Advanced Synthesis of Methylal Penem Intermediates for Commercial Scale-Up
Advanced Synthesis of Methylal Penem Intermediates for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic routes for critical beta-lactamase inhibitor intermediates, and patent CN102020659B presents a significant breakthrough in this domain. This intellectual property details a novel preparation method for 5,6-dihydro-8H-imidazo[2,1-c][1,4]oxazine-2-carbaldehyde, a pivotal building block for methylal penem antibiotics. Unlike traditional pathways that struggle with isomeric impurities and low yields, this innovation leverages a streamlined Grignard exchange strategy to achieve superior purity and operational efficiency. For R&D directors and procurement specialists, understanding this technology is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials for next-generation antimicrobial therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key penem intermediates has been plagued by significant chemical inefficiencies that hinder industrial viability. Prior art, such as the methods disclosed in US20040132708 and WO2006130588, typically involves multi-step sequences starting from 3-morpholinone, proceeding through methylation and ammonolysis, and culminating in a condensation with 2-bromo-3-hydroxyacrolein. A critical flaw in these legacy processes is the unavoidable generation of 3-position aldehyde isomers alongside the desired 2-position product. This structural similarity necessitates rigorous and costly purification steps, often relying on silica gel column chromatography, which is notoriously difficult to scale and results in substantial material loss. Consequently, the overall yield remains low, and the process cost is prohibitively high for large-scale commercial application.
The Novel Approach
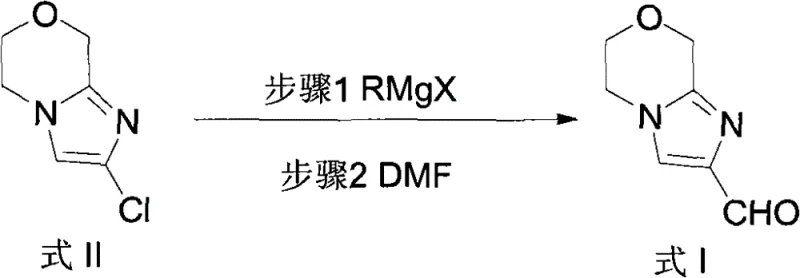
In stark contrast, the methodology outlined in CN102020659B introduces a rational design that circumvents these historical bottlenecks through a direct functionalization strategy. By utilizing 2-chloro-6,8-dihydro-5H-imidazo[2,1-c]-[1,4]oxazine as a precursor, the process employs a halogen-magnesium exchange followed by formylation with dimethylformamide (DMF). This approach ensures regioselective formation of the aldehyde group exclusively at the 2-position, effectively eliminating the formation of unwanted 3-position isomers. The result is a crude product of such high purity that complex chromatographic separation is rendered unnecessary, allowing for simple crystallization techniques to isolate the target compound. This fundamental shift in synthetic logic drastically simplifies the workflow and enhances the economic feasibility of producing high-purity beta-lactamase inhibitor intermediates.
Mechanistic Insights into Grignard Exchange and Cyclization
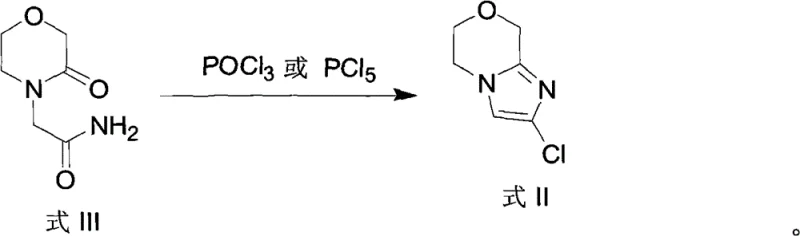
The core of this technological advancement lies in the precise execution of organometallic transformations and heterocyclic cyclization. The synthesis begins with the construction of the imidazo-oxazine scaffold, where 2-(3-oxomorpholino)acetamide undergoes dehydration and cyclization using phosphorus oxychloride (POCl3) or phosphorus pentachloride (PCl5). This step is critical for establishing the fused ring system required for biological activity. Following this, the chlorine atom at the 2-position serves as a handle for the subsequent Grignard reaction. When treated with a lower alkyl Grignard reagent, such as ethylmagnesium bromide or isopropylmagnesium bromide, a halogen-metal exchange occurs, generating a reactive organomagnesium species in situ.
This intermediate is then quenched with DMF, which acts as the formyl source, to yield the target aldehyde. The beauty of this mechanism is its specificity; the electron-deficient nature of the heterocycle and the steric environment direct the reaction cleanly to the desired position. Furthermore, the initial construction of the acetamide precursor from 3-morpholinone and haloacetamide under basic conditions provides a stable and accessible entry point into the synthesis. The entire sequence avoids harsh conditions that might degrade the sensitive beta-lactam-like scaffolds, ensuring that the integrity of the molecular architecture is maintained throughout the process.
From an impurity control perspective, this route offers distinct advantages by minimizing side reactions. Traditional aldol-type condensations often lead to polymeric byproducts and regioisomers that are chemically similar to the product, making them difficult to remove. In this novel pathway, the primary byproducts are magnesium salts and unreacted starting materials, which are easily removed during the aqueous workup and recrystallization stages. The absence of isomeric impurities means that the final product meets stringent purity specifications required for pharmaceutical applications without the need for extensive downstream processing. This level of control is paramount for ensuring the safety and efficacy of the final antibiotic formulation.
How to Synthesize 5,6-dihydro-8H-imidazo[2,1-c][1,4]oxazine-2-carbaldehyde Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature and stoichiometry, to maximize yield and purity. The process is divided into three logical stages: the preparation of the acetamide precursor, the cyclization to the chloro-intermediate, and the final Grignard formylation. Each step utilizes commonly available industrial reagents, facilitating easy sourcing and handling. The detailed standardized synthesis steps below outline the optimal conditions derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate this high-efficiency route in a pilot or production setting.
- React 3-morpholinone with haloacetamide in the presence of a base to form 2-(3-oxomorpholino)acetamide.
- Cyclize the acetamide derivative using POCl3 or PCl5 to generate 2-chloro-6,8-dihydro-5H-imidazo[2,1-c]-[1,4]oxazine.
- Perform halogen-magnesium exchange with RMgX followed by formylation with DMF to yield the target aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route represents a strategic opportunity to optimize costs and secure supply continuity. The elimination of silica gel column chromatography is perhaps the most significant economic driver, as this technique is a major bottleneck in terms of both solvent consumption and processing time in traditional fine chemical manufacturing. By replacing this with simple crystallization, the process reduces solvent waste and energy usage, leading to substantial cost savings in the overall production budget. Additionally, the use of readily available starting materials like 3-morpholinone and commercial Grignard reagents ensures that the supply chain is robust and less susceptible to disruptions caused by specialty reagent shortages.
- Cost Reduction in Manufacturing: The streamlined nature of this process directly translates to lower manufacturing costs by removing expensive purification steps and reducing cycle times. The avoidance of isomeric byproducts means that yield losses associated with separating difficult mixtures are virtually eliminated, improving the overall mass balance of the production line. Furthermore, the reagents employed, such as POCl3 and DMF, are commodity chemicals with stable pricing, shielding the project from the volatility often seen with exotic catalysts. This economic efficiency makes the production of high-purity pharmaceutical intermediates more sustainable and competitive in the global market.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the simplicity and robustness of the chemical transformations involved. Since the method does not rely on sensitive transition metal catalysts that require strict inert atmospheres or specialized handling, it can be executed in standard multipurpose reactors found in most chemical plants. This flexibility allows for faster technology transfer and quicker ramp-up times, ensuring that delivery schedules for critical API intermediates are met consistently. The ability to source raw materials from multiple global suppliers further mitigates the risk of single-source dependency, providing a secure foundation for long-term production planning.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is facilitated by the absence of complex separation technologies that often fail to translate linearly to larger vessels. The exothermic nature of the Grignard reaction is manageable with standard cooling protocols, and the workup procedures involve standard phase separations that are easily automated. From an environmental standpoint, the reduction in solvent volume and the elimination of silica waste contribute to a greener manufacturing profile, aligning with increasingly strict regulatory standards for waste disposal and emissions in the chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, offering clarity on how this technology compares to existing solutions. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this route for their specific supply chain needs.
Q: How does this method avoid the formation of 3-position aldehyde isomers?
A: The novel route utilizes a specific halogen-magnesium exchange on the 2-chloro intermediate, which directs formylation exclusively to the 2-position, eliminating the isomeric mixtures common in older condensation methods.
Q: What are the advantages of using Grignard reagents in this synthesis?
A: Using Grignard reagents like ethylmagnesium bromide allows for mild reaction conditions and high conversion rates without the need for complex catalysts, simplifying the purification process significantly.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses commercially available raw materials and avoids difficult separation techniques like silica gel column chromatography, making it highly scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,6-dihydro-8H-imidazo[2,1-c][1,4]oxazine-2-carbaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antibiotics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 5,6-dihydro-8H-imidazo[2,1-c][1,4]oxazine-2-carbaldehyde meets the exacting standards required by top-tier pharmaceutical companies worldwide.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to drive innovation and efficiency in your antibiotic supply chain.
