Advanced One-Pot Synthesis of Anethole: A Green Route for Industrial Scale-Up
The global demand for high-purity anethole, a key component in flavor and fragrance formulations known for its sweet fennel aroma, has driven significant innovation in synthetic methodologies. Patent CN107973703B introduces a transformative approach to synthesizing aryl-containing substituted olefins, specifically targeting the efficient production of anethole through a novel one-pot reaction. This technology leverages aliphatic acetals as alkylating agents reacting directly with aromatic compounds under the catalysis of solid acids, such as H-Beta molecular sieves. Unlike traditional extraction methods limited by natural resource availability or multi-step syntheses generating substantial waste, this patented route operates under mild conditions ranging from 90 to 160°C and pressures between 0.1 to 1.0 MPa. For R&D directors and process engineers, this represents a pivotal shift towards greener chemistry, offering a streamlined pathway that integrates alkylation and dehydration into a single operational unit, thereby enhancing overall process efficiency and reducing the carbon footprint associated with fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of anethole has relied heavily on extraction from natural essential oils like anise or fennel, a process inherently constrained by agricultural variability and limited yield, failing to meet the escalating demands of the global flavor industry. Alternatively, synthetic routes such as those described in prior art often involve harsh Friedel-Crafts acylation followed by reduction and dehydration, typically requiring stoichiometric amounts of aluminum trichloride and sodium borohydride. These conventional methods suffer from severe drawbacks, including the generation of large volumes of acidic wastewater and heavy metal waste, necessitating complex and costly purification steps to meet stringent pharmaceutical and food-grade purity standards. Furthermore, thermal decomposition routes operating at temperatures exceeding 300°C often result in incomplete conversion and the formation of undesirable isomers and polymeric by-products, complicating downstream separation and significantly impacting the economic viability of the process.
The Novel Approach
In stark contrast, the methodology disclosed in CN107973703B offers a robust solution by employing aliphatic acetals as stable alkylating agents in conjunction with reusable solid acid catalysts. This innovative strategy eliminates the need for external organic solvents, as the aromatic substrate itself serves as the reaction medium, drastically simplifying the reaction matrix and reducing volatile organic compound (VOC) emissions. The use of shape-selective zeolites like H-Beta not only enhances regioselectivity towards the desired para-isomer but also facilitates easy catalyst recovery through simple filtration, enabling potential reuse and minimizing solid waste. By conducting the reaction in a one-pot system at moderate temperatures, the process avoids the energy-intensive conditions of thermal cracking while maintaining high selectivity, presenting a commercially attractive alternative for the cost reduction in flavor & fragrance intermediates manufacturing.
Mechanistic Insights into Solid Acid-Catalyzed Alkylation
The core of this synthetic breakthrough lies in the activation of the aliphatic acetal by the Brønsted acid sites present on the surface of the solid catalyst, such as the H-Beta molecular sieve. Upon interaction with the acidic protons, the acetal undergoes cleavage to generate a reactive carbocation intermediate or an oxocarbenium ion, which subsequently attacks the electron-rich aromatic ring of the anisole in an electrophilic aromatic substitution mechanism. This initial alkylation step is seamlessly followed by an elimination reaction within the same reaction vessel, where the intermediate alcohol species loses a molecule of water or alcohol to form the thermodynamically stable trans-alkene double bond characteristic of anethole. The porous structure of the zeolite catalyst plays a critical role in stabilizing transition states and suppressing side reactions, such as poly-alkylation or isomerization, ensuring a cleaner impurity profile that is crucial for high-value applications.
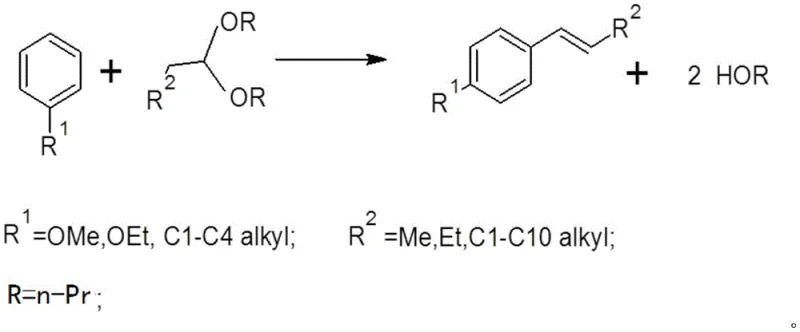
Furthermore, the mechanistic pathway allows for the recycling of the alcohol by-product (HOR), which can be recovered and reintroduced into the upstream synthesis of the acetal starting material, creating a closed-loop atom economy that aligns with modern sustainability goals. The versatility of the catalyst system, which can be modified with metal ions like Cerium or Nickel to tune acidity and pore size, provides R&D teams with the flexibility to optimize selectivity for specific substituted olefins beyond just anethole. Understanding this catalytic cycle is essential for scaling the process, as it highlights the importance of mass transfer limitations within the zeolite pores and the need for precise temperature control to maintain catalyst activity over extended periods in continuous flow systems.
How to Synthesize Anethole Efficiently
To implement this advanced synthesis route effectively, manufacturers must adhere to specific operational parameters that maximize conversion and selectivity while ensuring safety and reproducibility. The process begins with the precise charging of anisole and the selected aliphatic acetal into a pressure-rated reactor, followed by the addition of the activated solid acid catalyst under inert atmosphere conditions to prevent moisture deactivation. Detailed standard operating procedures regarding heating ramps, stirring speeds, and quenching protocols are critical to managing the exothermic nature of the alkylation and ensuring uniform heat distribution throughout the reaction mass. For a comprehensive guide on the specific molar ratios, catalyst loading percentages, and workup procedures validated by experimental data, please refer to the standardized synthesis protocol outlined below.
- Mix aromatic compound (anisole) and aliphatic acetal (e.g., propionaldehyde dimethanol acetal) in a reactor, utilizing the aromatic compound as both reactant and solvent.
- Add a solid acid catalyst such as H-Beta molecular sieve or metal-ion modified molecular sieves (e.g., Ce-Beta, Ni-Y) to the mixture.
- Heat the reaction mixture to 90-160°C under normal or low pressure (0.1-1.0 MPa) for several hours to complete the one-pot alkylation and dehydration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this solid acid-catalyzed route offers profound strategic advantages that extend beyond mere technical feasibility. The elimination of stoichiometric Lewis acids and the reduction of solvent usage directly translate to a simplified supply chain, reducing dependency on hazardous raw materials that are subject to volatile pricing and strict regulatory controls. Moreover, the compatibility of this reaction with fixed-bed and tubular reactors opens the door for continuous manufacturing, a paradigm shift that significantly enhances production throughput and consistency compared to traditional batch processing. This adaptability ensures a more reliable supply of high-purity anethole, mitigating risks associated with production bottlenecks and enabling manufacturers to respond agilely to market fluctuations in the flavor and fragrance sector.
- Cost Reduction in Manufacturing: The transition to a solvent-free system where the reactant acts as the solvent drastically cuts down on raw material costs and the energy expenditure associated with solvent recovery and distillation. Additionally, the use of heterogeneous catalysts that can be filtered and potentially regenerated reduces the recurring cost of catalyst consumption, while the simplified post-treatment workflow minimizes labor and utility costs related to waste neutralization and disposal. By avoiding the generation of heavy metal waste streams, facilities can also realize substantial savings in environmental compliance and effluent treatment operations, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Utilizing stable aliphatic acetals as starting materials provides a more robust supply chain compared to relying on natural essential oils, which are susceptible to crop failures and seasonal variations. The ability to source these petrochemical-derived acetals from multiple global suppliers ensures continuity of supply, while the modular nature of the reactor setup allows for decentralized production capabilities closer to key markets. This decentralization reduces logistics lead times and transportation costs, ensuring that customers receive their orders of high-purity aroma chemicals with greater speed and reliability, fostering stronger long-term partnerships.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process, such as atom economy and waste prevention, position manufacturers favorably against increasingly stringent environmental regulations worldwide. The absence of corrosive liquid acids and the generation of recyclable alcohol by-products simplify the permitting process for new production lines and reduce the liability associated with hazardous chemical storage. Scalability is further enhanced by the proven performance of molecular sieve catalysts in large-scale industrial applications, allowing for seamless transition from pilot plant trials to multi-ton commercial production without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, derived directly from the experimental data and beneficial effects described in the intellectual property documentation. These insights are intended to clarify the operational benefits and feasibility of adopting this technology for industrial applications, helping stakeholders make informed decisions about process integration. By understanding the specific advantages of solid acid catalysis and one-pot synthesis, companies can better evaluate the potential return on investment and strategic fit for their existing manufacturing portfolios.
Q: What are the advantages of using solid acid catalysts over traditional Lewis acids for anethole synthesis?
A: Solid acid catalysts like H-Beta molecular sieves eliminate the need for stoichiometric amounts of corrosive Lewis acids (e.g., AlCl3), significantly reducing waste generation and simplifying post-treatment filtration compared to traditional Friedel-Crafts routes.
Q: Can this synthesis method be adapted for continuous flow processing?
A: Yes, the patent explicitly states that the reaction can be carried out in batchwise, tubular reactors, or fixed bed reactors, making it highly suitable for continuous industrial manufacturing and large-scale production.
Q: How does this method address solvent usage concerns in fine chemical manufacturing?
A: The process utilizes the aromatic compound (anisole) as both the reactant and the solvent, thereby eliminating the need for additional organic solvents, which reduces raw material costs and environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anethole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting sustainable and efficient synthetic routes to maintain competitiveness in the global fine chemicals market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and robust. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch against the highest industry standards. Whether you require custom synthesis of complex flavor intermediates or large-volume supply of established compounds, our infrastructure is designed to support your growth and innovation goals.
We invite you to collaborate with us to explore how this advanced solid acid-catalyzed technology can be tailored to your specific production needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener route. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability as your trusted partner in delivering high-quality chemical solutions efficiently and responsibly.
