Scalable Synthesis of Aggregation-Induced Emission Pyrene Derivatives for Commercial OLED Applications
Scalable Synthesis of Aggregation-Induced Emission Pyrene Derivatives for Commercial OLED Applications
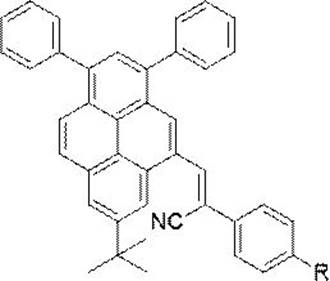
The rapid evolution of the organic optoelectronics sector demands materials that not only exhibit superior luminescent efficiency but also possess robust physical properties suitable for rigorous industrial processing. Patent CN113402422B introduces a significant advancement in this domain by disclosing a novel synthesis method for α-cyanoethylene-based aggregation-induced luminophores. These specialized compounds are engineered to overcome the notorious aggregation-caused quenching (ACQ) effect that traditionally plagues pyrene-based materials in solid-state applications. By strategically modifying the pyrene core with an α-cyanoethylene moiety, the invention successfully restricts intramolecular rotation, thereby unlocking high-efficiency emission in aggregated states. This technological breakthrough positions these derivatives as prime candidates for next-generation luminescent materials, mechanochemical sensors, and advanced biological imaging agents, offering a reliable OLED material supplier pathway for manufacturers seeking high-performance emitters.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing pyrene-based luminescent materials often suffer from significant drawbacks that hinder their widespread commercial adoption and cost-effectiveness in electronic chemical manufacturing. Conventional routes frequently rely on multi-step synthetic sequences involving expensive transition metal catalysts, such as palladium or platinum complexes, which not only inflate the raw material costs but also introduce complex purification challenges to remove trace metal residues that can degrade device performance. Furthermore, many existing pyrene derivatives exhibit strong π-π interactions in the solid state, leading to severe fluorescence quenching that renders them ineffective for solid-state lighting or display applications without elaborate structural modifications. The reliance on harsh reaction conditions, sensitive reagents requiring inert atmospheres, and low overall yields further exacerbates the supply chain vulnerabilities, making the consistent production of high-purity OLED material a logistical and financial burden for procurement teams managing tight budgets.
The Novel Approach
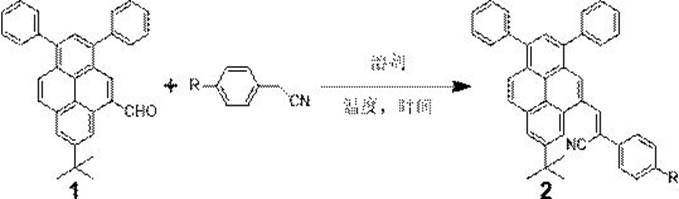
In stark contrast to these legacy methods, the technology described in CN113402422B utilizes a streamlined Knoevenagel condensation strategy that dramatically simplifies the production workflow while enhancing product quality. This novel approach leverages the reaction between 7-tert-butyl-1,3-diphenyl-5-formylpyrene and readily available phenylacetonitrile derivatives under mild alkaline conditions, eliminating the need for precious metal catalysts entirely. The reaction proceeds efficiently in common organic solvents like methanol under reflux, facilitating a direct and high-yielding transformation that minimizes waste generation and energy consumption. By adopting this method, manufacturers can achieve a substantial reduction in production complexity, as the workup involves standard extraction and simple solvent washing rather than chromatographic purification, thereby significantly lowering the barrier to entry for commercial scale-up of complex pyrene derivatives.
Mechanistic Insights into Knoevenagel-Catalyzed Cyclization
The core of this synthetic innovation lies in the base-catalyzed Knoevenagel condensation mechanism, which facilitates the formation of the carbon-carbon double bond linking the pyrene aldehyde and the nitrile group. In this process, a strong base such as potassium tert-butoxide deprotonates the active methylene group of the phenylacetonitrile derivative, generating a nucleophilic carbanion that attacks the electrophilic carbonyl carbon of the pyrene aldehyde. This addition is followed by a dehydration step, driven by the thermodynamic stability of the conjugated α-cyanoethylene system, which extends the π-conjugation across the molecule. This extended conjugation is critical for tuning the electronic properties of the final luminophore, ensuring that the HOMO-LUMO gap is optimized for the desired emission wavelength while the steric bulk of the tert-butyl and phenyl groups prevents detrimental face-to-face stacking.
From an impurity control perspective, the mechanism inherently favors high selectivity due to the distinct reactivity differences between the aldehyde functionality and the aromatic protons on the pyrene ring. The use of a specific molar ratio, optimized at 1:2.5 between the aldehyde and the nitrile component, ensures that the aldehyde is fully consumed, minimizing the presence of unreacted starting material which can act as a quencher in the final device. Furthermore, the choice of methanol as a washing solvent during purification exploits the differential solubility of the product versus potential side products; the target α-cyanoethylene derivative has low solubility in cold methanol, allowing impurities to remain in the solution while the high-purity AIE luminophore precipitates as a solid, thus achieving exceptional purity levels without the need for resource-intensive column chromatography.
How to Synthesize Alpha-Cyanoethylene Pyrene Derivatives Efficiently
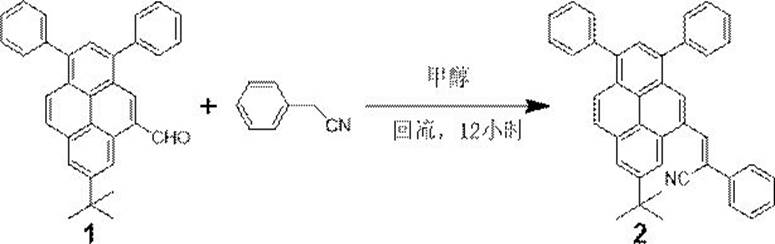
To implement this robust synthetic route in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric and thermal parameters to maximize yield and reproducibility. The process begins with the activation of the phenylacetonitrile nucleophile using potassium tert-butoxide in methanol, followed by the introduction of the pyrene aldehyde dissolved in a co-solvent like tetrahydrofuran to ensure homogeneity. Maintaining the reaction at reflux temperature for a duration of 12 hours is critical to drive the equilibrium towards completion, as shorter reaction times or lower temperatures have been shown to result in incomplete conversion and reduced isolated yields. The detailed standardized synthesis steps, including specific quantities and workup procedures, are outlined below to ensure consistent production of the target material.
- Combine 7-tert-butyl-1,3-diphenyl-5-formylpyrene and 4-R phenylacetonitrile in a molar ratio of 1: 2.5 with potassium tert-butoxide in methanol solvent.
- Heat the reaction mixture under reflux conditions for approximately 12 hours to ensure complete conversion via Knoevenagel condensation.
- Extract the cooled product with dichloromethane, wash with saturated brine, dry over magnesium sulfate, and purify by suction filtration after methanol washing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology translates into tangible strategic benefits that extend far beyond simple chemical transformation. The reliance on commodity chemicals such as methanol, potassium tert-butoxide, and substituted phenylacetonitriles means that the raw material supply chain is highly resilient and immune to the volatility often associated with specialized organometallic reagents. This stability allows for long-term contracting and predictable budgeting, effectively mitigating the risk of production stoppages due to material shortages. Moreover, the simplicity of the downstream processing—requiring only extraction, drying, and filtration—drastically reduces the demand for specialized equipment and skilled labor, enabling faster throughput and shorter lead times for high-purity electronic chemicals.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts represents a direct and significant saving in bill of materials costs, as there is no longer a need to procure costly palladium or rhodium complexes. Additionally, the removal of metal catalysts obviates the need for expensive metal scavenging resins or complex purification steps designed to meet strict residual metal specifications, which further drives down the operational expenditure per kilogram of produced material. The high atom economy of the condensation reaction also ensures that a larger proportion of the input mass is converted into valuable product rather than waste, contributing to a leaner and more cost-efficient manufacturing process overall.
- Enhanced Supply Chain Reliability: Since all reagents involved in this synthesis are commercially available off-the-shelf chemicals with established global supply networks, the risk of supply disruption is minimized compared to routes relying on custom-synthesized intermediates. The robustness of the reaction conditions, which tolerate standard laboratory glassware and heating mantles rather than requiring high-pressure reactors or cryogenic cooling, means that production can be easily transferred between different manufacturing sites or contract manufacturing organizations without extensive requalification. This flexibility ensures a continuous and reliable flow of materials to downstream device fabricators, safeguarding against market fluctuations.
- Scalability and Environmental Compliance: The use of methanol and dichloromethane, while requiring standard safety protocols, allows for well-established solvent recovery and recycling systems to be implemented, reducing the environmental footprint and disposal costs associated with hazardous waste. The absence of heavy metals in the process stream simplifies wastewater treatment and regulatory compliance, making it easier to obtain necessary environmental permits for expansion. Furthermore, the high thermal stability of the final product means it can be stored and transported under ambient conditions without degradation, reducing logistics costs and expanding the viable geographic market reach for the material.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the operational parameters and performance characteristics of the resulting luminophores. Understanding these details is crucial for R&D teams evaluating the material for specific device architectures and for procurement specialists assessing the feasibility of long-term supply agreements.
Q: What are the primary advantages of this Knoevenagel condensation route for AIE luminophores?
A: The primary advantages include the use of commercially available and inexpensive raw materials, a simple one-step reaction process that avoids complex catalytic systems, and high thermal stability of the resulting products which do not require special storage conditions.
Q: How does this synthesis method address the issue of fluorescence quenching in solid states?
A: By introducing the α-cyanoethylene group onto the pyrene core, the molecular structure restricts intramolecular rotation, thereby overcoming the aggregation-caused quenching (ACQ) effect typical of traditional pyrene derivatives and enabling strong emission in aggregated or solid states.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route is highly suitable for industrial scale-up due to its reliance on standard reflux conditions in methanol, the elimination of expensive transition metal catalysts, and a straightforward purification process involving simple solvent washing and filtration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Cyanoethylene Pyrene Derivatives Supplier
As the demand for high-performance organic light-emitting materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies backed by rigorous quality assurance. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and optical properties of every batch, guaranteeing that the α-cyanoethylene pyrene derivatives you receive meet the exacting standards required for advanced optoelectronic applications.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalyst-free methodology. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your product performance and your bottom line in the competitive landscape of electronic chemical manufacturing.
