Scalable Production of Chiral 1,2-Bis(2,5-Diphenylphosphino)Ethane via Safe Grignard Coupling
The landscape of chiral ligand manufacturing is undergoing a significant transformation driven by the urgent need for safer and more efficient synthetic routes. Patent CN110878104B introduces a groundbreaking preparation method for chiral 1,2-bis(2,5-diphenylphosphino)ethane, a critical precursor for rhodium-based asymmetric hydrogenation catalysts. This technology addresses long-standing safety hazards and inefficiencies associated with traditional synthesis, offering a robust pathway for the production of high-value pharmaceutical intermediates. By utilizing a streamlined Grignard coupling strategy followed by a mild silane reduction, the process eliminates the reliance on extremely pyrophoric reagents that have historically plagued this sector. For R&D directors and supply chain managers, this patent represents a pivotal shift towards sustainable and scalable chemistry, ensuring a reliable supply of high-purity ligands essential for synthesizing unnatural chiral amino acids and other complex therapeutic structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 1,2-bis(2,5-diphenylphosphino)ethane has been fraught with significant operational challenges and safety risks that hinder large-scale industrial adoption. Conventional literature routes typically involve a cumbersome four-step sequence that necessitates the use of highly dangerous reagents such as lithium aluminum hydride or phenyltrichlorosilane. These substances are notoriously pyrophoric, meaning they can ignite spontaneously upon contact with air or moisture, requiring stringent inert atmosphere controls and specialized equipment that drive up capital expenditure. Furthermore, the multi-step nature of these legacy processes results in cumulative yield losses and generates substantial volumes of hazardous waste, complicating environmental compliance and disposal logistics. The reliance on borane reagents and butyllithium for coupling and reduction further exacerbates the safety profile, making the process unsuitable for facilities without advanced hazard management capabilities. Consequently, the high cost of goods and the inherent danger of the chemistry have created bottlenecks in the supply chain for this vital catalytic ligand.
The Novel Approach
In stark contrast to the perilous legacy methods, the novel approach detailed in the patent simplifies the entire synthetic architecture into a highly efficient two-step sequence that prioritizes safety and yield. The process initiates with a direct Grignard coupling reaction, where 1,2-ethylene dibromide magnesium is reacted with chiral 2,5-diphenylphosphinic chloride in a tetrahydrofuran medium. This step effectively constructs the carbon-phosphorus backbone under controlled thermal conditions, avoiding the extreme reactivity associated with organolithium reagents. The subsequent reduction of the phosphine oxide intermediate is achieved using trichlorosilane and a benign organic amine base, replacing the hazardous lithium aluminum hydride entirely. This strategic substitution not only mitigates fire risks but also streamlines the workup procedure, allowing for straightforward aqueous quenching and extraction. The result is a chemically robust protocol that maintains high stereochemical integrity while drastically reducing the complexity of the manufacturing workflow.
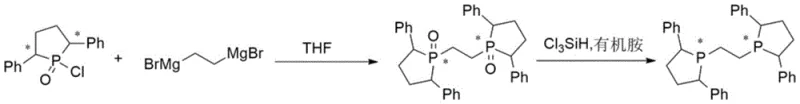
Mechanistic Insights into Grignard Coupling and Silane Reduction
The core of this technological advancement lies in the precise mechanistic execution of the nucleophilic substitution and subsequent deoxygenation steps. In the first stage, the Grignard reagent acts as a potent nucleophile, attacking the electrophilic phosphorus center of the chiral phosphinic chloride. This reaction proceeds through a concerted mechanism where the magnesium species facilitates the displacement of the chloride leaving group, forming a new phosphorus-carbon bond with high fidelity. The use of tetrahydrofuran as the solvent is critical here, as it stabilizes the magnesium species and ensures homogeneous reaction conditions, preventing the formation of insoluble aggregates that could trap impurities. Temperature control during the addition phase, specifically maintaining the range between 0°C and 10°C, is essential to suppress side reactions such as Wurtz-type coupling of the Grignard reagent itself, thereby ensuring the chemical purity of the bis-phosphine oxide intermediate exceeds 98%.
The second mechanistic phase involves the reduction of the phosphine oxide (P=O) bond to the phosphine (P) functionality, a transformation that is traditionally difficult to achieve without harsh conditions. The patent utilizes a system comprising trichlorosilane and triethylamine, which operates through the in situ formation of a reactive silyl species. The amine base activates the trichlorosilane, generating a hypervalent silicon complex that coordinates strongly with the oxygen atom of the phosphine oxide. This coordination weakens the phosphorus-oxygen double bond, facilitating a hydride transfer from the silicon center to the phosphorus atom. The resulting silyl ether byproduct is easily hydrolyzed during the aqueous sodium hydroxide quench, driving the equilibrium towards the desired phosphine product. This mechanism is exceptionally clean, avoiding the formation of aluminum salts or boron residues that are difficult to remove, thus contributing to the final product's high purity and suitability for sensitive catalytic applications.
How to Synthesize Chiral 1,2-Bis(2,5-Diphenylphosphino)Ethane Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and thermal management to maximize the benefits of the patented route. The process begins with the preparation of the Grignard reagent, which must be titrated accurately to ensure the correct molar ratio relative to the phosphinic chloride, typically ranging from 1:2.2 to 1:3.3 to drive the reaction to completion. Following the coupling and isolation of the oxide intermediate, the reduction step demands anhydrous conditions in toluene to prevent premature decomposition of the trichlorosilane. The detailed standardized operating procedures, including specific addition rates, reflux times, and crystallization protocols necessary to achieve the reported 99% chiral purity, are outlined in the structured guide below.
- Perform a Grignard coupling reaction by dropwise adding 1,2-ethylene dibromide magnesium solution into chiral 2,5-diphenylphosphinic chloride in THF at 0-10°C, then reflux to form the bis-phosphine oxide intermediate.
- Dissolve the isolated chiral 1,2-bis(2,5-diphenylphosphine oxide) ethane in toluene and add trichlorosilane along with an organic amine base such as triethylamine.
- Heat the mixture to reflux for reduction, quench carefully with aqueous sodium hydroxide, and proceed through extraction, concentration, and ethanol crystallization to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages regarding cost structure and operational continuity. The elimination of high-risk reagents like lithium aluminum hydride removes the need for expensive safety infrastructure and specialized storage facilities, directly lowering the overhead costs associated with manufacturing. Furthermore, the reduction in synthetic steps from four to two significantly decreases the consumption of solvents and utilities, leading to a leaner production process with a smaller environmental footprint. This efficiency gain allows for faster batch turnover times, enhancing the overall responsiveness of the supply chain to market demands for chiral ligands. By securing a supply source that utilizes this safer and more efficient technology, companies can mitigate the risks of production stoppages due to safety incidents or regulatory scrutiny.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the synthetic route and the substitution of costly raw materials. By removing the need for expensive and hazardous reducing agents, the direct material costs are significantly optimized, while the reduced number of unit operations lowers labor and energy expenditures. The high yields reported in the patent examples indicate minimal material loss, ensuring that the theoretical cost savings are realized in practical production scenarios. Additionally, the simplified workup procedures reduce the volume of waste streams, lowering disposal costs and easing the burden on wastewater treatment facilities.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials, such as Grignard reagents and trichlorosilane, ensures a robust supply chain that is less susceptible to the volatility often seen with specialty fine chemicals. Unlike processes dependent on bespoke or highly regulated precursors, this route leverages commodity chemicals that are readily sourced from multiple vendors, reducing single-source dependency risks. The improved safety profile also means that manufacturing sites face fewer regulatory hurdles and inspection delays, guaranteeing a more consistent and predictable delivery schedule for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: From a scale-up perspective, the exothermic profiles of the reactions described are manageable and well-suited for large-scale reactors, unlike the violent reactions associated with traditional hydride reductions. The ability to quench the reaction with standard aqueous sodium hydroxide simplifies the containment of hazardous byproducts, aligning with increasingly stringent global environmental regulations. This compliance readiness future-proofs the manufacturing asset, ensuring long-term viability without the need for costly retrofits to handle toxic or pyrophoric waste streams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on performance metrics and operational requirements. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this ligand into their catalytic processes or supply chains.
Q: Why is the new synthesis route safer than conventional methods?
A: Conventional methods rely on highly pyrophoric reagents like lithium aluminum hydride or phenyltrichlorosilane which pose severe fire risks. The patented process replaces these with standard Grignard reagents and trichlorosilane, significantly improving operational safety and reducing the need for specialized hazardous material handling infrastructure.
Q: What is the chiral purity achievable with this method?
A: The process demonstrates exceptional stereochemical retention, consistently achieving chiral purity levels exceeding 99% as verified by NMR and chiral HPLC analysis in the patent examples. This high optical purity is critical for its application in asymmetric hydrogenation catalysts where enantiomeric excess directly impacts drug quality.
Q: How does this process impact manufacturing costs?
A: By condensing the synthesis from a complex four-step sequence into just two efficient steps, the method drastically reduces solvent consumption, labor hours, and waste generation. Furthermore, the avoidance of expensive and dangerous reducing agents like lithium aluminum hydride leads to substantial raw material cost savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 1,2-Bis(2,5-Diphenylphosphino)Ethane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance chiral ligands play in the development of next-generation pharmaceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our facility is equipped to handle the specific safety requirements of phosphine chemistry, guaranteeing a secure and consistent supply of this essential intermediate for your asymmetric synthesis needs.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your specific applications. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized process can enhance your project's efficiency and profitability.
