Advanced Synthesis of Boronic Acid Adenine Derivatives for Commercial Scale-Up
Advanced Synthesis of Boronic Acid Adenine Derivatives for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking novel heterocyclic scaffolds that offer dual functionality and enhanced biological profiles. Patent CN100427488C introduces a significant breakthrough in this domain by disclosing a series of adenine derivatives functionalized with boronic acid groups. These compounds are not merely academic curiosities; they represent a class of high-value intermediates with demonstrated efficacy in inhibiting tumor necrosis factor α (TNF-α) and enhancing cytokinin activity in plants. For R&D directors and procurement specialists alike, understanding the synthesis and potential of these molecules is crucial for developing next-generation anti-inflammatory agents and plant growth regulators. The strategic integration of a boronic acid moiety onto the adenine core opens up avenues for further derivatization via Suzuki-Miyaura cross-coupling, thereby expanding the chemical space available for drug discovery.
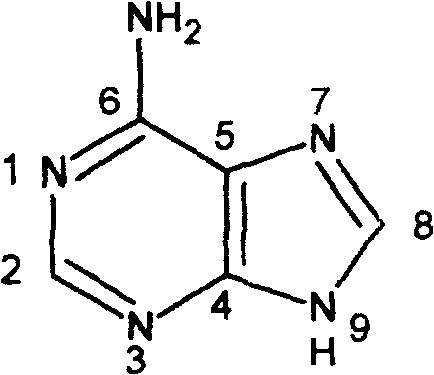
Adenine serves as a fundamental building block in biological systems, forming the backbone of DNA, RNA, and ATP. However, modifying this scaffold to introduce specific pharmacophores has historically been challenging due to issues with regioselectivity and harsh reaction conditions. The traditional reliance on halogenated purines often necessitates the use of toxic reagents and generates significant hazardous waste. In contrast, the methodology outlined in the patent leverages the nucleophilic properties of the adenine nitrogen atoms to directly alkylate with boronic acid-containing benzyl bromides. This shift in synthetic strategy addresses critical pain points in process chemistry, specifically regarding safety and environmental compliance, making it an attractive candidate for reliable pharmaceutical intermediate supplier networks looking to diversify their portfolios with safer, greener alternatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted adenine derivatives has relied heavily on the reaction of 6-chloropurine with various amines or benzyl halides. This conventional pathway is fraught with significant drawbacks that hinder efficient cost reduction in pharmaceutical intermediate manufacturing. Firstly, 6-chloropurine is a potent lachrymator and poses severe health risks to synthetic personnel, requiring expensive containment infrastructure and rigorous safety protocols. Secondly, the reaction often suffers from poor regioselectivity, leading to mixtures of N7 and N9 isomers that are difficult and costly to separate. Furthermore, the disposal of chlorinated byproducts adds a substantial burden to waste management systems. These factors collectively inflate the production cost and extend the lead time for high-purity intermediates, creating bottlenecks in the supply chain for downstream API manufacturers who demand consistent quality and rapid turnaround.
The Novel Approach
The innovative route described in the patent circumvents these issues by utilizing adenine itself as the starting material, reacting it directly with n-bromomethylphenylboronic acid in the presence of a mild base like anhydrous potassium carbonate. This method operates in polar aprotic solvents such as N,N-dimethylformamide (DMF) at temperatures ranging from 100°C to 140°C. By avoiding the use of chlorinated purine precursors, the process drastically reduces the toxicity profile of the synthesis. Moreover, the reaction demonstrates excellent selectivity for the N9 position under these conditions, minimizing the formation of unwanted isomers. For the N6-substituted derivatives, the process employs a Dimroth rearrangement strategy, which allows for the migration of substituents to the exocyclic amine position under controlled thermal conditions. This flexibility enables the production of both mono- and di-substituted derivatives with high structural fidelity, ensuring that the final product meets the stringent purity specifications required for biological applications.
Mechanistic Insights into Nucleophilic Substitution and Dimroth Rearrangement
The core of this synthesis lies in the nucleophilic attack of the adenine nitrogen atoms on the benzylic carbon of the bromomethylphenylboronic acid. In the first stage, the N9 nitrogen, being the most nucleophilic site in the imidazole ring under basic conditions, attacks the electrophilic carbon, displacing the bromide ion. This SN2-type mechanism is facilitated by the polar DMF solvent, which stabilizes the transition state and solvates the potassium cations. The presence of the boronic acid group on the benzyl ring is chemically robust under these basic alkylation conditions, preserving the functionality for future cross-coupling reactions. This mechanistic pathway is distinct from acid-catalyzed alkylations, which might protonate the ring nitrogens and deactivate the nucleophile. Understanding this nuance is vital for process chemists aiming to optimize reaction kinetics and maximize yield without compromising the integrity of the sensitive boronic acid handle.
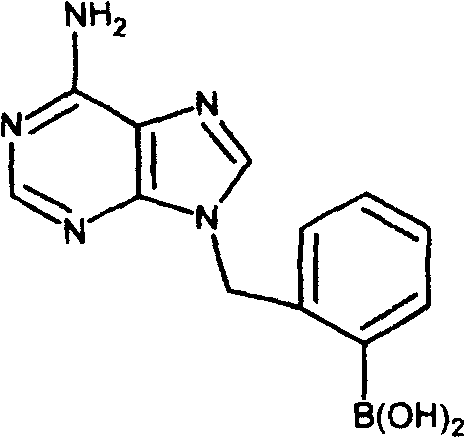
For the synthesis of N6-substituted derivatives, the mechanism involves a more complex sequence often referred to as the Dimroth rearrangement. Initially, alkylation may occur at the N1 or N3 position, or an intermediate N-benzyl species is formed which then undergoes ring opening and reclosure. Specifically, in the preparation of N9-benzyl-N6-(boronyl)benzyl adenine, the reaction involves treating 9-benzyladenine with the boronic acid bromide, followed by an acid-base treatment cycle. The intermediate is dissolved in methanolic HCl and then treated with ammonia at elevated temperatures (90°C). This sequence facilitates the migration of the benzyl group from the ring nitrogen to the exocyclic amine at the C6 position. This rearrangement is driven by thermodynamic stability, as the exocyclic amine substitution often yields a more stable conjugated system. Mastery of this rearrangement allows for the precise installation of substituents at the N6 position, which is critical for modulating the cytokinin activity of the final molecule.
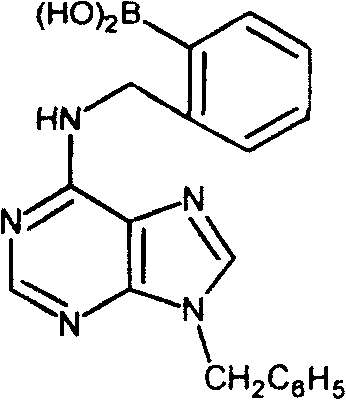
How to Synthesize N9-(Boronyl)benzyl Adenine Efficiently
The practical execution of this synthesis requires careful attention to moisture control and temperature regulation to ensure optimal conversion. The process begins with the preparation of a homogeneous suspension of the reactants, followed by a controlled addition of the alkylating agent to manage exotherms and prevent side reactions. Detailed operational parameters, including specific stoichiometric ratios and work-up procedures, are essential for reproducing the reported yields of 35% to 46%. For laboratories and pilot plants aiming to implement this technology, adhering to the standardized protocol ensures consistency in the impurity profile, which is a key metric for regulatory approval. The detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results effectively.
- Suspend adenine and anhydrous potassium carbonate in fresh DMF and heat to 100-140°C under stirring.
- Slowly add a DMF solution of n-bromomethylphenylboronic acid over 60 minutes and reflux for 12-16 hours.
- Filter the hot suspension, evaporate the solvent under reduced pressure, and purify the residue via chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers compelling advantages for supply chain resilience and cost management. The primary raw materials, adenine and bromomethylphenylboronic acids, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. This abundance ensures that procurement managers can negotiate favorable terms and maintain a steady flow of materials even during market fluctuations. Furthermore, the elimination of highly toxic chloropurines simplifies the safety infrastructure required for production, potentially lowering insurance premiums and operational overheads associated with hazardous material handling. The simplified purification process, often achievable through simple filtration or recrystallization rather than complex column chromatography on a large scale, translates directly into reduced processing time and lower solvent consumption.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of the synthetic route and the use of inexpensive, readily available starting materials. By avoiding the need for expensive protecting groups or specialized catalysts often required in traditional purine chemistry, the overall cost of goods sold (COGS) is significantly optimized. Additionally, the higher selectivity of the reaction reduces the burden on downstream purification units, leading to substantial savings in solvent usage and waste disposal costs. The ability to produce high-purity intermediates with fewer unit operations enhances the overall throughput of the manufacturing facility, allowing for better asset utilization and improved margin potential for the final API or agrochemical product.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the chemical transformation, which tolerates minor variations in reaction conditions without catastrophic failure. The use of stable boronic acid intermediates means that the supply chain is not vulnerable to the degradation issues often seen with more labile functional groups. This stability allows for longer storage times and easier logistics, ensuring that inventory levels can be maintained efficiently. For partners relying on a reliable agrochemical intermediate supplier, this reliability is paramount, as it minimizes the risk of production delays caused by raw material shortages or quality failures, thereby securing the timeline for product launches and market entry.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively in batch reactors with standard heating and stirring capabilities. The absence of heavy metal catalysts eliminates the need for rigorous metal scavenging steps, which are often a bottleneck in GMP manufacturing. From an environmental standpoint, the reduction in toxic waste generation aligns with increasingly stringent global regulations on chemical emissions. This green chemistry profile not only mitigates regulatory risk but also enhances the corporate sustainability image of the manufacturing entity. The ease of scaling from gram to kilogram quantities ensures that the transition from R&D to commercial production is seamless, supporting the rapid commercialization of new drug candidates or crop protection agents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these adenine derivatives. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating these compounds into their existing development pipelines. The answers cover aspects ranging from biological efficacy to process scalability, offering a comprehensive overview for technical and business leaders alike.
Q: What are the primary biological activities of these boronic acid adenine derivatives?
A: According to patent CN100427488C, these derivatives exhibit significant inhibition of pro-inflammatory factors like TNF-α (up to 36.6% inhibition) and demonstrate superior cytokinin activity compared to natural 6-benzyl adenine, promoting plant chlorophyll content.
Q: How does this synthesis method improve upon conventional adenine alkylation?
A: Traditional methods often rely on toxic 6-chloropurine and complex benzylamine reactions. This novel approach utilizes readily available bromomethylphenylboronic acid and simple adenine, reducing toxicity and simplifying the purification process while maintaining high regioselectivity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses standard solvents like DMF and common bases like potassium carbonate. The reaction conditions (100-140°C) are easily manageable in standard stainless steel reactors, making it highly scalable for commercial manufacturing without requiring exotic catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adenine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN100427488C for both pharmaceutical and agricultural applications. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves smoothly from the bench to the market. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying the stringent purity specifications required for bioactive intermediates. We understand that the consistency of the boronic acid functionality is critical for downstream coupling reactions, and our quality assurance protocols are designed to guarantee the integrity of every batch we produce.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next generation of products. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments for these high-value adenine derivatives. By partnering with us, you gain access to a supply chain that prioritizes innovation, safety, and efficiency, positioning your organization at the forefront of the fine chemical industry.
