Advanced Synthesis of L-Menthyl 3-Hydroxybutyrate: A Cost-Effective Route for High-Purity Cooling Agents
The global demand for high-performance cooling agents in the flavors and fragrances industry has driven significant innovation in synthetic methodologies, particularly for compounds like L-menthyl 3-hydroxybutyrate. Patent CN101423474A introduces a robust, two-step synthetic strategy that addresses the longstanding challenges of yield optimization and operational simplicity associated with this valuable molecule. Unlike traditional approaches that rely on corrosive acidic catalysts or hazardous ketene intermediates, this novel process utilizes a mild base-catalyzed transesterification followed by a controlled borohydride reduction. This technical breakthrough not only enhances the stereochemical integrity of the L-menthol moiety but also streamlines the production workflow, making it an ideal candidate for reliable cooling agent supplier networks seeking scalable solutions. The methodology leverages common industrial solvents such as toluene and methanol, ensuring that the transition from laboratory scale to commercial production is seamless and economically viable for manufacturers worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-hydroxybutyric acid L-menthyl ester has been plagued by inefficient routes that compromise both economic viability and product quality. One conventional pathway involves the reaction of methyl acetoacetate with L-menthol under acidic catalysis, a process that often necessitates rigorous temperature control to prevent the racemization of the chiral center, which is critical for the cooling sensation profile. Another established route utilizes the addition reaction of L-menthol with dimeric ketenes; however, ketenes are notoriously unstable, hazardous to handle, and expensive to source, creating significant supply chain bottlenecks. These traditional methods frequently suffer from complicated post-reaction workups, requiring extensive neutralization and purification steps to remove residual acids or unreacted hazardous intermediates. Furthermore, the yields in these older processes are often inconsistent, leading to higher waste generation and increased disposal costs for environmental compliance teams. The reliance on harsh reaction conditions also limits the choice of equipment materials, necessitating specialized corrosion-resistant reactors that drive up capital expenditure for production facilities.
The Novel Approach
In stark contrast, the methodology disclosed in the patent employs a transesterification reaction driven by inexpensive inorganic bases such as sodium carbonate, potassium carbonate, or calcium carbonate. This shift from acidic to basic catalysis fundamentally alters the reaction landscape, allowing for milder conditions that preserve the delicate stereochemistry of the L-menthol starting material. The use of commodity solvents like toluene or cyclohexane facilitates easy solvent recovery and recycling, further enhancing the green chemistry profile of the process. By avoiding hazardous ketene dimers, the new approach significantly reduces safety risks associated with raw material handling and storage, thereby lowering insurance and safety compliance overheads. The subsequent reduction step utilizes potassium borohydride or sodium borohydride at low temperatures (10-20°C), ensuring high selectivity for the ketone group without compromising the ester linkage. This strategic combination of mild transesterification and selective reduction results in a streamlined process that delivers high-purity products with minimal byproduct formation, setting a new standard for cost reduction in flavor and fragrance manufacturing.
Mechanistic Insights into Base-Catalyzed Transesterification and Reduction
The core of this synthetic innovation lies in the efficient base-catalyzed transesterification mechanism, where the carbonate ion acts as a mild yet effective promoter for the exchange of the ethoxy group in ethyl acetoacetate with the hydroxyl group of L-menthol. The reaction proceeds through a tetrahedral intermediate, facilitated by the removal of ethanol (or methanol) via distillation, which drives the equilibrium towards the formation of L-menthyl acetoacetate according to Le Chatelier's principle. The choice of sodium carbonate is particularly advantageous as it is non-nucleophilic enough to avoid unwanted side reactions such as Claisen condensation, which could occur with stronger bases like alkoxides. This mechanistic precision ensures that the beta-keto ester intermediate is formed with high fidelity, maintaining the structural integrity required for the subsequent reduction step. The use of aromatic solvents like toluene provides an optimal boiling point range that allows for the continuous removal of the alcohol byproduct while keeping the reaction mixture homogeneous and thermally stable.
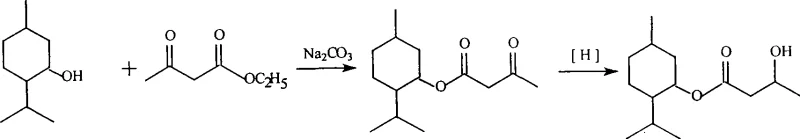
Following the formation of the intermediate, the reduction mechanism employs hydride transfer from borohydride species to the carbonyl carbon of the beta-keto group. Conducting this reaction at 10-20°C is critical for kinetic control, preventing the over-reduction of the ester functionality which would lead to diol impurities. The protic solvent, typically methanol or ethanol, serves a dual purpose: it solubilizes the borohydride salt and participates in the protonation of the resulting alkoxide intermediate to yield the final hydroxyl group. This low-temperature protocol effectively suppresses potential epimerization at the alpha-carbon, ensuring that the final cooling agent retains the specific spatial configuration necessary for its sensory activity. The result is a highly pure product profile with a narrow impurity spectrum, which is essential for meeting the stringent regulatory requirements of the food and pharmaceutical industries. By understanding these mechanistic nuances, R&D teams can optimize reaction parameters to maximize yield and minimize waste, achieving commercial scale-up of complex flavor molecules with confidence.
How to Synthesize L-Menthyl 3-Hydroxybutyrate Efficiently
The synthesis of this high-value cooling agent is designed for operational simplicity, allowing chemical engineers to implement the process with standard reactor setups found in most fine chemical plants. The procedure begins with the charging of ethyl acetoacetate and L-menthol into a reactor equipped with a distillation column, followed by the addition of the carbonate catalyst and toluene solvent. Heating the mixture to reflux facilitates the transesterification, with the evolved ethanol being continuously removed to drive the reaction to completion within 4 to 5 hours. Once the intermediate is isolated via vacuum distillation, it is dissolved in methanol and cooled to the specified low-temperature range before the careful addition of the reducing agent. Detailed standardized synthesis steps see the guide below.
- Perform transesterification of ethyl acetoacetate and L-menthol using sodium carbonate catalyst in toluene at reflux for 4-5 hours to obtain L-menthyl acetoacetate.
- Isolate the intermediate ester by washing to neutrality and distilling off the solvent under reduced pressure.
- Reduce the L-menthyl acetoacetate intermediate using potassium borohydride in methanol at 10-20°C to yield the final cooling agent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthetic route offers transformative benefits that extend far beyond simple yield improvements. The substitution of expensive and hazardous raw materials with widely available commodity chemicals creates a more resilient supply chain that is less susceptible to market volatility and sourcing disruptions. By eliminating the need for specialized corrosion-resistant equipment required for acidic processes, manufacturers can utilize existing stainless steel infrastructure, significantly reducing capital investment barriers for new production lines. The mild reaction conditions also translate to lower energy consumption, as the process does not require extreme heating or cryogenic cooling, aligning with corporate sustainability goals and reducing utility costs. Furthermore, the simplified workup procedures reduce the volume of aqueous waste generated, lowering the burden on wastewater treatment facilities and minimizing environmental compliance risks. These factors collectively contribute to a more robust and cost-effective manufacturing model that enhances the overall competitiveness of the final product in the global marketplace.
- Cost Reduction in Manufacturing: The elimination of expensive ketene dimers and strong acidic catalysts drastically lowers the raw material bill of materials, while the use of recyclable solvents like toluene further optimizes operational expenditures. The high conversion rates achieved in both steps minimize the loss of valuable chiral starting materials, ensuring that every kilogram of L-menthol is utilized efficiently. Additionally, the reduced need for complex purification steps lowers the consumption of auxiliary chemicals and processing time, leading to substantial cost savings per batch. This economic efficiency allows suppliers to offer competitive pricing without compromising on the quality or purity of the cooling agent delivered to customers.
- Enhanced Supply Chain Reliability: Sourcing strategies are significantly strengthened by the reliance on bulk chemicals such as sodium carbonate and ethyl acetoacetate, which are produced globally in massive quantities with stable pricing. The removal of hazardous ketene intermediates from the supply chain eliminates the logistical complexities and regulatory hurdles associated with transporting dangerous goods across borders. This stability ensures consistent production schedules and reliable delivery timelines, which are critical for maintaining inventory levels in the fast-moving consumer goods sector. Manufacturers can confidently plan long-term production runs knowing that the availability of key reagents is secure and unaffected by niche market fluctuations.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram-scale laboratory experiments to multi-ton commercial batches without loss of efficiency. The mild nature of the reagents reduces the risk of thermal runaway incidents, enhancing plant safety and simplifying the permitting process for capacity expansion. Moreover, the generation of benign byproducts and the ability to recover solvents align with modern green chemistry principles, facilitating easier regulatory approval in regions with strict environmental standards. This scalability ensures that the supply can grow in tandem with market demand, supporting the commercial expansion of products containing this advanced cooling agent.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of L-menthyl 3-hydroxybutyrate, based on the specific advantages outlined in the patent literature. Understanding these details helps stakeholders make informed decisions about integrating this material into their product formulations and supply chains. The answers reflect the consensus on best practices for handling and synthesizing this compound to ensure maximum efficacy and safety.
Q: Why is the base-catalyzed route superior to traditional acidic methods for this cooling agent?
A: Traditional acidic routes often require harsh conditions that can lead to racemization of the chiral menthol center or degradation of the ester linkage. The patented base-catalyzed method using carbonates operates under mild conditions, preserving optical purity and simplifying downstream purification.
Q: What are the key cost drivers eliminated in this synthetic pathway?
A: This pathway eliminates the need for expensive and hazardous ketene dimers or strong acidic catalysts. By utilizing commodity chemicals like ethyl acetoacetate and sodium carbonate, the raw material costs are drastically reduced while maintaining high yields above 95%.
Q: How does the low-temperature reduction step impact product quality?
A: Conducting the borohydride reduction at 10-20°C ensures high chemoselectivity, reducing the ketone group without affecting the ester moiety. This precise temperature control minimizes side reactions and byproduct formation, resulting in a high-purity final product suitable for food and pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Menthyl 3-Hydroxybutyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN101423474A to meet the evolving needs of the flavors and fragrances industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering high-purity L-menthyl 3-hydroxybutyrate that adheres to stringent purity specifications, supported by our rigorous QC labs that perform comprehensive testing on every batch. Our facility is equipped to handle the specific solvent systems and temperature controls required for this base-catalyzed process, guaranteeing a product that delivers the pure cool flavor profile your customers expect.
We invite you to collaborate with us to leverage these technological advancements for your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your supply chain efficiency and product quality.
