Revolutionizing 3-Cyanoindole Production via Green Electrochemical Decarboxylation for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking sustainable alternatives to traditional synthetic methodologies, particularly for the construction of complex heterocyclic scaffolds. Patent CN113930792B introduces a groundbreaking electrochemical preparation method for 3-cyanoindole compounds, specifically detailing the synthesis of 1-methyl-2-phenyl-1H-indole-3-carbonitrile. This technology represents a paradigm shift from stoichiometric chemical oxidation to electricity-driven synthesis, utilizing clean electrons as the primary redox reagent. By leveraging an undivided cell configuration, the process achieves high selectivity and yield under remarkably mild conditions, effectively bypassing the need for toxic cyanide sources or expensive transition metal catalysts. For R&D directors and process chemists, this patent offers a robust pathway to access valuable indole nitriles, which are critical precursors for various bioactive molecules including anticancer and antiviral agents. The integration of this electrochemical strategy into existing supply chains promises not only environmental compliance but also significant operational simplifications for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-cyanoindole derivatives has been plagued by significant safety and environmental challenges associated with traditional cyanation protocols. Conventional routes often necessitate the use of stoichiometric amounts of toxic metal cyanides, such as potassium cyanide (KCN), copper cyanide (CuCN), or trimethylsilyl cyanide (TMSCN), which pose severe handling risks and require stringent waste disposal measures to prevent environmental contamination. Furthermore, alternative methods employing electrophilic CN+ reagents, such as N-cyano-N-phenyl-p-toluenesulfonamide, demand the pre-preparation of specialized reagents and frequently rely on Lewis acid catalysts or excessive bases that complicate the reaction workup. Other reported strategies involving nitriles or isonitriles as cyanide sources typically require high reaction temperatures, often exceeding 150°C, and the use of stoichiometric oxidants, leading to poor atom economy and the generation of substantial chemical waste. These inherent drawbacks render many legacy methods unsuitable for modern green manufacturing standards, creating bottlenecks in both cost efficiency and regulatory compliance for the production of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome traditional techniques, the electrochemical method disclosed in patent CN113930792B offers a streamlined and environmentally benign solution for constructing the cyano-indole core. This innovative approach utilizes an oxime ester derivative, specifically 2-methyl-2-((((1-methyl-2-phenyl-1H-indol-3-yl)methylene)amino)oxy)propionic acid, as the sole precursor for both the indole scaffold and the nitrile functionality. By applying a constant electric current in an undivided cell at room temperature, the system triggers a cascade of oxidative decarboxylation and nitrogen-oxygen bond cleavage events without the need for external chemical oxidants. The use of simple carbon rod anodes and platinum sheet cathodes eliminates the dependency on precious metal catalysts, while the mild reaction conditions preserve sensitive functional groups that might otherwise degrade under harsh thermal or acidic environments. This methodology not only simplifies the operational procedure but also aligns perfectly with the principles of green chemistry by minimizing waste generation and energy consumption.
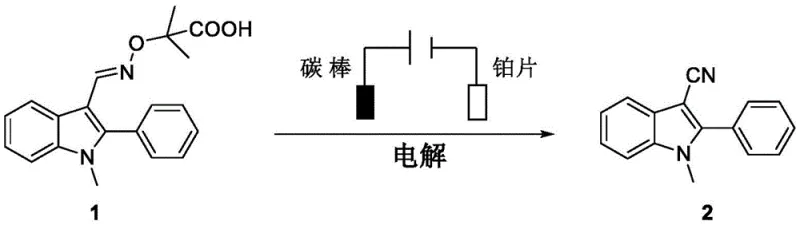
Mechanistic Insights into Electrochemical Decarboxylative Cyanation
The core of this transformative synthesis lies in the anodic oxidation mechanism that drives the fragmentation of the oxime ester substrate to generate the key reactive intermediates. Upon application of the electric potential, the carboxylate anion derived from the substrate undergoes single-electron oxidation at the anode surface to form a carboxyl radical species. This unstable intermediate rapidly loses a molecule of carbon dioxide (decarboxylation) to generate an oxygen-centered radical, which subsequently induces the homolytic cleavage of the adjacent nitrogen-oxygen bond. This critical bond scission releases an imine nitrogen radical species that is strategically positioned to undergo intramolecular rearrangement or cyclization, ultimately forging the carbon-nitrogen triple bond of the nitrile group. The entire catalytic cycle is sustained by the continuous flow of electrons, acting as a traceless reagent that avoids the accumulation of reduced byproducts typically associated with chemical oxidants. Understanding this radical-mediated pathway is crucial for process optimization, as it highlights the importance of current density and electrode material in controlling the rate of radical generation and preventing side reactions.
From an impurity control perspective, this electrochemical mechanism offers distinct advantages over metal-catalyzed counterparts by inherently avoiding the introduction of heavy metal residues. In traditional palladium or copper-catalyzed cyanations, trace amounts of these metals can persist in the final product, necessitating expensive and time-consuming purification steps such as scavenger treatment or recrystallization to meet strict ICH Q3D guidelines for elemental impurities. The metal-free nature of this electrochemical protocol ensures that the crude reaction mixture is free from transition metal contaminants, significantly simplifying the downstream purification train. Furthermore, the high selectivity of the radical generation process minimizes the formation of regioisomers or over-oxidized byproducts, resulting in a cleaner crude profile. This purity advantage is particularly valuable for the synthesis of pharmaceutical intermediates, where impurity profiles directly impact the safety and efficacy of the final drug substance, thereby reducing the overall cost of goods sold through improved yield and reduced processing time.
How to Synthesize 1-Methyl-2-phenyl-1H-indole-3-carbonitrile Efficiently
To implement this electrochemical synthesis effectively, precise control over reaction parameters such as electrolyte concentration, solvent choice, and current intensity is essential to maximize yield and reproducibility. The patent outlines a robust protocol where the substrate is dissolved in methanol along with a supporting electrolyte like n-tetrabutylammonium perchlorate and a base such as cesium carbonate to facilitate the initial deprotonation and conductivity. The detailed standardized synthesis steps below provide a clear roadmap for replicating this high-yielding transformation in a laboratory or pilot plant setting, ensuring that the benefits of this green chemistry approach can be fully realized in practical applications.
- Prepare the electrolyte solution by dissolving the oxime ester substrate, a base such as cesium carbonate, and a supporting electrolyte like n-tetrabutylammonium perchlorate in methanol within an undivided electrolytic cell.
- Insert a carbon rod anode and a platinum sheet cathode into the reaction mixture, ensuring a fixed distance between electrodes, and apply a constant direct current at room temperature.
- Monitor the reaction progress via TLC until completion, then proceed with standard workup procedures including solvent removal, extraction with ethyl acetate, and purification to isolate the target nitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology presents a compelling value proposition centered on cost reduction and supply reliability. By eliminating the requirement for expensive and volatile transition metal catalysts, the raw material bill of materials is significantly optimized, removing the price volatility associated with precious metals like palladium or rhodium. Moreover, the avoidance of toxic cyanide salts reduces the regulatory burden and insurance costs related to hazardous material storage and transportation, allowing for a more agile and compliant supply chain operation. The simplicity of the reactor setup, which does not require high-pressure vessels or extreme temperature control systems, further lowers the capital expenditure required for scaling up production capacity.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chemical oxidants and expensive metal catalysts directly translates to substantial cost savings in raw material procurement. Since the reaction relies on electricity as the primary driver, the variable costs are largely tied to energy consumption, which is generally more stable and predictable than the fluctuating prices of specialized chemical reagents. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary chemicals and solvents, further driving down the overall manufacturing cost per kilogram of the high-purity pharmaceutical intermediate produced.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as methanol, carbon rods, and simple salts like cesium carbonate ensures a robust and resilient supply chain that is less susceptible to geopolitical disruptions or single-source supplier bottlenecks. Unlike specialized ligands or custom-synthesized cyanating agents that may have long lead times, the reagents for this electrochemical process are widely available from multiple global vendors. This accessibility guarantees consistent production schedules and reduces the risk of stockouts, enabling manufacturers to maintain steady inventory levels and meet tight delivery deadlines for their downstream clients in the pharmaceutical sector.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of undivided cells make this process highly amenable to scale-up from benchtop to commercial tonnage production without significant engineering hurdles. The absence of toxic gas evolution or hazardous waste streams simplifies the environmental permitting process and reduces the cost of waste treatment facilities. This alignment with green chemistry principles not only enhances the corporate sustainability profile but also future-proofs the manufacturing process against increasingly stringent environmental regulations, ensuring long-term operational continuity and market competitiveness.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 3-cyanoindoles, providing clarity on the feasibility and benefits of adopting this novel methodology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, offering a reliable basis for decision-making.
Q: What are the primary safety advantages of this electrochemical method over traditional cyanation?
A: Unlike conventional methods that rely on highly toxic metal cyanides like KCN or CuCN, this electrochemical approach generates the cyano group internally through substrate fragmentation, eliminating the need to handle hazardous external cyanide sources and significantly reducing workplace safety risks.
Q: Does this process require expensive transition metal catalysts?
A: No, the method operates without transition metal catalysts such as palladium or copper. The reaction is driven by electron transfer at the electrode surface, which removes the risk of heavy metal contamination in the final API intermediate and lowers raw material costs.
Q: Is this electrochemical protocol suitable for large-scale industrial production?
A: Yes, the process utilizes simple undivided cells, operates at room temperature, and uses commercially available electrodes like carbon rods and platinum sheets. These factors indicate high scalability and ease of adaptation to flow chemistry or large batch reactors for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Methyl-2-phenyl-1H-indole-3-carbonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113930792B can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1-methyl-2-phenyl-1H-indole-3-carbonitrile delivered meets the highest quality standards required by global regulatory agencies.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and green synthetic route for their pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this electrochemical method can optimize your budget. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that enhance both the efficiency and sustainability of your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
