Advanced Lithium Salt Purification for High-Purity Glucuronide Compounds Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for the purification of complex glycoside structures, particularly glucuronide compounds which serve as critical intermediates in the development of novel therapeutic agents. Patent CN102786553A introduces a groundbreaking purification method that addresses the longstanding challenges associated with isolating high-purity glucuronide derivatives from crude reaction mixtures containing structurally similar impurities. This innovation leverages the unique coordination chemistry of lithium ions to selectively precipitate the target glucuronide compound as an insoluble lithium salt complex, effectively separating it from key by-products and colored pigments that traditionally compromise product quality. By shifting the paradigm from expensive chromatographic separation to a cost-effective precipitation strategy, this technology offers a scalable solution that aligns with the rigorous demands of modern Good Manufacturing Practice (GMP) environments. The strategic implementation of this purification protocol enables manufacturers to achieve purity levels exceeding 99.9%, thereby ensuring the safety and efficacy required for downstream pharmaceutical applications while significantly optimizing the overall production economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of glucuronide compounds has been plagued by significant technical bottlenecks that hinder efficient large-scale production and increase overall manufacturing costs. Traditional techniques such as column chromatography or preparative chromatography, while effective on a laboratory scale, are notoriously difficult to industrialize due to their high consumption of solvents, silica gel, and extensive processing time. Furthermore, direct recrystallization methods often fail to adequately remove specific structural impurities, particularly those generated during the deprotection steps of the synthesis, leading to products that do not meet stringent medicinal standards. The presence of conjugated double-bond impurities, which exhibit physicochemical properties very similar to the target molecule, makes separation via standard physical methods exceptionally challenging and often results in substantial yield losses. Additionally, the formation of dark-colored pigments during the reaction process further complicates the purification landscape, requiring additional decolorization steps that add complexity and reduce the overall economic viability of the manufacturing process.
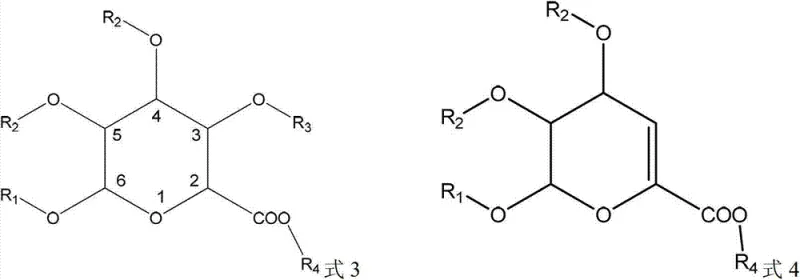
The Novel Approach
In stark contrast to these conventional limitations, the novel approach detailed in the patent utilizes a sophisticated lithium salt complexation mechanism that exploits subtle structural differences between the target glucuronide and its impurities. By carefully adjusting the pH of the solution to an alkaline range between 7 and 14 in the presence of a lithium-containing compound, the target glucuronide compound selectively forms an insoluble lithium salt complex that precipitates out of the solvent. This precipitation phenomenon is highly specific to the target structure, which possesses the necessary hydroxyl and carboxylic acid geometry to coordinate with lithium ions, whereas the key impurity containing a double bond does not form this complex and remains dissolved in the mother liquor. This selective precipitation not only achieves a high degree of separation efficiency but also simultaneously removes colored pigments that are trapped within the crystal lattice or remain in the solution, resulting in a significantly whiter and purer final product. The simplicity of this filtration-based separation eliminates the need for complex chromatographic equipment, thereby streamlining the workflow and enhancing the throughput capacity for industrial-scale operations.
Mechanistic Insights into Lithium Salt Complexation Purification
The core of this purification technology lies in the precise coordination chemistry between the glucuronide molecule and lithium ions under controlled alkaline conditions. The target glucuronide compound, characterized by specific hydroxyl groups at the 2 and 3 positions along with a carboxylic acid group, creates a unique spatial arrangement that allows for stable chelation with lithium cations. When the pH is adjusted to the optimal range, typically between 11 and 13 using lithium hydroxide, the deprotonated carboxylate and hydroxyl groups interact strongly with the lithium ions to form a rigid, water-insoluble complex lattice. This lattice structure is thermodynamically stable and precipitates rapidly from the aqueous or organic solvent mixture, effectively locking the target molecule into a solid phase that can be easily isolated via filtration. The structural integrity of this complex ensures that the target compound is protected from degradation during the separation process, maintaining its stereochemical configuration and chemical identity throughout the purification cycle.
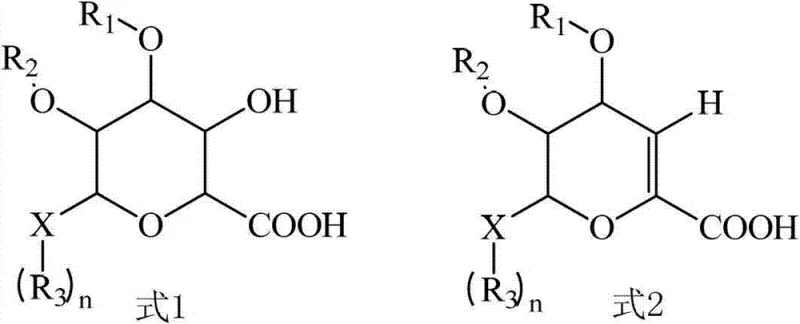
Conversely, the key impurity, which often arises from elimination reactions during synthesis, possesses a double bond that alters the electronic distribution and spatial geometry of the molecule. This structural modification prevents the impurity from adopting the necessary conformation to form a stable complex with lithium ions under the same reaction conditions. As a result, the impurity remains in the soluble phase, allowing for a clean physical separation between the solid lithium salt complex of the target and the liquid containing the contaminants. This mechanism also extends to the removal of colored by-products, which are either excluded from the growing crystal lattice of the lithium complex or remain solvated in the mother liquor due to their different polarity profiles. By repeating the dissolution and re-precipitation cycle, manufacturers can iteratively enhance the purity of the lithium salt complex, ensuring that the final regenerated glucuronide compound meets the highest standards of chemical purity required for pharmaceutical use.
How to Synthesize Glucuronide Compounds Efficiently
The implementation of this purification protocol requires careful attention to reaction parameters such as temperature, pH, and stirring speed to maximize the yield and purity of the lithium salt complex. Operators must dissolve the crude glucuronide mixture in a suitable solvent system, typically water or a water-organic solvent mixture, and maintain the temperature between 10 and 40 degrees Celsius during the addition of the lithium source. The detailed standardized synthesis steps, including specific molar ratios, stirring times, and filtration protocols, are outlined in the structured guide below to ensure reproducibility and safety across different production batches. Adhering to these precise operational guidelines is critical for achieving the consistent removal of impurities and pigments that defines the success of this purification strategy in a commercial setting.
- Dissolve crude glucuronide mixture in solvent and add lithium compound at 10-40°C, adjusting pH to 7-14 to form insoluble lithium salt complex.
- Separate the precipitated lithium salt complex by filtration and dissolve it in solvent by adding acid to adjust pH.
- Add alkali to the solution to adjust pH to 7-8, causing the high-purity glucuronide compound to precipitate and separate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this purification method offers substantial advantages by fundamentally simplifying the downstream processing requirements for glucuronide intermediates. The elimination of chromatographic purification steps translates directly into a significant reduction in the consumption of expensive stationary phases and organic solvents, which are major cost drivers in traditional pharmaceutical manufacturing. Furthermore, the use of inexpensive and readily available lithium salts as the primary purification reagent ensures that the raw material costs remain low and stable, shielding the supply chain from volatility associated with specialized chromatography media. The robustness of the precipitation process also reduces the dependency on highly skilled operators for complex column packing and elution, thereby lowering labor costs and minimizing the risk of human error during production runs.
- Cost Reduction in Manufacturing: The transition from chromatography to precipitation-based purification drastically lowers the operational expenditure associated with solvent recovery and waste disposal. By avoiding the use of large volumes of organic solvents required for column chromatography, the facility can reduce its environmental footprint and lower the costs related to solvent procurement and recycling infrastructure. The high yield of the precipitation process ensures that less raw material is wasted during purification, maximizing the output from each batch of crude reaction mixture and improving the overall cost-efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The simplicity of the equipment required for this purification method, primarily consisting of standard reactors and filtration units, enhances the reliability of the supply chain by reducing the risk of equipment failure or bottlenecks. Unlike chromatography systems which require specialized maintenance and calibration, the precipitation process can be executed in general-purpose chemical processing equipment that is widely available in most manufacturing facilities. This flexibility allows for faster scale-up and more consistent production schedules, ensuring that customers receive their orders on time without delays caused by complex purification logistics.
- Scalability and Environmental Compliance: This method is inherently scalable from laboratory to industrial production without the need for significant process re-engineering, facilitating the commercial scale-up of complex pharmaceutical intermediates. The reduction in solvent usage and the ability to use water as a primary solvent align with green chemistry principles, making it easier to comply with increasingly stringent environmental regulations regarding volatile organic compound emissions. The generation of less hazardous waste simplifies the disposal process and reduces the regulatory burden on the manufacturing site, contributing to a more sustainable and compliant operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this lithium salt complexation purification technology for glucuronide compounds. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this method into existing manufacturing workflows and assessing its impact on product quality and cost structures.
Q: How does the lithium salt method remove pigments from glucuronide compounds?
A: The lithium salt complex forms a specific crystal lattice that excludes colored impurities, which remain in the mother liquor or are filtered out during the precipitation process.
Q: What purity level can be achieved with this purification method?
A: Experimental data indicates that this method can consistently achieve purity levels exceeding 99.9% by effectively separating the target compound from key structural impurities.
Q: Is this purification method suitable for large-scale industrial production?
A: Yes, the method uses standard equipment like reactors and filters, avoids expensive chromatography, and is designed for stable, low-cost large batch industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glucuronide Compounds Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing innovation, leveraging advanced purification technologies like the lithium salt complexation method to deliver superior quality glucuronide compounds to the global market. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet the demanding volume requirements of multinational pharmaceutical companies with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to verify that every batch of glucuronide intermediate meets the highest industry standards for impurity profiles and chemical identity before release.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this efficient purification strategy. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term commercial goals and enhance the reliability of your raw material supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
