Advanced Synthesis of Flumioxazin Intermediates: A Technical Breakthrough for Global Agrochemical Supply Chains
The global demand for high-efficiency herbicides like Flumioxazin continues to drive innovation in the synthesis of key agrochemical intermediates. Patent CN108976129B introduces a transformative preparation method for 2-(5-fluoro-2,4-dinitrophenoxy) acetate, a critical precursor in the production of next-generation non-selective herbicides. This technology addresses long-standing challenges in the fine chemical industry, specifically targeting the high costs and purity issues associated with traditional synthetic routes. By leveraging a streamlined two-step process involving alkaline hydrolysis followed by a catalyzed nucleophilic substitution, this method offers a robust pathway for reliable agrochemical intermediate supplier networks seeking to optimize their manufacturing portfolios. The strategic implementation of specific additives and dual-catalyst systems ensures that the final product meets stringent purity specifications required for downstream pharmaceutical and agricultural applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(5-fluoro-2,4-dinitrophenoxy) acetate has been plagued by significant economic and technical barriers that hinder efficient commercial scale-up of complex agrochemical intermediates. Early methodologies, such as those disclosed in US4640707, relied heavily on m-fluorophenol as a starting material. While chemically feasible, m-fluorophenol is prohibitively expensive and subject to volatile market pricing, making it unsuitable for cost-sensitive bulk manufacturing. Furthermore, alternative routes described in US4803270 utilized 1,5-dichloro-2,4-dinitrobenzene, necessitating a subsequent fluorination step. This fluorination requires rigorous anhydrous conditions and high temperatures, which often lead to the cleavage of ether bonds and a drastic reduction in overall yield. Other approaches involving ethyl glycolate, as seen in Japanese Patent 7-48321, suffer from uncontrollable side reactions where both fluorine atoms are substituted, generating impurities that are notoriously difficult to separate from the target molecule.
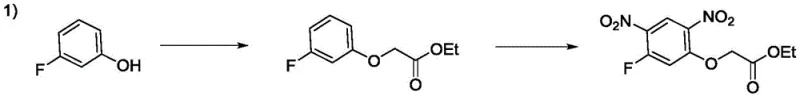
These conventional pathways not only inflate the cost reduction in herbicide manufacturing but also introduce severe supply chain risks due to the reliance on unstable or hazardous reagents like methyl bromoacetate. The accumulation of viscous by-products during acidification further complicates the isolation process, leading to extended processing times and increased waste generation. For procurement managers, these inefficiencies translate into unpredictable lead times and higher total cost of ownership. The inability to consistently achieve high purity without extensive chromatographic purification limits the applicability of these intermediates in high-value crop protection formulations, creating a bottleneck for companies aiming to reduce lead time for high-purity agrochemical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN108976129B presents a highly efficient and economically viable alternative that redefines the standard for intermediate synthesis. The core innovation lies in the utilization of 2,4-difluoronitrobenzene derivatives as the primary feedstock, which are significantly more abundant and cost-effective than phenolic precursors. The process initiates with a controlled hydrolysis step under alkaline conditions to selectively generate 5-fluoro-2,4-dinitrophenol. This intermediate is then subjected to a nucleophilic substitution reaction with inexpensive chloroacetates, facilitated by a unique combination of additives and catalysts. The introduction of 1,1,1-trichloroacetate esters as activating agents plays a pivotal role in enhancing reaction selectivity, effectively suppressing the formation of double-substituted by-products that plague earlier techniques.
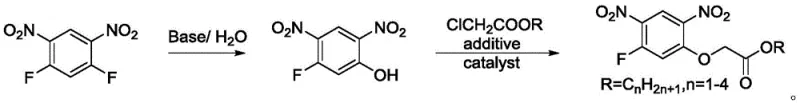
This novel approach simplifies the operational complexity, allowing the reaction to proceed at moderate temperatures between 110-170°C without the need for extreme pressure or specialized anhydrous setups. The resulting crude product exhibits superior crystallinity, enabling straightforward purification via recrystallization with isopropanol and water mixtures. This simplicity directly supports the commercial scale-up of complex polymer additives and agrochemicals by minimizing equipment requirements and energy consumption. By eliminating the need for hazardous fluorination reagents and expensive glycolic acid derivatives, this method establishes a new benchmark for safety and sustainability in fine chemical manufacturing, ensuring a stable supply of high-quality intermediates for the global market.
Mechanistic Insights into Alkaline Hydrolysis and Catalyzed Etherification
The chemical elegance of this synthesis is rooted in the precise control of nucleophilic aromatic substitution mechanisms. In the first stage, the electron-withdrawing nature of the nitro groups on the benzene ring activates the fluorine atoms towards nucleophilic attack by hydroxide ions. The use of a mixed solvent system comprising tetrahydrofuran and water optimizes the solubility of both the organic substrate and the inorganic base, facilitating a homogeneous reaction environment that maximizes conversion rates. Careful temperature control during this hydrolysis phase is critical to prevent over-reaction or degradation of the sensitive nitro functionalities. The subsequent acidification step precisely neutralizes the reaction mixture, causing the phenolic intermediate to precipitate as a solid, which can be easily isolated and dried to high purity levels before proceeding to the next stage.
In the second stage, the mechanism shifts to an etherification process driven by the synergy between a basic catalyst and a phase transfer catalyst. The addition of polyethers or quaternary ammonium salts enhances the nucleophilicity of the phenoxide ion in the organic phase, promoting efficient attack on the chloroacetate electrophile. The trichloroacetate additive functions as a transient activator, potentially forming a reactive intermediate that lowers the activation energy for the substitution. This dual-catalyst system ensures that the reaction proceeds rapidly and selectively, minimizing the residence time required at elevated temperatures. Such mechanistic optimization is crucial for maintaining the integrity of the dinitro structure while achieving the desired ether linkage, ultimately delivering a product with minimal impurity profiles suitable for sensitive downstream applications.
How to Synthesize 2-(5-Fluoro-2,4-Dinitrophenoxy) Acetate Efficiently
Implementing this synthesis route requires adherence to specific operational parameters to maximize yield and safety. The process begins with the preparation of the phenolic intermediate, followed by the etherification step where stoichiometry and catalyst loading are critical. Operators must ensure precise temperature ramping and effective removal of by-product gases to drive the equilibrium forward. The detailed standardized synthesis steps see the guide below for exact molar ratios and workup procedures.
- Hydrolyze 2,4-difluoronitrobenzene derivatives under alkaline conditions (KOH/NaOH) in a THF/water mixture to generate 5-fluoro-2,4-dinitrophenol.
- React the resulting phenol with ethyl chloroacetate in the presence of 1,1,1-trichloroacetate additives and dual catalysts (base + phase transfer agent).
- Maintain reaction temperature between 110-170°C, followed by recrystallization using isopropanol and water to achieve high purity solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain directors, the adoption of this patented methodology offers substantial strategic benefits that extend beyond mere technical feasibility. The shift away from volatile and expensive raw materials like m-fluorophenol stabilizes the cost structure of the final intermediate, allowing for more predictable budgeting and long-term contracting. By utilizing commodity chemicals such as chloroacetates and common inorganic bases, manufacturers can mitigate the risks associated with supply disruptions of specialty reagents. This resilience is vital for maintaining continuous production schedules in the fast-paced agrochemical sector, where downtime can result in significant financial losses and missed market opportunities.
- Cost Reduction in Manufacturing: The elimination of costly starting materials and the simplification of purification processes lead to a dramatic decrease in overall production expenses. By avoiding complex chromatographic separations and reducing solvent consumption through efficient recrystallization, the operational expenditure is significantly lowered. Furthermore, the high atom economy of the substitution reaction minimizes waste disposal costs, contributing to a leaner and more profitable manufacturing model that enhances competitiveness in the global marketplace.
- Enhanced Supply Chain Reliability: Sourcing raw materials that are widely available and commercially stable reduces the vulnerability of the supply chain to geopolitical or logistical shocks. The robustness of the synthesis against minor variations in reaction conditions ensures consistent output quality, reducing the rate of batch rejections. This reliability allows supply chain managers to optimize inventory levels and shorten lead times, ensuring that downstream customers receive their orders promptly and without quality disputes, thereby strengthening business relationships.
- Scalability and Environmental Compliance: The process is inherently scalable, transitioning smoothly from laboratory benchtop to multi-ton reactor systems without requiring fundamental changes in chemistry. The absence of hazardous fluorination steps and the use of less toxic reagents align with increasingly strict environmental regulations. This compliance reduces the regulatory burden and permitting timelines for new production facilities, accelerating time-to-market for new products while demonstrating a commitment to sustainable and responsible chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational requirements.
Q: What are the primary cost advantages of this new synthesis route compared to traditional methods?
A: The novel method avoids expensive raw materials like m-fluorophenol or unstable 5-fluoro-2-nitrophenol. By utilizing readily available fluorinated nitrobenzene derivatives and cheap chloroacetates, it significantly reduces raw material costs and eliminates complex purification steps associated with viscous by-products.
Q: How does the use of trichloroacetate additives impact product purity?
A: The addition of methyl or ethyl 1,1,1-trichloroacetate acts as an activating agent that facilitates the nucleophilic substitution reaction. This minimizes side reactions such as double substitution, leading to a cleaner reaction profile and a final product purity exceeding 98% after simple recrystallization.
Q: Is this process suitable for large-scale industrial production of herbicide intermediates?
A: Yes, the process is designed for scalability. It operates under relatively mild conditions (110-170°C) without requiring extreme anhydrous environments or hazardous fluorination reagents. The solid product precipitates easily upon cooling, simplifying filtration and drying operations for tonnage-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(5-Fluoro-2,4-Dinitrophenoxy) Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of effective crop protection solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of major agrochemical corporations. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2-(5-fluoro-2,4-dinitrophenoxy) acetate meets the exacting standards required for Flumioxazin synthesis. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of nitro-compound chemistry safely and efficiently.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your herbicide manufacturing programs.
