Scalable Manufacturing of Methyl 3-Amino-2-Fluorobenzoate via Novel Sulfonation-Nitration Strategy
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable pathways for complex fluorinated intermediates, particularly those serving as building blocks for novel anticancer agents and advanced pesticides. Patent CN115850103B, published in April 2023, introduces a groundbreaking preparation method for methyl 3-amino-2-fluorobenzoate, a critical molecular scaffold in modern medicinal chemistry. This technology addresses long-standing challenges regarding cost efficiency and environmental compliance by utilizing a clever sequence of sulfonation, nitration, and catalytic oxidation. For R&D directors and procurement specialists, this patent represents a significant shift away from legacy routes that rely on hazardous reagents and expensive starting materials. The disclosed methodology not only promises substantial cost reduction in API manufacturing but also enhances the overall safety profile of the production line, making it an ideal candidate for a reliable pharmaceutical intermediate supplier looking to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-amino-2-fluorobenzoic acid derivatives has been plagued by significant economic and technical hurdles that hinder large-scale adoption. Prior art, such as the methods described in WO2011059610A1, often relies on 2-fluoro-3-nitrotoluene as a primary raw material, which is inherently expensive and subject to volatile market pricing. Furthermore, these traditional oxidative processes frequently employ chromium trioxide, a hexavalent chromium compound known for its extreme toxicity and severe environmental impact, necessitating costly waste treatment protocols. Another existing approach, disclosed in CN111320548A, attempts to use 2,6-dichlorobenzoic acid but suffers from poor fluorination selectivity, leading to the formation of difficult-to-separate isomers and difluorinated impurities. These inefficiencies result in low overall yields and complicate the purification process, rendering such methods economically unviable for commercial scale-up of complex fluorinated compounds.
The Novel Approach
In stark contrast, the novel approach detailed in CN115850103B leverages o-fluorotoluene, a commodity chemical with excellent availability and a much lower cost basis, as the foundational starting material. The innovation lies in the strategic use of a sulfonic acid group as a temporary blocking and directing agent. By first sulfonating the o-fluorotoluene, the process effectively controls the regiochemistry of the subsequent nitration step, ensuring the nitro group is installed precisely at the 5-position relative to the sulfonic acid group. This precision eliminates the formation of unwanted isomers that plague other synthetic routes. Following nitration, the sulfonic acid group is cleanly removed via hydrolysis, revealing the desired 3-amino-2-fluorotoluene skeleton with high purity. This logical, step-wise construction allows for high-purity agrochemical intermediate production without the need for complex chromatographic separations at every stage.
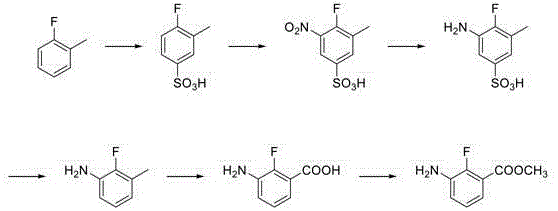
Mechanistic Insights into Regioselective Sulfonation and NHPI-Catalyzed Oxidation
The core chemical ingenuity of this patent resides in the manipulation of electronic effects on the aromatic ring to achieve perfect substitution patterns. In the initial stages, the sulfonation reaction is not merely a functionalization but a strategic maneuver; the bulky sulfonic acid group occupies the para-position relative to the methyl group (or ortho to fluorine depending on conditions), effectively blocking electrophilic attack at that site. When the nitrating agent is introduced, the incoming nitro group is forced into the only available activated position, which corresponds to the meta-position relative to the methyl group and ortho to the fluorine atom in the final desulfonated product. This "block-and-direct" strategy ensures that the subsequent reduction and hydrolysis steps yield 3-amino-2-fluorotoluene with exceptional regio-purity, drastically simplifying the impurity profile for downstream processing.
Furthermore, the oxidation of the methyl group to the carboxylic acid represents a significant advancement in green chemistry principles applied to fine chemical synthesis. Instead of stoichiometric oxidants like permanganate or chromic acid, this process utilizes molecular oxygen as the terminal oxidant, catalyzed by N-hydroxyphthalimide (NHPI). The NHPI catalyst generates phthalimide-N-oxyl (PINO) radicals under thermal conditions, which abstract a hydrogen atom from the benzylic methyl group. This radical mechanism is highly selective for the benzylic position and operates efficiently in acetic acid solvent. For R&D teams, understanding this radical chain mechanism is crucial for optimizing oxygen pressure and temperature to maximize turnover frequency while minimizing over-oxidation or ring degradation, thereby ensuring stringent purity specifications are met consistently.
How to Synthesize Methyl 3-Amino-2-Fluorobenzoate Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly during the telescoped sulfonation-nitration sequence and the high-pressure oxidation step. The patent outlines a streamlined workflow where intermediate isolation is minimized to enhance throughput. For instance, the sulfonation product can be directly subjected to nitration without extensive workup, and the reduction product can proceed straight to hydrolysis. This operational continuity reduces solvent consumption and handling time. To achieve the best results, operators should strictly adhere to the specified temperature gradients, especially during the exothermic nitration phase, to prevent runaway reactions. The detailed standardized synthesis steps, including precise molar ratios and catalyst loading percentages, are provided in the technical guide below to ensure reproducibility across different manufacturing sites.
- Perform sulfonation on o-fluorotoluene followed by nitration to obtain 3-methyl-4-fluoro-5-nitrobenzenesulfonic acid.
- Execute catalytic hydrogenation reduction and subsequent acid hydrolysis to yield 3-amino-2-fluorotoluene.
- Oxidize the toluene derivative using an NHPI catalyst system and finalize with methanol esterification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel manufacturing route offers compelling economic and logistical benefits that extend beyond simple raw material costs. The shift from specialized, high-cost starting materials to commodity-grade o-fluorotoluene fundamentally alters the cost structure of the final intermediate, providing a buffer against market volatility. Additionally, the elimination of heavy metal oxidants like chromium trioxide significantly reduces the regulatory burden and waste disposal costs associated with hazardous effluent treatment. This aligns perfectly with global trends towards sustainable manufacturing, potentially accelerating regulatory approvals for the final drug substance. The robustness of the catalytic systems employed, such as the recoverable Pd/C in the reduction step and the organic NHPI in oxidation, further contributes to long-term operational stability.
- Cost Reduction in Manufacturing: The replacement of expensive precursors with o-fluorotoluene creates a substantial baseline cost advantage that compounds through the entire synthesis. By avoiding the use of stoichiometric chromium reagents, the process eliminates the need for expensive heavy metal scavenging resins and specialized waste containment infrastructure. The ability to telescope multiple reaction steps without isolating intermediates significantly reduces solvent usage and labor hours, driving down the variable cost per kilogram. These cumulative efficiencies result in a highly competitive price point for the final ester, allowing downstream partners to optimize their own bill of materials.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like o-fluorotoluene and sulfuric acid mitigates the risk of supply disruptions often associated with niche fluorinated building blocks. The simplified purification requirements, driven by the high regioselectivity of the sulfonation strategy, mean that production batches are less likely to fail quality control due to isomeric impurities. This consistency ensures a steady flow of high-purity intermediates, reducing lead time for high-purity intermediates and preventing bottlenecks in the client's API synthesis schedule. The use of standard equipment for hydrogenation and oxidation also means the process can be easily replicated across multiple CDMO facilities if capacity expansion is required.
- Scalability and Environmental Compliance: The process is explicitly designed with industrial scale-up in mind, utilizing reaction conditions that are manageable in standard stainless steel reactors. The switch to aerobic oxidation using oxygen gas generates water as the primary byproduct, drastically reducing the E-factor (environmental factor) of the process compared to traditional methods. This green profile facilitates easier permitting and compliance with increasingly strict environmental regulations in major chemical manufacturing hubs. The high yields reported in the patent examples indicate that material throughput is maximized, ensuring that the commercial scale-up of complex fluorinated compounds remains economically viable even at multi-ton scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from a close analysis of the patent's background section, which highlights specific pain points in prior art, and the detailed examples which demonstrate the method's robustness. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific supply chain needs. The answers reflect the practical realities of scaling this chemistry from the laboratory bench to commercial production.
Q: What are the primary advantages of this synthesis route over traditional methods?
A: This method utilizes inexpensive o-fluorotoluene as a starting material, avoiding costly precursors like 2-fluoro-3-nitrotoluene. Furthermore, it eliminates the use of toxic chromium trioxide oxidants, replacing them with a greener NHPI-catalyzed aerobic oxidation system.
Q: How does the process ensure high regioselectivity during nitration?
A: The process employs a strategic sulfonation step that acts as a blocking group. This directs the subsequent nitration to the specific meta-position relative to the methyl group, ensuring the correct substitution pattern before the sulfonic acid group is removed via hydrolysis.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the suitability for industrial preparation. Key factors include the ability to telescope steps (using crude products directly in subsequent reactions), mild reaction conditions, and the use of robust, recoverable catalysts like Pd/C and NHPI.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 3-Amino-2-Fluorobenzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel anticancer drugs and pesticides depends heavily on the reliability and quality of key intermediates like methyl 3-amino-2-fluorobenzoate. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific catalytic hydrogenation and high-pressure oxidation steps required by this patent, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your overall production economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a stable, cost-effective supply chain for your critical fluorinated building blocks.
