Advanced Synthesis of Propranolol Hydrochloride for Commercial Scale-up and High Purity
Introduction to Advanced Propranolol Hydrochloride Synthesis
The pharmaceutical landscape continuously demands more efficient and scalable processes for critical cardiovascular medications, and the synthesis of Propranolol Hydrochloride stands as a prime example of this evolution. As detailed in patent CN111253267B, a novel methodology has been developed that addresses long-standing inefficiencies in producing this essential beta-blocker. This compound, chemically known as 1-isopropylamino-3-(1-naphthyloxy)-2-propanol hydrochloride, remains a cornerstone therapy for managing hypertension, angina pectoris, and arrhythmias. The structural integrity and high purity of the final active pharmaceutical ingredient are paramount for patient safety and efficacy. 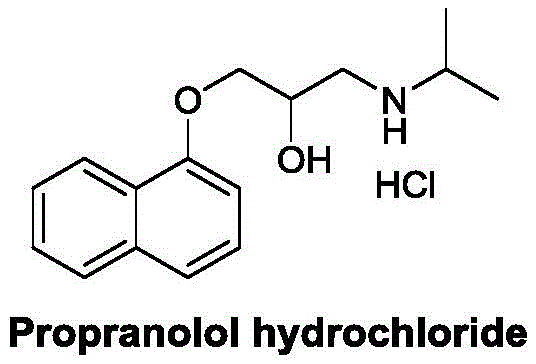 . The disclosed technology leverages a sophisticated sequence of etherification, ring-opening, and a unique salt-forming reaction to achieve a refined purity exceeding 99.8 percent. For R&D directors and procurement specialists, understanding the nuances of this patent provides a strategic advantage in securing reliable supply chains for high-quality API intermediates.
. The disclosed technology leverages a sophisticated sequence of etherification, ring-opening, and a unique salt-forming reaction to achieve a refined purity exceeding 99.8 percent. For R&D directors and procurement specialists, understanding the nuances of this patent provides a strategic advantage in securing reliable supply chains for high-quality API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Propranolol Hydrochloride has been plagued by significant yield losses and operational complexities inherent to traditional synthetic routes. One common method involves the etherification of 1-naphthol with epichlorohydrin followed by ring-opening with isopropylamine, but the final salt formation step using concentrated hydrochloric acid presents a major bottleneck. Due to the high water solubility of Propranolol Hydrochloride, substantial amounts of the product remain dissolved in the mother liquor during crystallization, leading to relatively low overall yields. Furthermore, alternative routes utilizing 1,3-dibromoacetone as a starting material introduce prohibitive raw material costs and generate difficult-to-separate disubstituted byproducts. These legacy processes often require excessive amounts of isopropylamine and harsh conditions that complicate waste treatment and increase the environmental footprint of manufacturing facilities.
The Novel Approach
The innovative process outlined in the patent data fundamentally re-engineers the synthesis pathway to overcome these specific thermodynamic and kinetic barriers. By optimizing the etherification step with effective phase transfer catalysts and, most critically, replacing aqueous hydrochloric acid with thionyl chloride in an alcohol solvent for salt formation, the process ensures superior product precipitation. This modification drastically reduces dissolution losses and simplifies the isolation of the crude product. 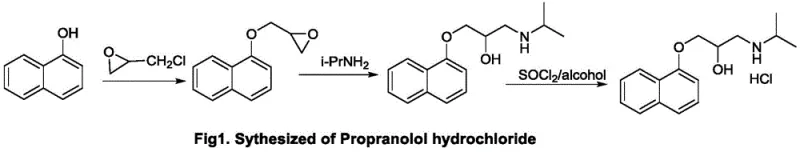 . The result is a streamlined workflow that not only enhances the molar yield to over 80 percent but also significantly improves the operational safety profile by avoiding the handling of large volumes of corrosive concentrated acids. This approach represents a robust solution for cost reduction in pharmaceutical manufacturing, aligning perfectly with modern green chemistry principles.
. The result is a streamlined workflow that not only enhances the molar yield to over 80 percent but also significantly improves the operational safety profile by avoiding the handling of large volumes of corrosive concentrated acids. This approach represents a robust solution for cost reduction in pharmaceutical manufacturing, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into Phase Transfer Catalysis and Salt Formation
The core of this synthetic breakthrough lies in the precise control of reaction kinetics during the initial etherification and the final salt formation stages. In the first step, 1-naphthol reacts with epichlorohydrin under alkaline conditions, facilitated by phase transfer catalysts such as benzyltriethylammonium chloride or polyethylene glycol. These catalysts effectively shuttle the phenoxide anion into the organic phase, accelerating the nucleophilic attack on the epoxide ring to form the key intermediate, 3-(1-naphthoxy)-1,2-epoxypropane. Maintaining the temperature between 40-80 degrees Celsius ensures optimal conversion while minimizing side reactions. Subsequently, the ring-opening reaction with isopropylamine is catalyzed by tertiary amines like N,N-diisopropylethylamine, which activates the epoxide for nucleophilic attack without promoting polymerization. This careful selection of bases and solvents creates a clean reaction profile that simplifies downstream purification efforts.
Perhaps the most mechanistically significant improvement is the utilization of thionyl chloride (SOCl2) in an alcohol solvent for the final salt formation. Unlike the traditional addition of HCl gas or aqueous acid, the reaction of SOCl2 with alcohol generates HCl in situ within an organic medium. This environment favors the immediate precipitation of the Propranolol Hydrochloride salt as the reaction proceeds, shifting the equilibrium towards product formation and preventing the redissolution issues seen in aqueous systems. The exothermic nature of this reaction is managed by controlling the temperature between 0-50 degrees Celsius, specifically preferring a range of 5-15 degrees Celsius to ensure crystal quality. This mechanism not only boosts yield but also produces a crystalline solid that is easier to filter and dry, directly impacting the efficiency of the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Propranolol Hydrochloride Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding temperature, stoichiometry, and solvent selection to maximize the benefits of the novel salt formation technique. The process begins with the preparation of the epoxy intermediate, followed by the amination step, and concludes with the critical salting-out procedure. Operators must ensure that the phase transfer catalyst is fully dissolved before the addition of the alkaline solution to prevent localized overheating. The subsequent ring-opening step benefits from a slight excess of isopropylamine to drive the reaction to completion, while the final salt formation demands precise temperature control to manage the evolution of gases and ensure uniform particle size. Detailed standardized synthetic steps see the guide below.
- Conduct etherification of 1-naphthol and epichlorohydrin using a phase transfer catalyst under alkaline conditions to form 3-(1-naphthoxy)-1,2-epoxypropane.
- Perform a base-catalyzed ring-opening reaction with isopropylamine to generate the crude Propranolol base.
- Execute salt formation using thionyl chloride in an alcohol solvent followed by recrystallization to obtain pure Propranolol Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond simple yield improvements. The shift away from expensive and hazardous raw materials like 1,3-dibromoacetone to widely available commodities such as epichlorohydrin and thionyl chloride significantly de-risks the supply chain. This transition ensures that production is not held hostage by the volatility of niche reagent markets, thereby enhancing supply chain reliability and continuity. Furthermore, the simplified workup procedures reduce the burden on waste treatment facilities, as the process generates fewer aqueous waste streams compared to traditional hydrochloride salt formation methods. These factors collectively contribute to a more resilient and cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of high-cost starting materials and the substantial improvement in overall yield directly translate to a lower cost of goods sold. By avoiding the significant product losses associated with aqueous salt formation, manufacturers can extract more value from every kilogram of raw material input. Additionally, the reduced need for extensive solvent recovery and wastewater treatment lowers utility and disposal expenses. This economic efficiency allows for more competitive pricing strategies in the global API market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on bulk chemicals like 1-naphthol, epichlorohydrin, and isopropylamine ensures that raw material sourcing remains stable even during market fluctuations. These commodities are produced at massive scales globally, reducing the risk of shortages that often plague specialized fine chemical intermediates. The robustness of the process also means that production timelines are more predictable, as there are fewer purification bottlenecks that could delay batch release. This stability is crucial for maintaining consistent inventory levels and meeting the rigorous delivery schedules of downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of standard organic solvents make this process highly amenable to scaling from pilot plants to multi-ton commercial production. The absence of heavy metal catalysts or exotic reagents simplifies regulatory compliance and environmental permitting. Moreover, the high purity of the crude product reduces the load on the final recrystallization step, minimizing solvent consumption and energy usage. This alignment with green chemistry principles not only satisfies increasingly stringent environmental regulations but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific synthesis pathway. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their own production lines. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for industrial application. We encourage stakeholders to review these points to fully appreciate the operational advantages offered by this technology.
Q: What is the key advantage of using thionyl chloride for salt formation in this process?
A: Unlike traditional concentrated hydrochloric acid methods where the product has high water solubility leading to dissolution losses, using thionyl chloride in an alcohol solvent facilitates precipitation, significantly improving yield and isolation efficiency.
Q: How does this method improve upon conventional synthesis routes?
A: This method avoids the use of expensive raw materials like 1,3-dibromoacetone and eliminates the low-yield issues associated with traditional hydrochloride salt formation, resulting in a total yield exceeding 80 percent and purity over 99.8 percent.
Q: What are the optimal reaction conditions for the etherification step?
A: The etherification reaction is optimally conducted at temperatures between 40-80 degrees Celsius for 4 to 6 hours using phase transfer catalysts such as benzyltriethylammonium chloride or polyethylene glycol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Propranolol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of life-saving cardiovascular therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. We are committed to delivering Propranolol Hydrochloride that meets stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest international standards. Our facility is equipped to handle the specific requirements of this synthesis, including the safe management of thionyl chloride and the precise temperature controls needed for optimal crystallization.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through this advanced technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term production goals. Let us be your partner in delivering high-quality pharmaceutical solutions to the global market.
