Scalable Synthesis of Fluorinated Pyridine Intermediates for Global Pharma Supply Chains
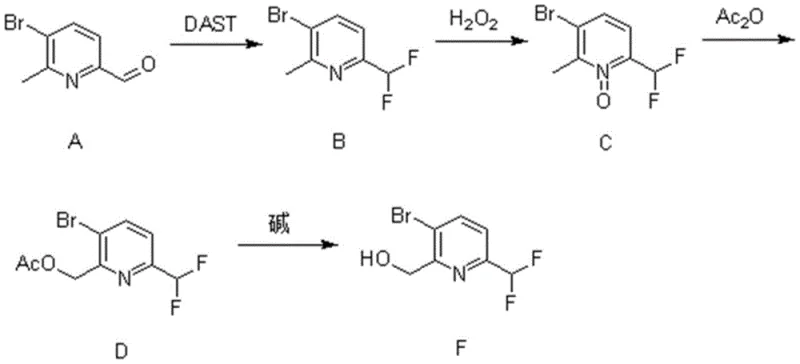
The rapid evolution of fluorine-containing heterocyclic compounds has fundamentally reshaped the landscape of modern medicinal chemistry and agrochemical development, driving an urgent demand for reliable synthetic pathways that can deliver complex building blocks with precision. Patent CN110981792B introduces a groundbreaking methodology for the synthesis of [(3-bromo-6-difluoromethyl) pyridin-2-yl] methanol, a sophisticated intermediate that serves as a critical precursor for next-generation bioactive molecules. This technical disclosure outlines a robust four-step sequence that leverages the unique reactivity of diethylaminosulfur trifluoride (DAST) and oxidative rearrangement strategies to construct the difluoromethyl motif with exceptional fidelity. For global supply chain leaders and R&D directors, understanding the nuances of this patent is essential, as it represents a significant leap forward in accessing fluorinated pyridine scaffolds that were previously difficult to manufacture with consistent quality. The method not only addresses the historical challenges associated with introducing fluorine atoms into sensitive heterocyclic systems but also provides a clear roadmap for scaling these reactions from gram-scale laboratory experiments to multi-ton commercial production facilities without compromising on purity or yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of difluoromethyl groups onto pyridine rings has been plagued by significant technical hurdles that often render conventional synthetic routes economically unviable for large-scale manufacturing. Traditional approaches frequently rely on harsh halogenation conditions or multi-step sequences involving unstable intermediates that degrade rapidly under standard processing temperatures, leading to inconsistent batch-to-batch reproducibility. Furthermore, many legacy methods suffer from poor regioselectivity, resulting in complex mixtures of isomers that require extensive and costly purification processes such as preparative HPLC or repeated recrystallization to achieve the necessary pharmaceutical grade purity. The use of expensive transition metal catalysts in older protocols also introduces the risk of heavy metal contamination, necessitating additional downstream processing steps to meet stringent regulatory limits for residual metals in active pharmaceutical ingredients. These inefficiencies collectively drive up the cost of goods sold and extend lead times, creating bottlenecks that hinder the rapid deployment of new drug candidates into clinical trials and commercial markets.
The Novel Approach
In stark contrast to these outdated methodologies, the novel approach detailed in the patent utilizes a streamlined strategy that maximizes atom economy while minimizing the formation of hazardous byproducts. By initiating the synthesis with a controlled DAST fluorination of a readily available aldehyde precursor, the process establishes the critical carbon-fluorine bonds early in the sequence under mild conditions that preserve the integrity of the bromine substituent. The subsequent oxidation and rearrangement steps exploit the electronic properties of the pyridine N-oxide to direct functionalization specifically to the desired position, effectively eliminating the formation of unwanted regioisomers that plague other methods. This logical progression from aldehyde to alcohol via an acetate intermediate ensures that each transformation proceeds with high conversion rates, as evidenced by the impressive yields reported across multiple experimental examples. The elimination of precious metal catalysts and the use of commodity chemicals like hydrogen peroxide and acetic anhydride further simplify the operational complexity, making this route inherently more suitable for cost reduction in fine chemical manufacturing environments where efficiency and safety are paramount.
Mechanistic Insights into DAST Fluorination and N-Oxide Rearrangement
The cornerstone of this synthetic achievement lies in the precise execution of the DAST-mediated fluorination, which converts the formyl group of the starting material into a difluoromethyl moiety through a well-defined mechanistic pathway. At low temperatures, typically initiated at -78°C, the oxygen atom of the aldehyde attacks the sulfur center of the DAST reagent, forming a reactive intermediate that subsequently undergoes nucleophilic substitution by fluoride ions. This low-temperature protocol is critical for suppressing side reactions such as elimination or polymerization, ensuring that the difluoromethyl group is installed cleanly without affecting the adjacent bromine atom or the pyridine nitrogen. Following fluorination, the oxidation of the methyl group to an N-oxide using hydrogen peroxide in trifluoroacetic acid creates a highly activated species that is primed for the subsequent Boekelheide-type rearrangement. This rearrangement is driven by the thermal energy provided during the acetylation step, causing the migration of the acetoxy group to the adjacent carbon and establishing the correct substitution pattern required for the final target molecule.
Controlling impurity profiles in fluorinated pyridine synthesis is notoriously difficult due to the potential for defluorination or over-oxidation, yet this process demonstrates remarkable selectivity through careful modulation of reaction stoichiometry and thermal profiles. The use of a slight excess of DAST ensures complete consumption of the aldehyde starting material, preventing the carryover of unreacted precursors that could complicate downstream purification. Similarly, the controlled addition of hydrogen peroxide prevents the formation of over-oxidized byproducts, while the specific choice of alkaline hydrolysis conditions in the final step ensures gentle cleavage of the acetate ester without inducing decomposition of the sensitive difluoromethyl group. This meticulous attention to reaction parameters results in a final product with a purity profile that meets the rigorous standards expected by R&D directors evaluating new candidates for drug development. The ability to consistently produce high-purity intermediates reduces the burden on analytical teams and accelerates the overall timeline for process validation and regulatory filing.
How to Synthesize (3-Bromo-6-Difluoromethyl) Pyridin-2-Yl Methanol Efficiently
Implementing this synthesis on an industrial scale requires a thorough understanding of the thermodynamic and kinetic parameters defined in the patent to ensure safe and efficient operations. The process begins with the dissolution of the aldehyde precursor in dichloromethane, followed by the careful addition of DAST under an inert nitrogen atmosphere to maintain anhydrous conditions essential for fluorination success. Subsequent steps involve precise temperature ramps and phase separations that must be monitored closely to optimize yield and minimize waste generation. While the patent provides specific laboratory-scale examples, translating these to commercial reactors involves engineering considerations regarding heat transfer and mixing efficiency, particularly during the exothermic oxidation and acetylation phases. Detailed standardized synthesis steps see the guide below.
- Fluorination of 3-bromo-2-methylpyridine-6-carbaldehyde using DAST at low temperature to form the difluoromethyl intermediate.
- Oxidation of the methyl group to an N-oxide using hydrogen peroxide in trifluoroacetic acid, followed by thermal rearrangement.
- Acetylation of the rearranged intermediate with acetic anhydride, followed by alkaline hydrolysis to yield the target alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers substantial strategic advantages that extend far beyond simple chemical transformation, directly impacting the bottom line through enhanced operational efficiency. The reliance on widely available commodity reagents such as hydrogen peroxide, acetic anhydride, and common organic solvents mitigates the risk of supply disruptions often associated with specialized or proprietary catalysts. This accessibility ensures a stable supply chain for raw materials, allowing manufacturers to negotiate better pricing and secure long-term contracts with multiple vendors to hedge against market volatility. Furthermore, the high yields achieved in each step of the sequence mean that less raw material is wasted, leading to a significant reduction in the overall mass intensity of the process and a corresponding decrease in the cost of goods sold. By streamlining the synthesis into fewer, high-yielding steps, companies can reduce the number of unit operations required, thereby lowering energy consumption and labor costs associated with production.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes the need for costly scavenging resins and extensive metal testing, which traditionally adds significant expense to the manufacturing budget. Additionally, the high atom economy of the rearrangement step ensures that the majority of the starting material mass is incorporated into the final product, drastically reducing the volume of chemical waste that requires disposal. This efficiency translates directly into lower production costs per kilogram, providing a competitive edge in pricing negotiations with downstream pharmaceutical clients who are increasingly sensitive to the cost structures of their supply chains.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a reasonable range of temperatures and stoichiometries as shown in the patent examples, makes the process less susceptible to minor variations in raw material quality or equipment performance. This inherent stability reduces the likelihood of batch failures and reprocessing, ensuring a consistent flow of material to meet production schedules. Moreover, the use of standard chemical equipment and non-hazardous reagents simplifies logistics and storage requirements, allowing for greater flexibility in choosing manufacturing sites and reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process design inherently supports commercial scale-up of complex fluorinated intermediates by avoiding extreme pressures or cryogenic conditions beyond the initial fluorination step, which can be managed with standard industrial chilling systems. The aqueous workups and use of recyclable solvents align with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations regarding volatile organic compound emissions and wastewater treatment. This environmental compatibility not only reduces regulatory risk but also enhances the corporate sustainability profile, a factor that is becoming increasingly important for multinational corporations when selecting long-term manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis, providing clarity for stakeholders evaluating its potential for integration into existing production portfolios. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for assessing the feasibility of adopting this technology for specific project requirements and for anticipating any potential challenges during technology transfer.
Q: What are the critical safety considerations for the DAST fluorination step?
A: The use of Diethylaminosulfur Trifluoride (DAST) requires strict temperature control, typically starting at -78°C, to prevent exothermic runaway reactions and ensure selective fluorination without decomposing the sensitive pyridine ring structure.
Q: How does the N-oxide rearrangement improve regioselectivity?
A: The oxidation to the N-oxide activates the adjacent methyl group, allowing for a highly specific rearrangement upon heating with acetic anhydride. This avoids the formation of complex isomeric mixtures often seen in direct functionalization of the pyridine ring.
Q: Is this process suitable for multi-ton commercial production?
A: Yes, the pathway utilizes common industrial reagents like hydrogen peroxide and acetic anhydride. The high yields reported in the patent examples suggest that with proper engineering controls for the exothermic steps, the process is robust enough for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3-Bromo-6-Difluoromethyl) Pyridin-2-Yl Methanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced fluorinated intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality that spans from gram-scale optimization to multi-ton production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in a practical manufacturing setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of [(3-bromo-6-difluoromethyl) pyridin-2-yl] methanol meets the exacting standards required for pharmaceutical applications. Our capability to manage the specific safety protocols associated with DAST chemistry and oxidative rearrangements ensures that your supply remains uninterrupted and compliant with all international safety regulations.
We invite you to collaborate with us to leverage this innovative synthetic route for your next project, unlocking new possibilities for cost-effective and high-quality intermediate production. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
