Scalable Synthesis of Aggregation-Induced Emission Pyrene Derivatives for Advanced Optoelectronics
Introduction to Next-Generation AIE Luminophores
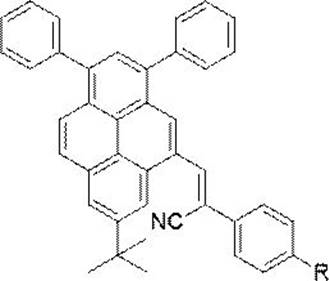
The rapid evolution of the organic optoelectronics sector demands materials that maintain high quantum efficiency even in aggregated or solid states, a challenge traditionally posed by the Aggregation-Caused Quenching (ACQ) effect inherent in many planar aromatic systems. Patent CN113402422B addresses this critical bottleneck by disclosing a robust synthesis method for a novel class of α-cyanoethylene-based aggregation-induced emission (AIE) luminophores derived from a pyrene core. These compounds are engineered to restrict intramolecular rotation, thereby converting non-radiative energy decay into efficient fluorescence when aggregated. This technological breakthrough is particularly significant for the development of high-performance organic light-emitting diodes (OLEDs), mechanochemical sensors, and advanced biological imaging probes where solid-state brightness is paramount.
From a manufacturing perspective, the patent outlines a pathway that balances molecular complexity with process simplicity, utilizing readily available commercial reagents to construct the functionalized pyrene scaffold. The strategic incorporation of the electron-withdrawing cyano-vinyl group not only tunes the electronic properties for red-shifted emission but also facilitates a straightforward Knoevenagel condensation reaction. This approach circumvents the need for harsh conditions or exotic catalysts, positioning this chemistry as a viable candidate for the commercial scale-up of complex OLED material intermediates. For industry stakeholders, this represents a shift towards more sustainable and economically feasible production of high-value electronic chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the modification of pyrene cores to prevent fluorescence quenching has relied heavily on bulky substituent introduction via palladium-catalyzed cross-coupling reactions, such as Suzuki or Heck couplings. While effective, these conventional methodologies introduce significant supply chain vulnerabilities and cost burdens due to the reliance on precious metal catalysts and the subsequent necessity for rigorous purification to meet electronic-grade purity standards. Furthermore, traditional synthetic routes often involve multi-step sequences with low atom economy, generating substantial chemical waste and requiring energy-intensive separation techniques like flash column chromatography. These factors collectively hinder the cost reduction in electronic chemical manufacturing, making large-scale deployment of high-performance luminescent materials economically challenging for mass-market display technologies.
The Novel Approach
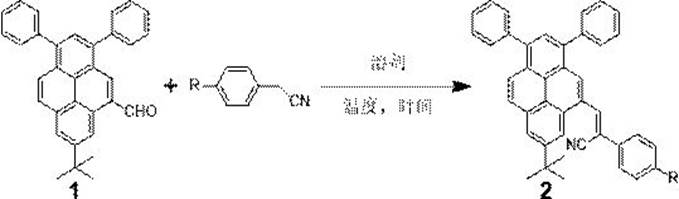
In stark contrast, the methodology presented in CN113402422B leverages a direct Knoevenagel condensation between a formyl-functionalized pyrene intermediate and substituted phenylacetonitriles. This reaction proceeds under mild alkaline conditions using potassium tert-butoxide in methanol, eliminating the need for transition metals entirely. The simplicity of this one-step carbon-carbon bond formation drastically reduces the operational complexity and safety hazards associated with handling air-sensitive catalysts. By streamlining the synthesis into a single convergent step followed by a simple precipitation workup, the process enhances overall throughput and minimizes solvent consumption. This novel approach directly supports the goal of being a reliable OLED material intermediate supplier by ensuring a consistent, high-yield production capability that is less susceptible to raw material volatility.
Mechanistic Insights into Knoevenagel Condensation
The core chemical transformation driving this synthesis is the base-catalyzed Knoevenagel condensation, a fundamental carbon-carbon bond-forming reaction that links the active methylene group of the phenylacetonitrile to the carbonyl carbon of the pyrene aldehyde. Mechanistically, the potassium tert-butoxide deprotonates the alpha-carbon of the nitrile, generating a resonance-stabilized carbanion nucleophile. This nucleophile attacks the electrophilic aldehyde carbon, forming a beta-hydroxy nitrile intermediate which subsequently undergoes dehydration to yield the conjugated α-cyanoethylene double bond. This extended pi-conjugation is critical for the optoelectronic properties, as it lowers the energy gap between the HOMO and LUMO levels, facilitating charge transport and tuning the emission wavelength. The reaction is driven to completion by the thermodynamic stability of the conjugated product and the removal of water, although in this specific alcoholic solvent system, the equilibrium favors the product due to the stability of the extended conjugated system.
Impurity control in this process is elegantly managed through the physical properties of the reactants and products rather than complex chromatographic separations. The patent specifies that the target α-cyanoethylene pyrene derivatives exhibit low solubility in methanol, the reaction solvent, especially upon cooling or concentration. This differential solubility allows for a purification strategy where the crude product precipitates out of the solution while unreacted starting materials and polar byproducts remain dissolved in the mother liquor or are washed away during the filtration step. This mechanism ensures that the final high-purity AIE luminophore meets stringent quality specifications without the need for silica gel purification, which is often a bottleneck in scaling up fine chemical synthesis. The result is a product with excellent thermal stability and defined crystal structures, as confirmed by X-ray diffraction data in the patent examples.
How to Synthesize Alpha-Cyanoethylene Pyrene Derivatives Efficiently
To replicate the high yields and purity described in the patent, precise control over reaction stoichiometry and thermal conditions is essential. The optimized protocol involves dissolving the phenylacetonitrile derivative and potassium tert-butoxide in methanol, followed by the addition of the pyrene aldehyde dissolved in a co-solvent like tetrahydrofuran to ensure homogeneity. The mixture is then heated to reflux for approximately 12 hours to drive the condensation to completion. Following the reaction, the workup involves liquid-liquid extraction with dichloromethane to separate the organic product from inorganic salts, followed by drying and solvent removal. The detailed standardized synthesis steps, including specific molar ratios and washing procedures, are outlined below to ensure reproducibility for process chemists aiming to implement this route.
- Perform Knoevenagel condensation between 7-tert-butyl-1,3-diphenyl-5-formylpyrene and 4-R-phenylacetonitrile using potassium tert-butoxide in methanol under reflux for 12 hours.
- Extract the cooled reaction mixture with dichloromethane, wash the organic phase with saturated brine, and dry over anhydrous magnesium sulfate.
- Distill off the solvent under reduced pressure, filter the resulting solid, and wash with methanol to obtain the pure target luminophore.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits rooted in process intensification and raw material accessibility. The elimination of precious metal catalysts not only removes a significant cost driver but also mitigates the risk of supply disruptions associated with rare earth or platinum-group metals. Furthermore, the use of commodity solvents like methanol and dichloromethane, combined with a workup that avoids energy-intensive distillation columns or chromatography skids, translates to a drastically simplified manufacturing footprint. This simplification allows for faster batch turnover times and reduced capital expenditure on specialized equipment, directly contributing to cost reduction in electronic chemical manufacturing while maintaining high product quality standards required for downstream device fabrication.
- Cost Reduction in Manufacturing: The process utilizes inexpensive, commercially available starting materials such as phenylacetonitrile derivatives and potassium tert-butoxide, avoiding the high costs associated with specialized organometallic reagents. By removing the requirement for transition metal catalysts, the downstream processing costs related to metal scavenging and residual metal testing are completely eliminated. This streamlined chemistry significantly lowers the cost of goods sold (COGS), making the final luminescent material more competitive in price-sensitive markets like consumer electronics and large-area lighting panels.
- Enhanced Supply Chain Reliability: All reagents specified in the patent are bulk commodity chemicals with established global supply chains, reducing the risk of single-source bottlenecks. The robustness of the Knoevenagel condensation under reflux conditions implies a wide operating window, meaning the process is less sensitive to minor fluctuations in temperature or mixing rates compared to sensitive catalytic cycles. This operational resilience ensures consistent production schedules and reducing lead time for high-purity luminescent materials, allowing manufacturers to respond more agilely to market demand spikes without compromising on delivery commitments.
- Scalability and Environmental Compliance: The synthesis generates minimal hazardous waste, primarily consisting of aqueous salt solutions and recoverable organic solvents, which aligns well with modern green chemistry principles and environmental regulations. The purification step relies on crystallization and filtration rather than large volumes of silica or eluents, significantly reducing the solid waste burden. This environmentally friendly profile facilitates easier regulatory approval for new manufacturing sites and supports the long-term sustainability goals of multinational corporations seeking to minimize their carbon footprint in the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these alpha-cyanoethylene pyrene derivatives. The answers are derived directly from the experimental data and technical specifications provided in patent CN113402422B, ensuring accuracy for R&D and procurement teams evaluating this technology for integration into their existing material portfolios.
Q: What is the primary advantage of this alpha-cyanoethylene pyrene derivative over traditional pyrene materials?
A: Unlike traditional pyrene derivatives that suffer from Aggregation-Caused Quenching (ACQ) in solid states, this alpha-cyanoethylene derivative exhibits Aggregation-Induced Emission (AIE). The restriction of intramolecular rotation (RIR) mechanism prevents fluorescence quenching, making it highly efficient for solid-state optoelectronic applications like OLEDs and sensors.
Q: Does the synthesis require expensive transition metal catalysts?
A: No, the synthesis described in patent CN113402422B utilizes a metal-free Knoevenagel condensation strategy. It relies on potassium tert-butoxide as a base catalyst, which significantly reduces raw material costs and eliminates the need for complex heavy metal removal processes typically required in cross-coupling reactions.
Q: How is the purity of the final product ensured without column chromatography?
A: The process achieves high purity through a simplified workup procedure. After extraction and drying, the crude product is purified by distilling off the solvent and washing the precipitated solid with methanol. Since the product has low solubility in methanol while impurities remain soluble, this recrystallization-like step effectively yields high-purity material suitable for electronic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Cyanoethylene Pyrene Derivative Supplier
As the demand for high-efficiency luminescent materials continues to surge across the display and sensor industries, partnering with an experienced CDMO is crucial for translating laboratory patents into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to tonnage manufacturing is seamless and risk-mitigated. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis and thermal stability testing, guaranteeing that every batch of alpha-cyanoethylene pyrene derivative meets the exacting standards required for next-generation optoelectronic devices.
We invite potential partners to engage with our technical team to discuss how this specific Knoevenagel-based route can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how implementing this metal-free synthesis can optimize your overall material budget. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of chemical excellence and reliability.
