Advanced Synthetic Route for 2-(2,2-difluoroethoxy)-6-trifluoromethylbenzenesulfonyl chloride Manufacturing
Introduction to Patent CN112939818B
The global agrochemical sector continuously demands more efficient and cost-effective pathways for producing critical herbicide intermediates. Patent CN112939818B, published in May 2022, introduces a groundbreaking synthetic method for 2-(2,2-difluoroethoxy)-6-trifluoromethylbenzenesulfonyl chloride, a pivotal building block for sulfonamide herbicides. This technology represents a significant leap forward in process chemistry, addressing long-standing inefficiencies in traditional manufacturing protocols. By streamlining the synthesis into a concise six-step sequence, the patent offers a robust solution that enhances both yield and operational safety. For R&D directors and procurement strategists, understanding this innovation is crucial for optimizing supply chains and reducing the total cost of ownership for high-value agrochemical ingredients.
The core breakthrough lies in the strategic selection of starting materials and the elimination of unnecessary protective group chemistry. Unlike legacy routes that rely on complex protection-deprotection sequences, this novel approach leverages the inherent reactivity of substituted nitro-bromobenzenes. The process utilizes widely available reagents such as N-bromosuccinimide (NBS) or elemental bromine for regioselective halogenation, followed by efficient reduction and diazotization steps. This methodological shift not only simplifies the workflow but also minimizes waste generation, aligning with modern green chemistry principles. As a reliable agrochemical intermediate supplier, recognizing the potential of such patented technologies allows for better forecasting of market availability and pricing stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(2,2-difluoroethoxy)-6-trifluoromethylbenzenesulfonyl chloride has been plagued by inefficient multi-step sequences that drive up costs and complicate scale-up. Traditional literature routes, such as those described in DE10019291 and US6005108, typically commence with m-trifluoromethylphenol as the primary raw material. A major drawback of these methods is the mandatory requirement for hydroxyl protection prior to introducing the sulfur functionality. This necessitates additional reaction steps for protection and subsequent deprotection, which inherently lowers the overall yield and increases solvent consumption. Furthermore, these conventional pathways often rely on the use of strong bases like n-butyllithium to facilitate the introduction of the thioether group.
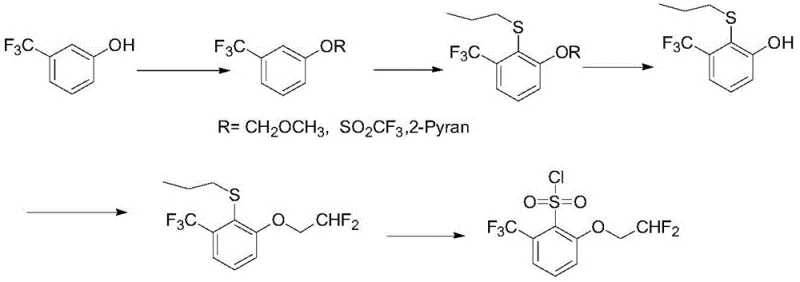
The reliance on n-butyllithium presents severe safety hazards and economic burdens, particularly in large-scale industrial settings where cryogenic conditions and strict moisture exclusion are mandatory. The inability to recover protecting groups further exacerbates the environmental footprint and raw material costs. Additionally, alternative routes reported in patents like CN105801454 utilize starting materials such as 2-trifluoromethyl-6-nitro-chlorobenzene, which are themselves difficult to prepare on a large scale, creating a bottleneck in the supply chain. These cumulative inefficiencies result in a process that is neither economically viable nor operationally robust for modern high-volume manufacturing requirements.
The Novel Approach
In stark contrast, the methodology disclosed in CN112939818B offers a streamlined, direct route that circumvents the pitfalls of protection chemistry. The new strategy initiates with 2-trifluoromethyl-4-nitro-bromobenzene, a commercially accessible precursor, and proceeds through a logical sequence of functional group transformations. The key innovation is the direct introduction of the alkylthio group via a Grignard reaction or Grignard exchange followed by reaction with dialkyl disulfide, completely bypassing the need for hydroxyl protection. This approach significantly shortens the synthetic timeline and improves the overall atom economy of the process. The reaction conditions are notably milder, avoiding the extreme低温 required for lithiation and instead utilizing manageable temperature ranges suitable for standard reactor setups.
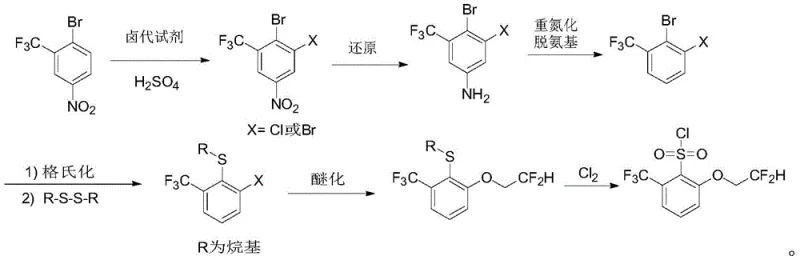
Furthermore, the novel route incorporates a highly efficient oxidative chlorination step to convert the sulfide directly to the sulfonyl chloride using chlorine gas. This final transformation is conducted under controlled conditions (10-60°C), ensuring high conversion rates and minimizing the formation of over-chlorinated byproducts. By integrating these improvements, the patent delivers a synthesis that is not only shorter in step count but also superior in terms of safety and scalability. For procurement managers, this translates to a more reliable source of high-purity agrochemical intermediates with reduced vulnerability to raw material shortages associated with exotic starting materials.
Mechanistic Insights into the Optimized Synthetic Pathway
The chemical elegance of this patent lies in its precise control over regioselectivity and functional group tolerance throughout the six-step sequence. The process begins with the electrophilic aromatic substitution of 2-trifluoromethyl-4-nitro-bromobenzene. By employing halogenating reagents such as NBS, Br2, or Cl2 in concentrated sulfuric acid at temperatures between 50-100°C, the system achieves selective halogenation at the desired position ortho to the trifluoromethyl group. This step is critical as it sets the substitution pattern for the entire molecule, and the acidic medium ensures high regioselectivity while suppressing poly-halogenation side reactions. Following this, the nitro group is reduced to an amine using cost-effective systems like iron powder with ammonium chloride or zinc with hydrochloric acid, which are far more economical than catalytic hydrogenation for certain scales.
Subsequent removal of the amino group via diazotization and decomposition is a masterstroke in simplifying the molecular architecture. Using reagents like t-butyl nitrite or sodium nitrite with hypophosphorous acid, the amino functionality is cleanly excised, leaving behind the desired halogenated aromatic core. This deamination step effectively resets the ring for the crucial carbon-sulfur bond formation. The introduction of the thioether moiety is achieved through a Grignard reaction or metal-halogen exchange using isopropyl magnesium halides, followed by quenching with dialkyl disulfides. This method avoids the harsh conditions of nucleophilic aromatic substitution on electron-rich rings and provides excellent yields. Finally, the etherification with 2,2-difluoroethanol under basic conditions (NaH or NaNH2) and the subsequent oxidation with chlorine gas complete the synthesis with high fidelity, ensuring the final product meets stringent purity specifications required for herbicide active ingredient synthesis.
How to Synthesize 2-(2,2-difluoroethoxy)-6-trifluoromethylbenzenesulfonyl chloride Efficiently
The execution of this synthetic route requires careful attention to reaction parameters, particularly during the halogenation and Grignard steps, to maximize yield and minimize impurities. The patent provides detailed embodiments demonstrating the robustness of the method across different halogenating agents and reduction systems. Operators should note that the choice of solvent and temperature control during the diazotization phase is vital for preventing the accumulation of unstable diazonium salts. The following guide outlines the standardized operational framework derived from the patent examples, serving as a blueprint for process chemists aiming to implement this technology in a pilot or production environment.
- Halogenation of 2-trifluoromethyl-4-nitro-bromobenzene using NBS, Br2, or Cl2 in sulfuric acid at 50-100°C.
- Reduction of the nitro group to an amine using Fe/NH4Cl, Zn/HCl, or catalytic hydrogenation.
- Diazotization of the amine followed by decomposition (deamination) to remove the amino group.
- Grignard reaction or exchange followed by reaction with dialkyl disulfide to introduce the alkylthio group.
- Etherification with 2,2-difluoroethanol under basic conditions (NaH or NaNH2).
- Oxidative chlorination of the sulfide to sulfonyl chloride using chlorine gas.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this patented synthesis offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage is the substantial reduction in manufacturing complexity, which directly correlates to lower production costs and improved margin structures. By eliminating the need for protecting groups and hazardous reagents like n-butyllithium, the process reduces the number of unit operations and the associated utility consumption. This simplification allows for faster batch cycles and higher throughput in existing manufacturing facilities without the need for specialized cryogenic equipment. Consequently, this leads to significant cost reduction in agrochemical intermediate manufacturing, making the final herbicide products more competitive in the global market.
- Cost Reduction in Manufacturing: The elimination of protecting group chemistry removes the cost of additional reagents, solvents, and the energy required for extra reaction and purification steps. Furthermore, replacing expensive and dangerous n-butyllithium with more affordable Grignard reagents or exchange protocols drastically lowers raw material expenses. The use of common reducing agents like iron or zinc powder instead of precious metal catalysts further contributes to a leaner cost structure, ensuring that the final intermediate can be sourced at a highly competitive price point without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 2-trifluoromethyl-4-nitro-bromobenzene mitigates the risk of supply disruptions often associated with custom-synthesized precursors. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, ensures consistent production output even when facing minor variations in raw material quality. This stability is crucial for maintaining continuous supply lines to downstream herbicide manufacturers, reducing lead time for high-purity agrochemical intermediates and preventing production stoppages due to intermediate shortages.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, utilizing reaction conditions that are easily transferable from laboratory to multi-ton reactors. The avoidance of extremely low temperatures and pyrophoric reagents simplifies safety management and reduces the burden on EHS (Environment, Health, and Safety) departments. Additionally, the shorter synthetic route generates less chemical waste, aligning with increasingly strict environmental regulations. This sustainability profile not only facilitates regulatory approval but also enhances the brand reputation of companies adopting this greener manufacturing technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing production portfolios.
Q: How does this new synthetic route improve safety compared to conventional methods?
A: The novel route eliminates the need for n-butyllithium, a highly pyrophoric and hazardous reagent used in traditional lithiation steps. Instead, it utilizes safer Grignard reagents or exchange reactions under controlled temperatures, significantly reducing operational risks in large-scale manufacturing.
Q: What are the cost advantages of avoiding protecting groups in this synthesis?
A: Conventional methods require hydroxyl protection and subsequent deprotection steps, which add extra reagents, solvents, and processing time. By bypassing these steps through a direct functionalization strategy starting from nitro-bromobenzene, the process achieves better atom economy and reduces overall raw material consumption.
Q: Is this process suitable for multi-ton commercial production?
A: Yes, the patent explicitly designs the route for industrial production. It employs mild reaction conditions (50-100°C for halogenation, ambient to moderate temps for others) and avoids extreme cryogenic conditions typical of lithiation, making it highly scalable and robust for commercial supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2,2-difluoroethoxy)-6-trifluoromethylbenzenesulfonyl chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like CN112939818B in driving the next generation of agrochemical solutions. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for this synthesis, including rigorous temperature control for halogenation and safe handling of chlorine gas for the final oxidation step. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-(2,2-difluoroethoxy)-6-trifluoromethylbenzenesulfonyl chloride meets the highest industry standards.
We invite you to collaborate with us to leverage this innovative route for your herbicide development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized synthesis can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of this critical intermediate, empowering your R&D and production teams to focus on delivering superior agricultural solutions to the global market.
