Revolutionizing Anisyl Propionaldehyde Production: A Cost-Effective Anisole Alkylation Strategy for Global Markets
Revolutionizing Anisyl Propionaldehyde Production: A Cost-Effective Anisole Alkylation Strategy for Global Markets
The global demand for high-quality fragrance intermediates is constantly evolving, driven by the need for cost-effective and environmentally sustainable manufacturing processes. Patent CN113527077A introduces a groundbreaking methodology for the preparation of anisyl propionaldehyde, a valuable compound known for its fruity, fennel, and flowery aroma, directly from anisole. This technical breakthrough addresses the critical economic bottleneck associated with traditional synthesis routes that rely on expensive precursors like anisaldehyde or anisyl alcohol. By shifting the feedstock to anisole, a cheap and readily available bulk chemical, this innovation offers a compelling value proposition for manufacturers seeking to optimize their supply chains. The process not only simplifies the production flow but also ensures the generation of high-purity end products suitable for sensitive applications in the flavor and fragrance industry. 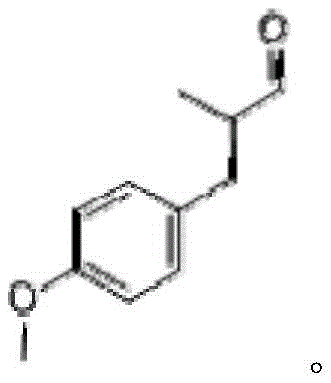
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial production of anisyl propionaldehyde has been heavily dependent on semi-synthetic routes starting from heliotropin or anisaldehyde, which are themselves derived from natural sources like anise oil or through complex oxidation processes. These conventional pathways typically involve Claisen-Schmidt condensation followed by catalytic hydrogenation, often requiring precious metal catalysts such as palladium-carbon under high pressure. Such methods are fraught with inefficiencies, including low yields, complex post-treatment procedures, and the generation of significant amounts of salt-containing wastewater due to the use of liquid alkalis. Furthermore, the reliance on anisaldehyde ties the production cost to the volatile pricing of natural essential oils or energy-intensive oxidation steps, creating supply chain vulnerabilities. The difficulty in recycling phase transfer catalysts and the challenges associated with separating byproducts like anisyl propanol further exacerbate the operational costs and environmental footprint of these legacy technologies.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes anisole as the primary starting material, bypassing the need for pre-oxidized intermediates entirely. This strategy employs a direct alkylation reaction where anisole reacts with 2-methylallyl diacetate or a combination of 2-methylacrolein and acetic anhydride under Lewis acid catalysis. This fundamental shift in synthetic logic drastically shortens the production flow, eliminating the energy-intensive hydrogenation step and the associated safety risks of high-pressure reactors. The process operates under milder conditions, typically between -10°C and 80°C, and utilizes recoverable catalysts that reduce waste generation. By integrating a transesterification step to convert the intermediate acetoxyl compound into the final aldehyde, the method achieves high conversion rates while allowing for the efficient recovery and reuse of excess anisole. This results in a streamlined, economically superior process that decouples production costs from the fluctuating prices of specialty aldehydes.
Mechanistic Insights into Lewis Acid-Catalyzed Alkylation and Transesterification
The core of this innovative synthesis lies in the efficient Friedel-Crafts-type alkylation of the aromatic ring of anisole. In the first stage, a Lewis acid catalyst, such as boron trifluoride or ferric chloride, activates the electrophilic species derived from 2-methylallyl diacetate or the in-situ generated equivalent from 2-methylacrolein and acetic anhydride. This activation facilitates the nucleophilic attack by the electron-rich anisole ring, preferentially at the para-position due to the directing effect of the methoxy group, forming the key intermediate 1-acetoxy-2-methyl-3-(4-methoxyphenyl)propylene. The reaction conditions are tightly controlled to minimize side reactions, with the temperature maintained below 40°C during catalyst addition to prevent polymerization or over-alkylation. The use of specific Lewis acids allows for high regioselectivity and conversion, ensuring that the residual amount of the alkylating agent is reduced to less than 1%, which is critical for downstream purification efficiency.
Following the alkylation, the process transitions to a transesterification or hydrolysis step to unveil the aldehyde functionality. The intermediate acetoxyl compound is reacted in an alcohol solvent, such as methanol or ethanol, in the presence of an ester exchange catalyst like potassium carbonate or sodium methoxide. This step effectively cleaves the acetate group, yielding the target anisyl propionaldehyde and a corresponding ester byproduct which is easily removed via distillation. The mechanistic elegance of this two-step sequence lies in its modularity; the intermediate can be isolated or processed in situ, offering flexibility in plant operations. Moreover, the preheating of the intermediate before addition to the alcohol solvent enhances reaction kinetics, ensuring complete conversion and minimizing the formation of impurities. This precise control over the reaction mechanism is what enables the production of anisyl propionaldehyde with purity levels exceeding 99%, meeting the stringent requirements of fine fragrance applications. 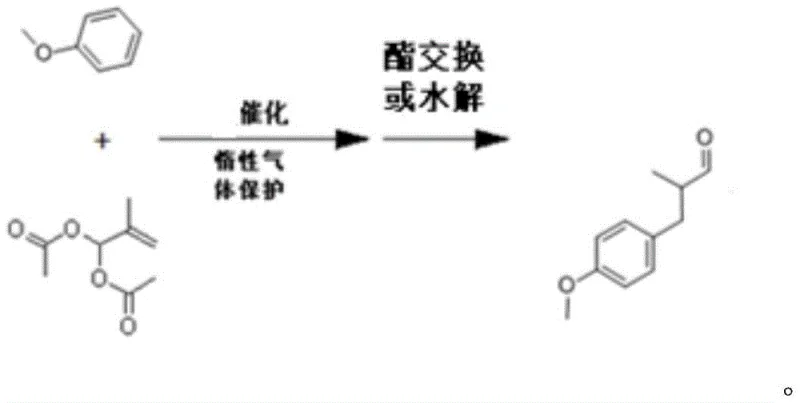
How to Synthesize Anisyl Propionaldehyde Efficiently
The synthesis of anisyl propionaldehyde via this patented route involves a carefully orchestrated sequence of alkylation and ester exchange reactions that maximize yield while minimizing waste. The process begins with the mixing of anisole and the alkylating agent under inert atmosphere protection, followed by the controlled addition of the catalyst to initiate the formation of the acetoxyl intermediate. Detailed operational parameters, including specific molar ratios and temperature gradients, are critical to achieving the high conversion rates reported in the patent examples. For R&D teams looking to implement this technology, understanding the nuances of the workup procedure—specifically the alkaline washing and vacuum distillation steps—is essential for isolating the intermediate with high purity before the final conversion. The standardized synthesis steps outlined below provide a robust framework for scaling this chemistry from the laboratory to commercial production.
- Mix anisole and 2-methylallyl diacetate (or 2-methylacrolein and acetic anhydride) with a Lewis acid catalyst at low temperatures (-10 to 40°C) to form the acetoxyl intermediate.
- Wash the reaction mixture with alkaline solution, separate the oil phase, and recover unreacted anisole via reduced pressure distillation.
- React the intermediate with an alcohol solvent and ester exchange catalyst at 10-80°C, followed by distillation and rectification to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this anisole-based synthesis route presents a transformative opportunity to enhance margin stability and operational resilience. The most significant advantage is the drastic reduction in raw material costs, as anisole is a commodity chemical with a stable and abundant global supply, unlike anisaldehyde which is subject to agricultural and extraction variabilities. By eliminating the dependency on expensive natural extracts or multi-step oxidation precursors, manufacturers can secure a more predictable cost structure. Furthermore, the simplified process flow reduces the capital expenditure required for specialized high-pressure equipment, lowering the barrier to entry for production and decreasing maintenance overheads. The ability to recover and recycle excess anisole directly within the process loop further amplifies these cost savings, ensuring that material utilization is optimized to the highest degree possible without compromising product quality.
- Cost Reduction in Manufacturing: The substitution of high-cost anisaldehyde with low-cost anisole fundamentally alters the cost basis of the final product. Additionally, the removal of the hydrogenation step eliminates the need for expensive palladium catalysts and high-pressure reactors, leading to substantial savings in both CAPEX and OPEX. The process also avoids the use of phase transfer catalysts which are often costly and difficult to recover, thereby reducing the overall chemical consumption per kilogram of product. These cumulative efficiencies result in a significantly leaner manufacturing cost structure that provides a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: Sourcing anisole is far more reliable than sourcing anisaldehyde or anise oil, as it is produced on a massive scale for various industrial applications. This abundance ensures that production schedules are not disrupted by raw material shortages or seasonal fluctuations common in botanical supply chains. The robustness of the synthetic route also means that production lead times can be shortened, as the reaction times are relatively fast and the workup procedures are straightforward. This reliability allows supply chain managers to maintain lower inventory buffers while still meeting customer demand, improving working capital efficiency and responsiveness to market changes.
- Scalability and Environmental Compliance: The process is inherently scalable due to its use of standard unit operations like distillation and liquid-liquid extraction, which are well-understood in chemical engineering. From an environmental perspective, the method generates significantly less wastewater and solid waste compared to traditional alkali-catalyzed condensations, simplifying effluent treatment and reducing disposal costs. The reduced generation of hazardous waste and the absence of heavy metal residues in the final product align with increasingly strict global regulatory standards for fragrance ingredients. This environmental compatibility not only mitigates regulatory risk but also enhances the brand value of the end products in eco-conscious consumer markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on the feasibility and benefits of the anisole route. Understanding these details is crucial for technical teams evaluating the transition from legacy processes to this more efficient methodology. The insights provided here cover aspects ranging from catalyst selection to product purity specifications, ensuring a comprehensive understanding of the technology's potential.
Q: What are the primary cost advantages of the anisole-based synthesis route?
A: The primary advantage lies in substituting expensive anisaldehyde or anisyl alcohol with anisole, a widely available and inexpensive commodity chemical. Additionally, the process eliminates the need for high-pressure hydrogenation steps and expensive palladium-carbon catalysts required in traditional methods.
Q: How does this method improve environmental compliance compared to conventional processes?
A: This method significantly reduces the generation of 'three wastes' (wastewater, waste gas, solid waste) by avoiding the use of large amounts of liquid alkali and phase transfer catalysts that are difficult to recycle. The shorter production flow and ability to recover excess anisole further minimize environmental impact.
Q: What purity levels can be achieved with this synthetic pathway?
A: The patent data indicates that through vacuum rectification of the crude product obtained after transesterification, purity levels exceeding 99% can be consistently achieved, making the product suitable for high-grade perfumery and food flavor applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anisyl Propionaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic routes like the one described in CN113527077A to maintain competitiveness in the global fragrance market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity anisyl propionaldehyde that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement this cost-effective anisole-based process allows us to offer a superior value proposition to our partners, combining technical excellence with economic efficiency.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your fragrance ingredient sourcing with a solution that balances performance, purity, and profitability.
