Advanced Synthesis of N-methylhexahydroazepin-4-one Hydrochloride for Scalable Azelastine Production
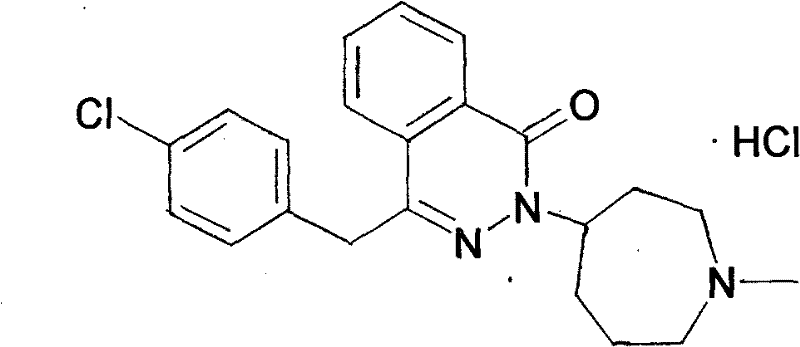
The pharmaceutical landscape for antihistamines continues to evolve, with Azelastine Hydrochloride (W-2979M) remaining a cornerstone treatment for allergic rhinitis due to its superior pharmacokinetic profile and dual-action mechanism. Central to the efficient manufacturing of this active pharmaceutical ingredient (API) is the availability of high-quality intermediates, specifically N-methylhexahydroazepin-4-one hydrochloride. Patent CN101781248B introduces a transformative synthetic methodology that addresses long-standing bottlenecks in producing this critical seven-membered heterocyclic building block. By shifting the starting material paradigm from expensive, specialized diesters to the commodity chemical N-methyl-2-pyrrolidone (NMP), this technology offers a robust pathway for cost reduction in pharmaceutical intermediates manufacturing. The innovation lies not just in the raw material switch, but in the optimization of reaction conditions that enhance yield and simplify purification, thereby securing a more reliable supply chain for downstream API producers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the azepinone core for Azelastine has relied on routes that are economically and operationally burdensome for large-scale facilities. Traditional methods, such as those disclosed in patent DE4343409, utilize complex precursors like 4-[(3-ethoxy-3-oxopropyl)methylamino] ethyl ester. These starting materials are not only costly to procure but also introduce significant logistical friction into the supply chain due to their specialized nature. Furthermore, the conventional processes often necessitate harsh hydrolysis conditions followed by energy-intensive evaporation to dryness, which places heavy demands on equipment and utility consumption. The cumulative effect of these factors is a process with lower overall throughput and higher waste generation, creating a substantial barrier to achieving cost reduction in electronic chemical manufacturing or similar high-volume sectors where efficiency is paramount. The difficulty in sourcing high-purity diester precursors often leads to batch-to-batch variability, complicating quality control protocols.
The Novel Approach
In stark contrast, the novel approach detailed in CN101781248B leverages N-methyl-2-pyrrolidone (NMP) as a ubiquitous and inexpensive feedstock. This strategic shift eliminates the dependency on hard-to-find diester intermediates, fundamentally altering the cost structure of the synthesis. The new route employs a streamlined sequence involving acid-catalyzed ring opening, followed by esterification and a Michael addition-cyclization cascade. This methodology allows for the use of single solvents throughout significant portions of the reaction sequence, drastically simplifying solvent recovery and recycling operations. By avoiding the tedious evaporation steps associated with prior art, the new process minimizes thermal stress on the product, reducing the formation of degradation by-products. This operational simplicity translates directly into enhanced supply chain reliability, as the reliance on a stable, bulk chemical supply replaces the volatility of niche reagent markets.
Mechanistic Insights into NMP-Based Ring Expansion and Cyclization
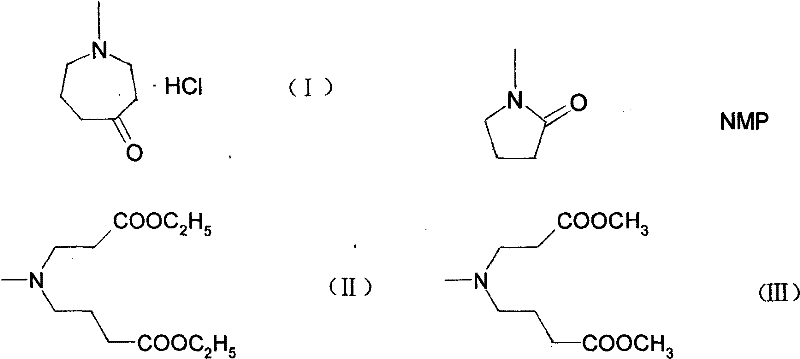
The core of this synthetic breakthrough involves a sophisticated manipulation of the pyrrolidine ring to construct the larger azepine system. The process initiates with the acid-catalyzed hydrolysis of NMP, where refluxing in hydrochloric acid cleaves the amide bond to yield 4-methylaminobutyric acid hydrochloride. This ring-opening step is critical as it linearizes the five-membered precursor, preparing the carbon chain for subsequent elongation. Following this, the carboxylic acid functionality is activated via reaction with thionyl chloride in methanol, forming the methyl ester hydrochloride salt. This esterification is performed under controlled low-temperature conditions to prevent side reactions, ensuring the integrity of the amine salt. The resulting intermediate then undergoes a Michael addition with methyl acrylate in the presence of triethylamine, effectively extending the carbon chain by two units and setting the stage for ring closure. This precise control over chain elongation is vital for maintaining the regioselectivity required for the final cyclization step.
The final transformation is an intramolecular condensation driven by a strong base, typically potassium tert-butoxide, which facilitates the formation of the seven-membered azepinone ring. This Dieckmann-like cyclization is highly sensitive to reaction conditions; the patent specifies the use of non-polar solvents like xylene or toluene to manage the solubility of the intermediates and by-products. The base abstracts a proton from the alpha-position of the ester, generating an enolate that attacks the distal ester carbonyl, closing the ring and expelling an alkoxide leaving group. Subsequent acidification and pH adjustment allow for the selective precipitation of the target hydrochloride salt. This mechanistic pathway ensures high purity, with reported gas chromatography values exceeding 99%, by effectively separating the cyclic product from linear oligomers or unreacted starting materials through crystallization. The ability to tune the pH during the salification step provides an additional layer of impurity control, essential for meeting stringent pharmaceutical specifications.
How to Synthesize N-methylhexahydroazepin-4-one Hydrochloride Efficiently
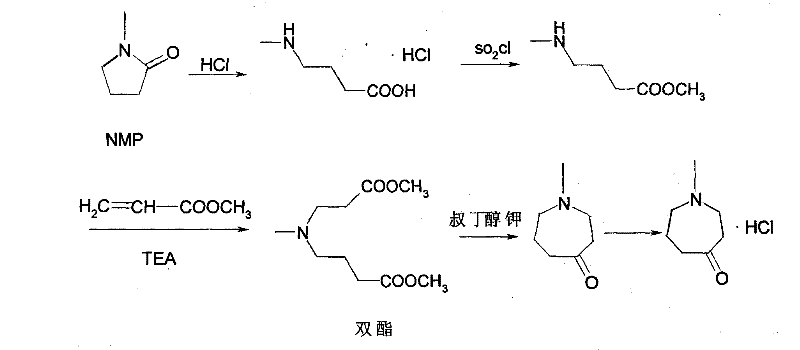
The synthesis protocol outlined in the patent provides a clear roadmap for replicating this high-yield process in a pilot or production environment. The procedure begins with the reflux of NMP in concentrated hydrochloric acid, followed by solvent removal and crystallization to isolate the amino acid salt. This solid is then subjected to esterification using thionyl chloride and methanol, a reaction that must be carefully temperature-controlled to maximize conversion. The resulting ester salt is subsequently reacted with methyl acrylate and triethylamine to form the diester intermediate, which serves as the direct precursor to the ring closure. The final cyclization step requires the slow addition of the diester to a refluxing solution of potassium tert-butoxide in xylene, followed by a careful workup involving acid extraction and basification. For the complete, standardized operating procedures including exact stoichiometric ratios, temperature profiles, and safety handling instructions, please refer to the detailed guide below.
- Reflux N-methyl-2-pyrrolidone (NMP) with hydrochloric acid to effect ring-opening hydrolysis, yielding 4-methylaminobutyric acid hydrochloride.
- Perform mono-esterification by reacting the amino acid salt with thionyl chloride in methanol to generate 4-methylaminobutyric acid methyl ester hydrochloride.
- Execute a Michael addition with methyl acrylate followed by base-catalyzed intramolecular cyclization using potassium tert-butoxide to form the seven-membered azepinone ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this NMP-based synthesis route offers compelling economic and operational benefits that extend beyond simple raw material substitution. The shift from specialized diesters to NMP represents a move towards a more resilient supply chain model, mitigating the risks associated with single-source suppliers of niche chemicals. Since NMP is produced on a massive global scale for various industrial applications, its availability is virtually guaranteed, ensuring continuity of supply even during market fluctuations. Furthermore, the simplification of the reaction workflow reduces the total processing time and labor hours required per batch. The elimination of complex evaporation steps and the ability to recycle solvents like isopropanol and xylene contribute to a significant reduction in utility costs and waste disposal fees. These factors combine to create a manufacturing process that is not only cheaper to operate but also more environmentally sustainable, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of expensive, multi-step synthesized diester precursors with N-methyl-2-pyrrolidone, a low-cost commodity chemical. This fundamental change in the bill of materials drastically lowers the direct material costs associated with each kilogram of intermediate produced. Additionally, the process design minimizes the number of unit operations; by avoiding energy-intensive evaporation to dryness and utilizing efficient crystallization techniques, the facility saves on steam and electricity consumption. The high yield reported in the patent examples implies that less raw material is wasted to side products, further enhancing the overall material efficiency. Consequently, the total cost of goods sold (COGS) for the intermediate is substantially reduced, allowing for more competitive pricing strategies in the final API market.
- Enhanced Supply Chain Reliability: Relying on NMP as the starting material decouples the production of Azelastine intermediates from the volatile supply chains of fine chemical esters. NMP is a standard solvent and reagent available from numerous global manufacturers, reducing the risk of supply disruptions caused by production issues at a single vendor. The robustness of the synthetic route also means that the process is less sensitive to minor variations in reagent quality, providing a buffer against supply chain inconsistencies. Moreover, the simplified purification steps reduce the lead time for releasing batches for quality control, enabling faster turnover and more responsive inventory management. This reliability is crucial for maintaining the continuous production schedules required by large-scale pharmaceutical clients who cannot afford interruptions in their API synthesis.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor configurations and common solvents that pose no extraordinary engineering challenges. The reduction in wastewater volume, achieved through efficient solvent recycling and minimized aqueous workups, simplifies effluent treatment and lowers compliance costs. The use of less hazardous reagents and the avoidance of extreme conditions contribute to a safer working environment, reducing the regulatory burden associated with handling dangerous chemicals. As production volumes increase from kilograms to tons, the linearity of this process ensures that yields remain consistent, preventing the typical drop-off in efficiency seen when scaling complex, multi-step syntheses. This scalability makes the technology an ideal candidate for establishing long-term, high-volume manufacturing capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and comparative analysis provided in the patent documentation. Understanding these details is essential for R&D teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source. The answers reflect the practical realities of running this chemistry in a GMP-compliant environment.
Q: What are the primary advantages of using NMP over traditional diester precursors for this synthesis?
A: Using N-methyl-2-pyrrolidone (NMP) significantly reduces raw material costs as it is a widely available commodity chemical compared to specialized diesters. Furthermore, the NMP route avoids complex hydrolysis and evaporation steps required by older methods, streamlining the workflow and improving overall yield.
Q: How does this patented method ensure high purity for the final intermediate?
A: The process utilizes specific crystallization conditions, such as adjusting pH to 1.0–2.0 in isopropanol at low temperatures (0°C), which effectively precipitates the target hydrochloride salt while leaving impurities in the mother liquor. The patent reports gas chromatography (GC) purity levels exceeding 99%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for scalability. It employs common solvents like methanol and xylene, avoids exotic catalysts, and generates minimal wastewater. The operational simplicity and high yields make it highly favorable for commercial-scale manufacturing of Azelastine intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-methylhexahydroazepin-4-one Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Azelastine depends on the unwavering quality and availability of key intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of N-methylhexahydroazepin-4-one hydrochloride meets the highest industry standards. Our commitment to process excellence means we can consistently deliver the high-purity intermediates necessary for your downstream synthesis, minimizing your risk of batch failures.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By leveraging our expertise in this specific NMP-based route, we can provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced synthesis capabilities can become a strategic asset to your organization, driving both quality and efficiency in your pharmaceutical manufacturing operations.
