Scalable Chemical Synthesis of High-Purity L-Theanine via Asymmetric Michael Addition
Scalable Chemical Synthesis of High-Purity L-Theanine via Asymmetric Michael Addition
The global demand for L-theanine, a unique amino acid predominantly found in tea leaves known for its relaxation and neuroprotective properties, has necessitated the development of robust, non-agricultural production methods. Patent CN101717347B introduces a sophisticated chemical methodology that transcends the limitations of traditional extraction and enzymatic routes by employing a chiral auxiliary-based asymmetric synthesis. This technology leverages the stereoselective power of a glycine Schiff base Ni(II) complex derived from the chiral auxiliary BPB (2-[N-(N’-benzyl-prolyl)amino]benzophenone) to construct the L-theanine skeleton with high fidelity. By shifting production from biological sources to a controlled chemical environment, manufacturers can achieve consistent quality and supply continuity, addressing the critical needs of a reliable amino acid intermediate supplier in the nutraceutical and pharmaceutical sectors.
The significance of this patent lies in its ability to decouple L-theanine production from the fluctuations of the tea harvest, offering a pathway to high-purity L-theanine that meets stringent regulatory standards for food and drug applications. The process is characterized by its thermodynamic control over chirality, ensuring that the final product possesses the biologically active L-configuration without the need for complex resolution steps often required in racemic chemical syntheses. For R&D directors and procurement specialists, this represents a pivotal shift towards a more predictable and scalable supply chain model, where the quality of the active ingredient is defined by molecular design rather than agricultural variance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial acquisition of L-theanine has relied heavily on extraction from low-grade tea leaves or plant cell culture, methods that are intrinsically bound by the seasonality of agriculture and the low concentration of the target molecule in biomass. Extraction processes are notoriously cumbersome, involving multiple purification stages to isolate the amino acid from a complex matrix of polyphenols and other alkaloids, resulting in low overall yields and high operational costs that make large-scale cost reduction in pharmaceutical intermediate manufacturing difficult to achieve. Furthermore, enzymatic synthesis routes, while specific, often face significant hurdles such as the requirement for expensive ATP regeneration systems when using theanine synthase, or the propensity for hydrolysis and transpeptidation side reactions when using γ-glutamyl transpeptidase, which complicates downstream processing and impurity control.
The Novel Approach
In stark contrast, the chemical method disclosed in CN101717347B utilizes a Michael addition strategy that constructs the glutamine backbone directly from simple, commercially available precursors like ethylamine and acryloyl chloride. This approach eliminates the dependency on biological feedstocks and allows for the commercial scale-up of complex chiral amino acids in standard chemical reactors. The core innovation is the use of the BPB-Ni(II) complex, which acts as a chiral template to direct the incoming electrophile specifically to the Re-face of the glycine equivalent, thereby installing the correct stereochemistry at the alpha-carbon in a single synthetic operation. This not only simplifies the synthetic route but also ensures that the optical purity is locked in by the rigid coordination geometry of the metal complex, providing a level of consistency that biological methods struggle to match.
Mechanistic Insights into BPB-Ni(II) Catalyzed Asymmetric Michael Addition
The heart of this synthesis is the asymmetric Michael addition reaction, where the nucleophilic glycine equivalent, activated within the coordination sphere of the nickel ion, attacks the beta-carbon of N-ethylacrylamide. The chiral auxiliary BPB creates a sterically demanding environment around the nickel center, effectively shielding one face of the planar glycine moiety and forcing the reaction to proceed through a specific transition state that yields the L-isomer. This thermodynamic control is superior to kinetic control methods because the resulting product complex is the most stable diastereomer, minimizing the formation of unwanted D-isomers and simplifying the purification profile. The rigidity of the square-planar Ni(II) geometry is crucial here, as it maintains the spatial arrangement necessary for high enantioselectivity throughout the bond-forming event.
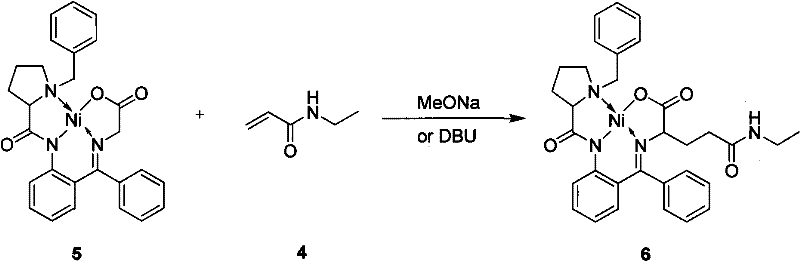
Following the carbon-carbon bond formation, the resulting theanine Schiff base Ni(II) complex retains the chiral information until the final hydrolysis step. The stability of this intermediate allows for rigorous purification, such as column chromatography or crystallization, before the final deprotection, ensuring that any minor impurities formed during the addition step can be removed prior to releasing the free amino acid. This "protect-then-release" strategy is a hallmark of fine chemical synthesis, allowing for reducing lead time for high-purity amino acid intermediates by preventing the propagation of impurities into the final API or nutraceutical ingredient. The ability to recover the BPB ligand and the nickel salt after hydrolysis further underscores the efficiency of this catalytic cycle, making it economically viable for multi-ton production.
How to Synthesize L-Theanine Efficiently
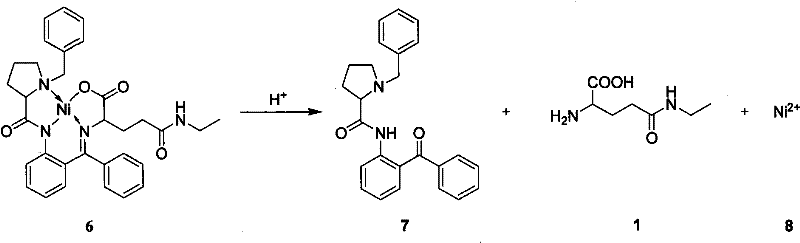
The synthesis protocol outlined in the patent is divided into three distinct operational phases: the preparation of the Michael acceptor (N-ethylacrylamide), the asymmetric coupling reaction, and the final hydrolytic release. The first step involves the reaction of ethylamine with acryloyl chloride in an aqueous alkaline medium, a straightforward acylation that requires careful temperature control to prevent polymerization of the acrylic double bond. The second and most critical step is the coupling of this acceptor with the pre-formed glycine-Ni(II) complex in a polar aprotic solvent like DMF, using a base such as sodium methoxide or DBU to generate the reactive enolate species. The final step involves acidic hydrolysis to cleave the Schiff base and release the free L-theanine, a process that simultaneously regenerates the valuable chiral auxiliary for recycling.
- Prepare N-ethylacrylamide by reacting ethylamine with acryloyl chloride in an aqueous alkaline medium at controlled low temperatures.
- Perform a stereoselective Michael addition between the glycine Schiff base Ni(II) complex and N-ethylacrylamide using MeONa or DBU as the base in DMF.
- Execute acid hydrolysis of the resulting theanine Schiff base Ni(II) complex to release L-theanine, followed by ion exchange purification and catalyst recovery.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this chemical synthesis route offers profound strategic advantages beyond mere technical feasibility. By moving away from agricultural extraction, companies can secure a supply chain that is immune to weather patterns, crop diseases, and geopolitical instability affecting tea-producing regions. The ability to produce L-theanine in a chemical plant allows for precise inventory planning and just-in-time manufacturing, drastically reducing lead time for high-purity amino acid intermediates compared to the months-long cycle of planting and harvesting tea bushes. Furthermore, the chemical route's reliance on bulk petrochemical derivatives like ethylamine and acryloyl chloride ensures a stable and predictable raw material cost structure, shielding the business from the volatility of commodity tea prices.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the recoverability of its key reagents; the chiral auxiliary BPB and the nickel catalyst can be reclaimed from the reaction mixture after hydrolysis with high efficiency, effectively amortizing the cost of these expensive materials over multiple production batches. This closed-loop material usage eliminates the need for continuous purchasing of high-value chiral starters, leading to substantial cost savings in the long term. Additionally, the avoidance of ATP-dependent enzymatic systems removes the burden of maintaining complex biocatalytic infrastructure, further lowering the operational expenditure associated with utility and enzyme replacement costs.
- Enhanced Supply Chain Reliability: Unlike extraction methods that are bottlenecked by the availability of raw tea material, this chemical synthesis can be executed year-round in standard stainless steel reactors, ensuring a continuous flow of product to meet market demand. The raw materials required, such as ethylamine and acryloyl chloride, are commodity chemicals produced on a massive global scale, meaning that supply disruptions are highly unlikely and sourcing can be diversified across multiple vendors. This reliability is critical for pharmaceutical customers who require validated, consistent supply chains to maintain their own regulatory filings and production schedules without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable, as the reaction conditions (temperatures between 0°C and 70°C, atmospheric pressure) are easily manageable in large-scale industrial vessels without requiring specialized high-pressure or cryogenic equipment. From an environmental perspective, the ability to recover and reuse the nickel catalyst minimizes heavy metal waste discharge, aligning with increasingly strict global environmental regulations regarding effluent treatment. The use of standard organic solvents like methanol and ethyl acetate also facilitates solvent recovery and recycling, contributing to a greener manufacturing footprint that appeals to sustainability-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral auxiliary technology for L-theanine production. These insights are derived directly from the experimental data and mechanistic principles described in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: How does the BPB-Ni(II) complex method ensure optical purity compared to extraction?
A: The method utilizes the rigid spatial structure of the chiral BPB-Ni(II) complex to thermodynamically control the stereochemistry during the Michael addition, ensuring the exclusive formation of the L-configuration without the seasonal variability inherent in tea leaf extraction.
Q: Can the chiral auxiliary and nickel catalyst be recovered in this process?
A: Yes, the process is designed for economic efficiency; the chiral auxiliary BPB is recovered from the organic phase after hydrolysis with high yield, and the Ni(II) ions are eluted from the ion exchange resin for reuse, significantly reducing material costs.
Q: What are the key advantages of this chemical route over enzymatic synthesis?
A: Unlike enzymatic methods that often require expensive ATP regeneration systems or suffer from hydrolysis side reactions, this chemical route operates under mild conditions with readily available reagents and avoids the complexity of biocatalyst maintenance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Theanine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the BPB-Ni(II) mediated synthesis route for producing high-quality L-theanine and are fully equipped to translate this laboratory-scale innovation into commercial reality. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate stereochemical controls required for this asymmetric synthesis are maintained even at the largest volumes. We operate with stringent purity specifications and utilize rigorous QC labs to verify the optical purity and chemical identity of every batch, guaranteeing that the L-theanine supplied meets the exacting standards of the global nutraceutical and pharmaceutical industries.
We invite you to collaborate with our technical team to explore how this advanced chemical route can optimize your supply chain and reduce your overall cost of goods. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data from our pilot runs and conduct detailed route feasibility assessments to demonstrate how we can become your strategic partner in securing a stable, high-quality supply of L-theanine.
