Advanced Synthesis of Trans-Ketone Intermediates for High-Performance Liquid Crystal Displays
Advanced Synthesis of Trans-Ketone Intermediates for High-Performance Liquid Crystal Displays
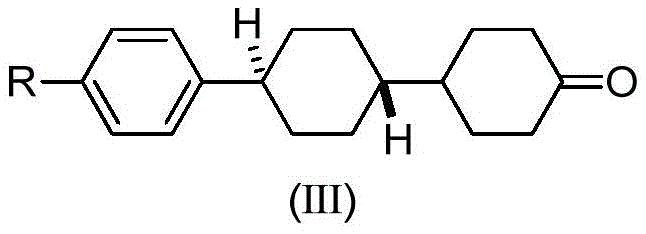
The rapid evolution of display technologies, particularly STN and TFT mixed crystal formulas, demands intermediates of exceptional purity and structural precision. Patent CN112898133B introduces a groundbreaking preparation method for trans-ketone intermediates, specifically targeting the synthesis of trans-4'-(4-methylphenyl)(1,1'-bicyclohexyl)-4-one. This technology represents a paradigm shift from traditional multi-step protection-deprotection sequences to a more streamlined approach leveraging Suzuki coupling and controlled catalytic hydrogenation. For R&D directors and procurement specialists in the liquid crystal industry, this innovation addresses critical bottlenecks regarding impurity profiles, specifically the persistent issue of benzyl alcohol residues and ketone condensation by-products that have long plagued conventional manufacturing routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of trans-ketone intermediates has relied heavily on routes exemplified by Japanese patent JP2014162752A, which utilizes dicyclohexyl ketone glycol monoketal as a pivotal intermediate. This traditional pathway is fraught with inefficiencies; the selective protection of the dihydric alcohol typically yields only about 80% selectivity, which plummets to below 60% when targeting purities exceeding 99.5%. Furthermore, the subsequent hydrolysis using concentrated formic acid not only generates substantial hazardous waste but also fosters a strong acid environment that promotes the formation of complex ketone by-product impurities. The resulting mixture often contains a myriad of known and unknown structural residues, including various cis-trans mixtures and condensed hetero-products, making final purification arduous and costly.
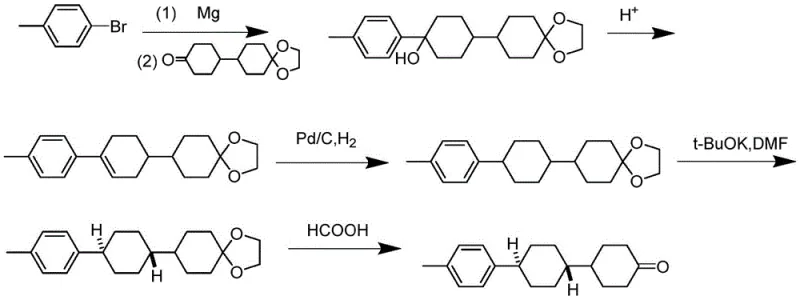
The Novel Approach
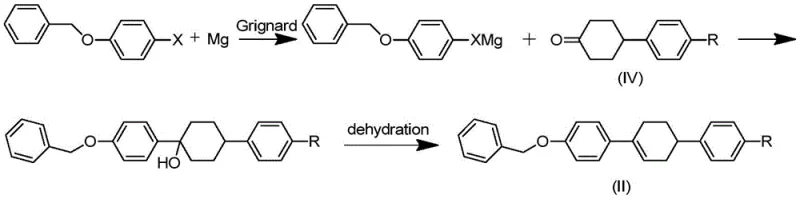
In stark contrast, the methodology disclosed in CN112898133B circumvents these pitfalls by employing benzyloxy halogenated benzene and alkyl halogenated benzene as primary feedstocks. The core innovation lies in the construction of the carbon skeleton via Suzuki coupling followed by a sophisticated hydrogenation strategy that simultaneously removes the benzyl protecting group and reduces the aromatic rings. This approach completely bypasses the problematic glycol monoketal intermediate. By introducing a benzyl ether structure, the process benefits from improved crystallinity and molecular weight characteristics that facilitate easier purification through crystallization rather than exhaustive chromatography. The result is a robust synthetic route that minimizes the generation of benzyl alcohol impurities and avoids the harsh acidic conditions responsible for macromolecular impurity formation.
Mechanistic Insights into Suzuki Coupling and Controlled Hydrogenation
The chemical elegance of this process is rooted in its precise control over reaction thermodynamics and kinetics. The initial phase involves the formation of a biaryl or aryl-cyclohexyl backbone through a palladium-catalyzed Suzuki coupling reaction. This step is critical for establishing the correct carbon framework while maintaining functional group tolerance. Following this, the catalytic hydrogenation is executed in a staged manner: initially at lower temperatures (20-40°C) and pressures (0.1-1.0 MPa) to selectively remove the benzyl group, followed by a higher temperature phase (80-120°C) and elevated pressure (0.2-1.5 MPa) to hydrogenate the phenolic ring into a ketone. This two-stage protocol is essential for preventing over-reduction or incomplete deprotection, ensuring that the intermediate ketone (Formula IV) is obtained with a purity exceeding 99.5%.
Impurity control is further enhanced during the Grignard addition and subsequent dehydration steps. Unlike the traditional route where acidolysis of the Grignard adduct leads to a complex soup of deprotected products, the benzyloxy-substituted benzyl alcohol generated here possesses a fixed dehydration site. This structural rigidity ensures that acidolysis and dehydration proceed cleanly to form the alkene (Formula II) without generating the diverse array of structural isomers seen in older methods. The final isomerization step, utilizing anhydrous aluminum trichloride as a Lewis acid catalyst at low temperatures (-15 to 5°C), effectively drives the equilibrium towards the thermodynamically stable trans-configuration, yielding the final product with a purity of not less than 99.9%.
How to Synthesize Trans-Ketone Intermediate Efficiently
The synthesis of this high-value liquid crystal intermediate requires strict adherence to anhydrous conditions and precise temperature control during the Grignard and isomerization phases. The process begins with the preparation of the biaryl precursor via Suzuki coupling, followed by a carefully monitored two-stage hydrogenation to generate the ketone intermediate. Subsequent Grignard addition and acid-catalyzed dehydration yield the alkene precursor, which is finally subjected to hydrogenation and Lewis acid-mediated isomerization. Detailed standardized operating procedures, including specific molar ratios, solvent choices, and workup protocols, are outlined below to ensure reproducibility and safety at scale.
- Perform catalytic hydrogenation on a benzyloxy-substituted biphenyl precursor (Formula I) using Pd/C to generate the ketone intermediate (Formula IV) while removing the benzyl protecting group.
- React the ketone intermediate with a Grignard reagent derived from benzyloxy halogenobenzene, followed by acidolysis and dehydration to form the alkene compound (Formula II).
- Execute a final catalytic hydrogenation and Lewis acid-catalyzed isomerization (using AlCl3) to convert the mixture into the target trans-ketone intermediate (Formula III) with purity exceeding 99.9%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and operational advantages. By eliminating the need for specialized and often expensive dicyclohexyl ketone glycol monoketal, the process relies on commodity chemicals like benzyloxy bromobenzene and methyl bromobenzene, which are readily available from multiple global suppliers. This diversification of the raw material base significantly mitigates supply chain risks associated with single-source dependencies. Moreover, the drastic simplification of the purification process—shifting from complex column chromatography to efficient crystallization—translates directly into reduced processing time and lower solvent consumption.
- Cost Reduction in Manufacturing: The elimination of the glycol protection and deprotection steps removes several unit operations from the production line, including the handling of concentrated formic acid and the associated neutralization and waste treatment costs. The higher overall yield, driven by the avoidance of low-selectivity protection steps, means that less raw material is required per kilogram of final product. Furthermore, the ability to recover and oxidize cyclohexanol by-products back into the ketone intermediate using sodium hypochlorite ensures that material efficiency is maximized, leading to substantial cost savings in raw material utilization.
- Enhanced Supply Chain Reliability: The reliance on robust, well-understood reactions such as Suzuki coupling and catalytic hydrogenation enhances the reliability of the manufacturing process. These reactions are less sensitive to minor variations in operating conditions compared to the delicate selectivity required for diol mono-protection. Consequently, batch-to-batch consistency is improved, reducing the likelihood of production delays caused by out-of-specification intermediates. The simplified workflow also shortens the overall cycle time, allowing for faster turnaround and more responsive inventory management.
- Scalability and Environmental Compliance: From an environmental perspective, the removal of concentrated formic acid hydrolysis significantly reduces the generation of acidic wastewater, simplifying effluent treatment and lowering compliance costs. The process is inherently safer and more scalable, as it avoids the handling of large volumes of corrosive acids and the generation of complex, hard-to-treat organic waste streams. The high purity of the crude product prior to final crystallization reduces the load on downstream purification units, making the entire process more sustainable and aligned with modern green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of purity, yield, and operational simplicity.
Q: How does this new method improve purity compared to traditional ketal protection routes?
A: The novel route avoids the use of dicyclohexyl ketone glycol monoketal, which traditionally suffers from low selectivity (dropping below 60% for high purity) and generates difficult-to-remove benzyl alcohol impurities. By utilizing a benzyloxy ether structure that facilitates crystallization and a controlled two-stage hydrogenation, the new method achieves purities greater than 99.9% with significantly fewer structural isomers.
Q: What are the key raw materials required for this synthesis?
A: The process primarily utilizes commercially available and cost-effective starting materials such as benzyloxy halogenobenzene (e.g., 4-benzyloxy bromobenzene) and alkyl halogenobenzene (e.g., 4-methyl bromobenzene). These are coupled via a Suzuki reaction, eliminating the need for complex, multi-step protected ketone precursors.
Q: Does this process offer environmental benefits over the prior art?
A: Yes, the method eliminates the need for concentrated formic acid hydrolysis used in traditional deprotection steps, which generates significant acidic waste. Additionally, the higher selectivity and yield reduce solvent consumption and waste generation associated with extensive purification columns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-Ketone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced liquid crystal materials requires a partner with deep technical expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN112898133B can be successfully translated from the laboratory to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of trans-ketone intermediate meets the exacting standards required for high-performance display applications.
We invite you to collaborate with us to optimize your supply chain for liquid crystal materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value and reliability for your organization.
