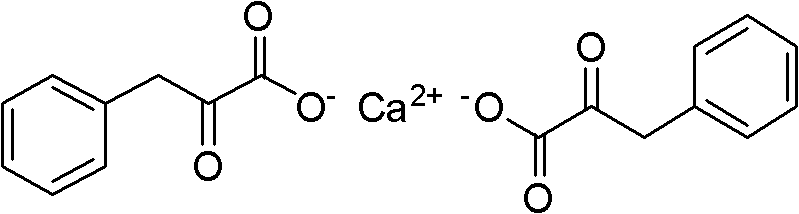
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Alpha-Ketophenylalanine Calcium has been plagued by significant operational drawbacks that hinder scalability and safety compliance. One prevalent legacy method involves the Knoevenagel-Cope condensation of ethyl cyanoacetate with benzaldehyde, a pathway that necessitates six distinct synthetic steps and relies on ethyl cyanoacetate, a raw material known for its high toxicity and handling risks. Furthermore, this route requires an oxidation step utilizing hydrogen peroxide, which introduces severe safety concerns regarding explosion hazards and thermal runaway in large-scale reactors, making it unfavorable for modern safety production standards. Another existing alternative utilizes glycolylurea as a starting material; however, this method suffers from extremely difficult hydrolysis conditions that require massive quantities of alkali lye and prolonged reaction times under harsh environments. These conventional processes not only result in low hydrolysis yields but also generate substantial volumes of saline wastewater due to the necessary neutralization steps, creating a heavy burden on waste treatment facilities and increasing the overall cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the patented methodology presents a highly efficient and environmentally benign synthetic strategy centered on the use of pharmaceutical-grade glycine, benzaldehyde, and acetic anhydride. The core innovation lies in the adoption of a one-pot catalytic cyclization reaction that directly converts these readily available commodity chemicals into the key intermediate, 4-benzylidene-2-methyl dihydro-oxazole ketone, with exceptional efficiency. This intermediate is subsequently subjected to a ring-opening and hydrolysis reaction within an autoclave pressure vessel in the presence of calcium hydroxide, bypassing the need for sodium hydroxide and the associated neutralization waste. The entire process is characterized by mild reaction conditions that are easy to control, significantly reducing the possibility of side reactions and thereby improving the overall reaction yield compared to atmospheric hydrolysis methods. This streamlined workflow represents a paradigm shift for the commercial scale-up of complex pharmaceutical intermediates, offering a cleaner and more economically viable production model.
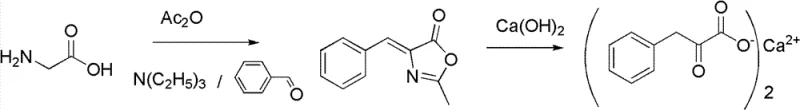
Mechanistic Insights into One-Pot Catalytic Cyclization and Pressurized Hydrolysis
The chemical elegance of this process is rooted in the precise manipulation of reaction kinetics through the use of specific organic base catalysts during the cyclization phase. The reaction initiates with the condensation of glycine and acetic anhydride to form an N-acetylglycine intermediate, which subsequently undergoes dehydration and condensation with benzaldehyde to form the oxazolone ring structure. The selection of the catalyst is critical, with the patent specifying a range of effective organic bases including aliphatic tertiary amines like tri-n-propylamine and triethylamine, as well as heterocyclic amines such as DBU and pyridine. These catalysts facilitate the formation of the 4-benzylidene-2-methyl dihydro-oxazole ketone by promoting the necessary proton transfers and stabilizing the transition states without introducing metallic impurities that would require costly removal steps later in the purification process. The ability to tune the catalyst loading between 0.1 to 2.0 molar equivalents allows for fine optimization of the reaction rate and selectivity, ensuring high conversion of the starting glycine.
Following the cyclization, the mechanistic advantage shifts to the hydrolysis step, where the stability of the amido linkage in the oxazolone intermediate is exploited to achieve selective ring opening. Unlike ester bonds which might hydrolyze too rapidly or uncontrollably, the specific electronic environment of this oxazolone allows for a controlled reaction with calcium hydroxide under pressurized conditions. By operating the autoclave at a gauge pressure of 0.2 to 0.3 MPa and temperatures between 120 to 130°C, the system achieves a rapid reaction rate that effectively suppresses the degradation of the sensitive alpha-keto acid moiety. This pressurized environment ensures that the water remains in a liquid phase at elevated temperatures, enhancing the solubility of the reactants and the collision frequency, which leads to the observed high yields of approximately 85% in the hydrolysis step. Furthermore, the direct use of calcium hydroxide ensures that the final product precipitates directly as the calcium salt, eliminating the need for a separate salt formation step and minimizing the generation of inorganic salt byproducts.
How to Synthesize Alpha-Ketophenylalanine Calcium Efficiently
The implementation of this synthesis route requires careful attention to the sequential addition of reagents and the management of exothermic events during the acetylation and condensation phases. The process begins with the suspension of pharmaceutical-grade glycine in a reactor equipped with efficient stirring and cooling capabilities, followed by the controlled addition of the organic base catalyst and acetic anhydride to maintain the temperature below 50°C. Once the N-acetylation is complete, benzaldehyde is introduced, and the mixture is heated to promote the cyclization, after which the volatile byproducts and excess reagents are recovered via distillation for reuse. The resulting solid intermediate is then transferred to a pressure-resistant reactor where it is mixed with water and calcium hydroxide for the final hydrolysis transformation. For the detailed standardized synthesis steps and specific parameter optimizations, please refer to the guide below.
- Perform a one-pot catalytic cyclization reaction using glycine, benzaldehyde, acetic anhydride, and an organic base catalyst to form 4-benzylidene-2-methyl dihydro-oxazole ketone.
- Conduct a ring-opening and hydrolysis reaction in an autoclave pressure vessel using calcium hydroxide to convert the intermediate into Alpha-Ketophenylalanine Calcium.
- Recover and recycle the organic amine catalyst and acetic acid byproducts via distillation to minimize waste and reduce operational costs.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented technology offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility often encountered in the fine chemical sector. The substitution of toxic and regulated raw materials like ethyl cyanoacetate with widely available commodity chemicals such as glycine and benzaldehyde drastically simplifies the sourcing logistics and reduces the regulatory burden associated with hazardous material transport and storage. Moreover, the elimination of hydrogen peroxide from the process removes a significant safety liability, potentially lowering insurance premiums and facility safety compliance costs for manufacturing partners. The inherent design of the process, which allows for the recovery and recycling of the organic amine catalyst and acetic acid, creates a closed-loop system that minimizes raw material consumption and waste disposal fees. This holistic approach to process design ensures that the cost reduction in pharmaceutical intermediates manufacturing is achieved not just through yield improvements, but through fundamental structural efficiencies in the material flow.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the recoverability of key reagents, specifically the organic amine catalyst and acetic anhydride, which can be distilled and reused in subsequent batches without loss of efficacy. By avoiding the use of expensive transition metal catalysts or stoichiometric oxidants that generate heavy metal waste, the process eliminates the need for costly purification steps such as chromatography or specialized scavenging resins. The direct formation of the calcium salt during hydrolysis further consolidates the downstream processing, removing unit operations related to acidification, basification, and salt metathesis that typically inflate production costs. Consequently, the overall cost of goods sold is substantially lowered, providing a competitive pricing structure for bulk purchasers.
- Enhanced Supply Chain Reliability: The reliance on pharmaceutical-grade glycine and basic aromatic aldehydes ensures a robust supply chain, as these feedstocks are produced globally at massive scales by numerous chemical manufacturers, mitigating the risk of single-source bottlenecks. The simplified two-step nature of the synthesis reduces the total cycle time required to produce a batch of finished API intermediate, allowing for more responsive inventory management and shorter lead times for high-purity pharmaceutical intermediates. Additionally, the mild reaction conditions and the use of standard stainless steel pressure vessels mean that the technology can be easily transferred to multiple contract manufacturing organizations (CMOs) without requiring specialized or exotic reactor hardware. This flexibility enhances supply continuity and allows buyers to diversify their manufacturing base effectively.
- Scalability and Environmental Compliance: Environmental sustainability is a core pillar of this technology, as the one-pot cyclization step generates no wastewater, and the hydrolysis step produces minimal effluent due to the absence of neutralization salts. The ability to recycle solvents and reagents internally aligns perfectly with increasingly stringent global environmental regulations, reducing the carbon footprint and waste disposal costs associated with production. The process is inherently scalable, moving seamlessly from laboratory glassware to industrial-tonnage pressure reactors without encountering the heat transfer or mixing limitations that often plague multi-step batch processes. This scalability ensures that suppliers can meet surging demand for uremia treatments without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific synthetic route for Alpha-Ketophenylalanine Calcium. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for their own production lines or for procurement teams assessing supplier qualifications.
Q: What are the safety advantages of this new synthesis method compared to traditional routes?
A: Unlike conventional methods that utilize toxic ethyl cyanoacetate or hazardous hydrogen peroxide oxidants, this patented process employs pharmaceutical-grade glycine and benign organic amine catalysts, significantly reducing toxicity risks and eliminating the need for dangerous oxidation steps.
Q: How does the pressurized hydrolysis step improve product yield?
A: By conducting the hydrolysis of the oxazolone intermediate in an autoclave under controlled pressure (0.2-0.3 MPa) and temperature (120-130°C), the reaction kinetics are accelerated while suppressing side reactions, leading to higher conversion rates compared to atmospheric hydrolysis.
Q: Is the organic catalyst recoverable in this process?
A: Yes, the process is designed for sustainability; the organic amine catalyst, along with excess acetic anhydride and acetic acid, can be recovered via steam distillation after the cyclization step and reused in subsequent batches, lowering material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Ketophenylalanine Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving uremia treatments, and we are uniquely positioned to leverage advanced synthetic technologies like the one described in CN102050725B. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Alpha-Ketophenylalanine Calcium meets the exacting standards required for pharmaceutical applications, providing our partners with absolute confidence in material consistency.
We invite global pharmaceutical companies and procurement leaders to engage with us to explore how this optimized synthesis route can enhance your supply chain resilience and cost structure. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us collaborate to bring safer, more efficient, and cost-effective medical materials to the market, driving value for your organization and improving patient outcomes worldwide.
