Scalable Chromium-Catalyzed Fluorination for High-Purity 2-Amino-4-Fluoropyridine Production
Introduction to Novel Catalytic Fluorination Technology
The pharmaceutical industry continuously seeks efficient pathways for synthesizing fluorinated heterocycles, which are critical scaffolds in modern drug design, particularly for kinase inhibitors and URAT-1 inhibitors. Patent CN112552233A introduces a groundbreaking synthetic method for 2-amino-4-fluoropyridine, a key intermediate that has traditionally been difficult to access cost-effectively. This technology leverages a specialized Chromium-based catalyst to achieve direct C-H fluorination on a pyridine ring, bypassing the need for pre-functionalized halogenated starting materials. By utilizing cheap 2-pyridinecarboxylic acid as the feedstock, this innovation addresses both economic and environmental pain points associated with legacy manufacturing processes. The strategic integration of a robust oxidation system allows for high regioselectivity, ensuring that the fluorine atom is installed precisely at the 4-position relative to the nitrogen atom.
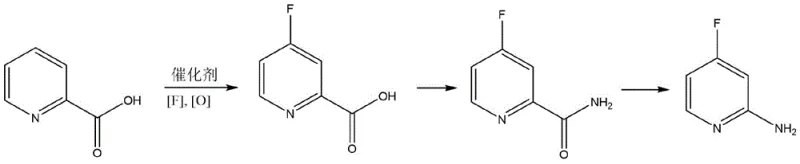
Furthermore, the process flow is remarkably concise, condensing the synthesis into just three distinct chemical transformations that are highly amenable to continuous processing or large-batch reactor operations. The initial fluorination step sets the stage for subsequent functional group manipulations, specifically the conversion to an amide and finally to the amine via degradation. This streamlined approach not only reduces the cumulative yield loss typical of multi-step syntheses but also minimizes the solvent usage and energy consumption per kilogram of product. For R&D teams evaluating new supply chains, this patent represents a significant leap forward in process chemistry, offering a viable alternative to routes that rely on scarce resources or generate excessive toxic byproducts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2-amino-4-fluoropyridine has been hindered by reliance on inefficient and costly methodologies that struggle to meet modern green chemistry standards. One prominent prior art route, such as that described in WO201100790761, depends heavily on Palladium-catalyzed cross-coupling reactions starting from 2-chloro-4-fluoropyridine. While chemically feasible, the use of noble metal catalysts introduces substantial cost volatility and necessitates complex purification protocols to reduce metal residues to ppm levels acceptable for pharmaceutical use. Additionally, the requirement for protecting group strategies, such as Boc-protection, adds unnecessary synthetic steps, lowering the overall throughput and increasing the material footprint of the process.
Another conventional pathway involves mixed-acid nitration technologies, which are notoriously hazardous and environmentally burdensome. These methods typically employ large excesses of sulfuric and nitric acids, leading to the generation of massive quantities of acidic wastewater that require expensive neutralization and treatment facilities. The harsh reaction conditions associated with nitration also pose significant safety risks, including the potential for thermal runaways and the formation of unstable intermediates. Consequently, these legacy methods are increasingly viewed as unsustainable for large-scale commercial production, driving the urgent need for catalytic alternatives that can operate under milder, safer, and more atom-economical conditions.
The Novel Approach
In stark contrast to these established limitations, the novel approach detailed in the patent utilizes a direct C-H activation strategy that fundamentally reshapes the economic landscape of producing this valuable intermediate. By employing a Chromium-NHC (N-Heterocyclic Carbene) catalyst system, the process achieves selective fluorination directly on the unactivated pyridine ring of 2-pyridinecarboxylic acid. This eliminates the need for pre-halogenated substrates and expensive noble metals, replacing them with earth-abundant Chromium and inexpensive fluoride salts like potassium fluoride or ammonium fluoride. The reaction conditions are notably mild, typically operating between 60°C and 80°C under an oxygen atmosphere or with peroxide oxidants, which significantly enhances operational safety.
Moreover, the subsequent steps involving amidation and Hofmann degradation are classic, high-yielding transformations that utilize commodity chemicals such as chloroformates and sodium hypochlorite. This combination of cutting-edge catalysis with robust, well-understood organic transformations creates a hybrid process that is both innovative and practically deployable. The result is a synthesis that avoids the pitfalls of heavy metal contamination and acidic waste streams, offering a cleaner, faster, and more cost-effective route to market. For procurement specialists, this shift translates directly into a more stable supply base with reduced exposure to the price fluctuations of precious metals and specialized reagents.
Mechanistic Insights into Chromium-Catalyzed C-H Fluorination
The core of this technological breakthrough lies in the unique structure and reactivity of the Chromium-NHC catalyst, which facilitates the challenging activation of the C-H bond at the 4-position of the pyridine ring. As illustrated in the catalyst structure, the central Chromium ion is coordinated by two N-heterocyclic carbene ligands derived from a bis-imidazolium salt backbone. This specific coordination geometry creates a highly electron-rich metal center capable of interacting with the pyridine substrate and the oxidant to form a reactive metallacycle intermediate. The presence of bulky substituents on the ligand framework, such as isopropyl or phenyl groups, plays a crucial role in directing the regioselectivity of the fluorination, ensuring that the fluorine atom is installed exclusively at the desired position rather than forming a mixture of isomers.
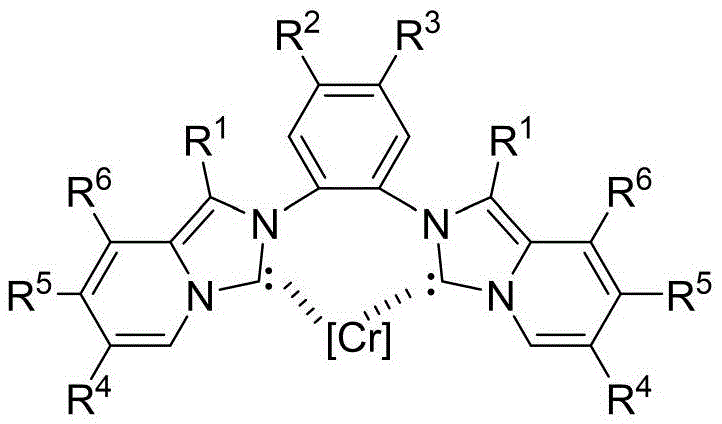
From an impurity control perspective, this catalytic system offers superior selectivity compared to electrophilic fluorination reagents which often lack discrimination between similar C-H bonds. The mechanism likely proceeds through a high-valent Chromium-fluoride species that transfers the fluorine atom to the activated carbon center with high fidelity. This precision minimizes the formation of regioisomeric byproducts, such as 3-fluoro or 5-fluoro derivatives, which are notoriously difficult to separate due to their similar physical properties. By suppressing these side reactions at the molecular level, the process simplifies downstream purification, potentially allowing for crystallization-based isolation rather than resource-intensive chromatography. This level of control is essential for maintaining the stringent purity specifications required for pharmaceutical intermediates intended for clinical applications.
How to Synthesize 2-Amino-4-Fluoropyridine Efficiently
The practical implementation of this synthesis involves a logical sequence of three steps that transform a simple carboxylic acid into the target amine with high efficiency. The process begins with the catalytic fluorination of 2-pyridinecarboxylic acid in a polar aprotic solvent like DMF or acetonitrile, using a fluoride source and an oxidant under controlled temperature conditions. Following the isolation of the 4-fluoropyridine-2-carboxylic acid intermediate, the second step involves activation of the carboxylic acid with a chloroformate in the presence of an organic base, followed by treatment with an ammonia source to generate the corresponding primary amide. The final transformation utilizes a Hofmann degradation protocol, where the amide is treated with a hypohalite solution to effect the loss of the carbonyl group and formation of the primary amine.
- Step 1: React 2-pyridinecarboxylic acid with a fluoride source and oxidant using a specialized Chromium-NHC catalyst to achieve regioselective C-H fluorination at the 4-position.
- Step 2: Convert the resulting 4-fluoropyridine-2-carboxylic acid into its corresponding amide using chloroformate activation and an ammonia source.
- Step 3: Perform a Hofmann amide degradation reaction on the 4-fluoropyridine-2-formamide using hypochlorite to yield the final 2-amino-4-fluoropyridine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic advantages that extend beyond simple unit cost calculations. The primary driver of value is the substitution of expensive, supply-constrained raw materials with abundant commodity chemicals. By eliminating the dependency on Palladium catalysts and protected halogenated pyridines, manufacturers can insulate their production costs from the volatility of the precious metals market and the geopolitical risks associated with specialized fine chemical suppliers. Furthermore, the use of common reagents like potassium fluoride and sodium hypochlorite ensures that the supply chain is resilient and capable of scaling rapidly to meet surges in demand without long lead times for custom synthesis.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic simplification of the raw material bill and the reduction in waste disposal costs. Eliminating noble metal catalysts removes the need for expensive recovery systems or scavenging resins, which are significant line items in the operating budget of API manufacturing. Additionally, the high atom economy of the direct fluorination step means that less raw material is wasted as byproducts, leading to a higher effective yield per ton of input. The avoidance of mixed-acid nitration also removes the heavy burden of treating large volumes of corrosive acidic waste, resulting in substantial savings in environmental compliance and waste management expenditures.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials significantly de-risks the supply chain against disruptions. 2-Pyridinecarboxylic acid is a bulk chemical produced by multiple global vendors, ensuring a competitive pricing environment and consistent availability. Unlike complex custom intermediates that may have single-source suppliers, the commoditized nature of the inputs for this route allows for flexible sourcing strategies. This flexibility is crucial for maintaining business continuity, especially in a global market where logistics bottlenecks can delay the delivery of specialized reagents. The robustness of the chemistry also means that production can be easily transferred between different manufacturing sites without requiring highly specialized equipment or expertise.
- Scalability and Environmental Compliance: From a sustainability and scaling perspective, this route is exceptionally well-suited for expansion from pilot plant to commercial tonnage. The reaction conditions are mild and do not require extreme pressures or cryogenic temperatures, allowing the use of standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. The reduction in hazardous waste generation aligns perfectly with increasingly strict environmental regulations, facilitating easier permitting and community acceptance for manufacturing facilities. The simplified work-up procedures, often involving straightforward extraction and crystallization, reduce the cycle time per batch, thereby increasing the overall capacity utilization of the production assets and enabling faster time-to-market for downstream drug products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chromium-catalyzed synthesis for 2-amino-4-fluoropyridine. These answers are derived directly from the technical disclosures and experimental data provided in the patent literature, focusing on the practical implications for industrial application. Understanding these details is critical for technical teams assessing the feasibility of integrating this new route into their existing manufacturing portfolios.
Q: What are the advantages of the Chromium-NHC catalyst over traditional Palladium methods?
A: The Chromium-NHC catalyst system described in CN112552233A utilizes earth-abundant Chromium instead of expensive noble metals like Palladium. This significantly lowers raw material costs and eliminates the need for rigorous heavy metal scavenging steps often required for Pd residues in pharmaceutical APIs, thereby simplifying downstream purification and reducing overall production costs.
Q: How does this route improve safety compared to mixed-acid nitration?
A: Traditional routes often rely on mixed-acid nitration which generates large volumes of acidic waste and poses significant thermal runaway risks. The new catalytic fluorination route operates under milder conditions (60-80°C) with oxygen or peroxides as oxidants, drastically reducing the generation of hazardous 'three wastes' (waste water, gas, and residue) and improving the overall safety profile for industrial scale-up.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is explicitly designed for industrial production. It uses cheap and readily available starting materials like 2-pyridinecarboxylic acid and common reagents like potassium fluoride and sodium hypochlorite. The short three-step sequence with high yields (up to 91% in the first step) ensures a robust supply chain capable of meeting the demands of large-volume API synthesis without complex operational constraints.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4-Fluoropyridine Supplier
As the demand for fluorinated pyridine intermediates continues to grow across the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO is essential for securing a stable supply. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and reliability. Our technical team is adept at optimizing complex catalytic processes, such as the Chromium-NHC fluorination described here, to achieve stringent purity specifications required for GMP manufacturing. We maintain rigorous QC labs equipped with advanced analytical instrumentation to monitor every batch for impurities, ensuring that the final product meets the highest quality standards for API synthesis.
We invite you to engage with our technical procurement team to discuss how this innovative synthetic route can benefit your specific project needs. By leveraging our expertise in process development and scale-up, we can provide a Customized Cost-Saving Analysis tailored to your production volumes and quality targets. Please contact us to request specific COA data for our reference batches and to receive detailed route feasibility assessments that demonstrate the commercial viability of this technology for your supply chain.
