Advanced Aqueous Synthesis of 8-Chlorotheophylline for Commercial API Production
Advanced Aqueous Synthesis of 8-Chlorotheophylline for Commercial API Production
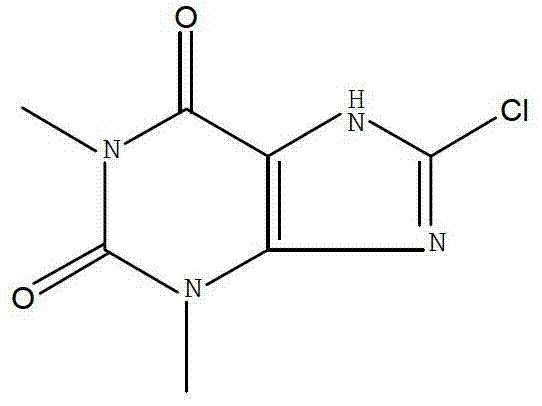
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally compliant pathways for critical active pharmaceutical ingredient (API) intermediates. A pivotal advancement in this domain is detailed in patent CN103360394A, which discloses a novel preparation method for 8-Chlorotheophylline (CAS: 85-18-7). This compound serves as a vital compatibility agent in the synthesis of Dimenhydrinate, a widely used antiemetic medication. The patented technology represents a significant departure from legacy manufacturing protocols by substituting hazardous chlorine gas and toxic organic solvents with N-chlorosuccinimide (NCS) in an aqueous medium. This strategic shift not only enhances the safety profile of the operation but also delivers superior product quality, with reported yields reaching 88-90% and HPLC purity exceeding 99%. For global supply chain leaders, this innovation underscores the potential for substantial process intensification and risk mitigation in the production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
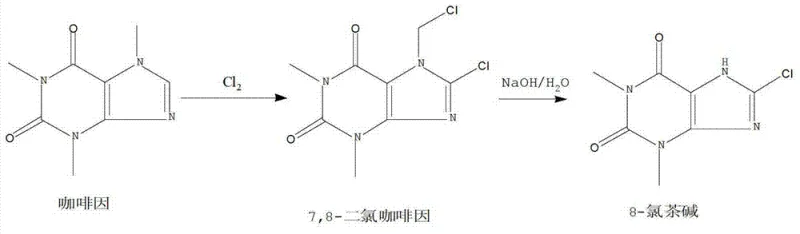
Historically, the industrial synthesis of 8-Chlorotheophylline has relied on two primary methodologies, both of which present severe operational and environmental challenges that hinder modern commercial scalability. The first conventional route, often utilizing caffeine as a starting material, involves chlorination with chlorine gas in the presence of nitrobenzene and iodine catalysts at elevated temperatures ranging from 80°C to 95°C. This process is notoriously difficult to control, requiring prolonged reaction times and generating significant quantities of hazardous wastewater and toxic organic waste. Furthermore, the reliance on caffeine as a feedstock introduces supply chain volatility, as it is a controlled substance in many jurisdictions. The second traditional method employs theophylline directly but still necessitates the use of chlorine gas dissolved in highly toxic halogenated solvents such as 1,2-dichloroethane or tetrachloroethane. These solvents are subject to strict regulatory limits regarding residual concentrations in final drug products, often requiring complex purification steps to meet the stringent threshold of less than 5ppm. Additionally, the use of gaseous chlorine poses acute safety risks, including equipment corrosion and the potential formation of uncontrolled free chlorine byproducts, which can compromise product integrity and operator safety.
The Novel Approach
In stark contrast to these archaic techniques, the methodology outlined in CN103360394A introduces a paradigm shift by leveraging N-chlorosuccinimide (NCS) as a solid, manageable chlorinating agent within a purely aqueous system. This approach effectively eliminates the need for volatile organic compounds (VOCs) and hazardous gaseous reagents, thereby simplifying the engineering controls required for the reactor setup. The reaction proceeds under mild thermal conditions, typically between 50°C and 80°C, which significantly reduces energy consumption compared to the high-temperature requirements of the caffeine-based route. By utilizing water as the solvent, the process inherently avoids the contamination issues associated with residual halogenated solvents, ensuring that the final API intermediate meets rigorous pharmacopeial standards without extensive downstream processing. The transition from a gas-liquid heterogeneous reaction to a more controlled liquid-phase substitution allows for precise stoichiometry management, minimizing the formation of poly-chlorinated impurities and maximizing the atom economy of the transformation.
Mechanistic Insights into NCS-Mediated Electrophilic Substitution
The core chemical innovation of this process lies in the selective electrophilic substitution at the C8 position of the theophylline ring system using N-chlorosuccinimide. Theophylline, chemically known as 1,3-dimethylxanthine, possesses a purine skeleton where the electron density is modulated by the electronegative nitrogen atoms and the carbonyl groups. In traditional acidic or radical conditions, controlling regioselectivity can be challenging; however, the use of NCS in a neutral to slightly acidic aqueous environment (pH 6-7) facilitates a specific attack on the C8 hydrogen. The mechanism involves the generation of a positive chlorine species from NCS, which acts as a soft electrophile. The presence of methyl groups at the N1 and N3 positions increases the electron density of the six-membered pyrimidine ring relative to the five-membered imidazole ring, yet the specific electronic environment at C8 renders it susceptible to this controlled halogenation. Crucially, the byproduct of this reaction is succinimide, which is highly soluble in water. This physical property difference allows for the facile separation of the desired 8-Chlorotheophylline, which precipitates upon cooling or pH adjustment, from the reaction matrix without the need for complex extraction protocols involving organic solvents.
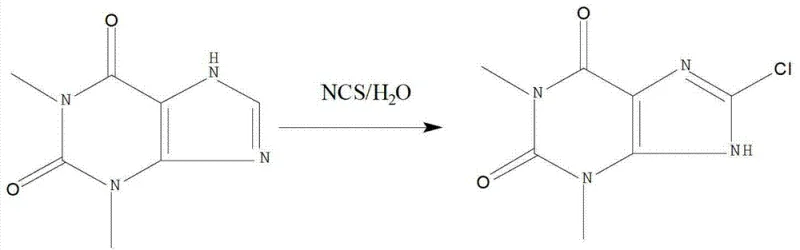
Furthermore, the mechanistic pathway ensures exceptional impurity control, a critical parameter for R&D directors overseeing API registration. In conventional chlorine gas methods, the high reactivity of molecular chlorine often leads to over-chlorination or the formation of oxidative degradation products that are structurally similar to the target molecule and difficult to remove. The NCS-mediated route operates under kinetic control where the reactivity of the chlorinating agent is moderated by the succinimide leaving group. This moderation prevents the runaway reactions often seen with elemental chlorine, thereby suppressing the formation of di-chlorinated species or ring-opened byproducts. The result is a crude product profile that is significantly cleaner, facilitating the subsequent recrystallization steps. The ability to monitor the reaction progress simply via Thin Layer Chromatography (TLC) until the disappearance of the theophylline spot indicates a robust and transparent process window, allowing manufacturers to halt the reaction precisely at maximum conversion to prevent secondary degradation.
How to Synthesize 8-Chlorotheophylline Efficiently
The operational simplicity of this aqueous synthesis makes it highly attractive for rapid technology transfer and scale-up. The process begins with the dissolution of anhydrous theophylline in water, followed by the controlled addition of the chlorinating agent. The reaction conditions are forgiving yet precise, allowing for a broad operating temperature range while maintaining high fidelity in product quality. Detailed standard operating procedures for this synthesis focus on the stoichiometric balance between the substrate and the reagent, as well as the critical pH control during the workup phase to ensure optimal precipitation of the final crystalline solid. For a comprehensive, step-by-step technical guide on executing this synthesis in a pilot or production plant, please refer to the standardized protocol below.
- Dissolve anhydrous Theophylline in water and heat to 50-80°C, then slowly drip N-chlorosuccinimide (NCS) while maintaining pH 6-7.
- Monitor the reaction progress via TLC until the raw material spot disappears, ensuring complete conversion to the chlorinated intermediate.
- Isolate the crude product, dissolve in 5% NaOH solution, filter, and reprecipitate by adjusting pH to 3-3.5 with dilute hydrochloric acid to obtain pure white solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NCS-based aqueous technology translates into tangible strategic advantages beyond mere chemical elegance. The elimination of hazardous reagents and solvents fundamentally reshapes the cost structure and risk profile of the manufacturing supply chain. By removing the dependency on chlorine gas infrastructure, facilities can avoid the capital expenditure associated with specialized corrosion-resistant reactors and scrubbing systems, leading to significant cost reduction in pharmaceutical intermediates manufacturing. Moreover, the switch to water as a solvent removes the logistical burden and cost of procuring, storing, and disposing of large volumes of regulated organic solvents like nitrobenzene and tetrachloroethane. This simplification of the bill of materials enhances supply chain resilience, as the key reagents—theophylline and NCS—are commercially available commodity chemicals with stable pricing and reliable global sourcing channels.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream processing workflow. In traditional methods, the removal of toxic solvents and the purification of the product from complex impurity profiles require multiple distillation and recrystallization steps, each incurring energy and material costs. In this novel aqueous route, the water-soluble nature of the succinimide byproduct allows it to be washed away easily, and the product can be isolated through simple filtration and acid-base precipitation. This reduction in unit operations directly lowers utility consumption and labor hours. Additionally, the higher yield reported (88-90% versus the historical 75%) means that less raw material is wasted per kilogram of output, effectively increasing the throughput of existing assets without the need for capacity expansion.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates like 8-Chlorotheophylline is paramount for preventing downtime in final API production lines. The conventional reliance on chlorine gas introduces a single point of failure; any disruption in gas supply or regulatory tightening on its transport can halt production immediately. By transitioning to a solid reagent system (NCS), the process becomes immune to gas logistics issues. Solid reagents are easier to store, have longer shelf lives, and can be stockpiled safely, providing a buffer against market fluctuations. This stability ensures reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to demand spikes from downstream drug formulators.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to scale a process without escalating waste treatment costs is a competitive differentiator. This aqueous method generates wastewater that is significantly less toxic than the halogenated organic waste streams of legacy processes. The absence of VOCs simplifies air emission compliance, and the reduced corrosivity of the reaction mixture extends the lifespan of stainless steel equipment. These factors collectively lower the total cost of ownership for the manufacturing facility. The process is inherently safer, reducing insurance premiums and liability risks, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates in regions with strict environmental enforcement.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its implementation. The following questions address common inquiries regarding the safety, efficiency, and regulatory implications of switching to this NCS-mediated pathway. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a factual basis for decision-making.
Q: Why is N-chlorosuccinimide (NCS) preferred over chlorine gas for this synthesis?
A: NCS offers superior safety and selectivity compared to hazardous chlorine gas. It operates under milder conditions in an aqueous phase, eliminating the need for toxic organic solvents like nitrobenzene and reducing equipment corrosion risks associated with free chlorine generation.
Q: What is the expected purity and yield of 8-Chlorotheophylline using this method?
A: According to patent CN103360394A, this optimized aqueous process achieves a yield of 88-90% and an HPLC purity exceeding 99%, significantly outperforming traditional methods which often struggle with impurity control and lower yields around 75%.
Q: How does this process address environmental compliance in pharmaceutical manufacturing?
A: By replacing volatile organic compounds (VOCs) like nitrobenzene and tetrachloroethane with water as the primary solvent, and avoiding the use of gaseous chlorine, the process drastically reduces hazardous waste generation and aligns with modern green chemistry principles for sustainable API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Chlorotheophylline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a regulatory necessity but a strategic imperative for maintaining competitiveness in the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the technology described in CN103360394A and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this aqueous NCS method are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 8-Chlorotheophylline meets the exacting standards required for API synthesis.
We invite forward-thinking partners to collaborate with us to leverage this advanced technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of adopting this route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, high-quality supply of this critical intermediate while optimizing your overall production costs.
