Scalable Metal-Free Electrooxidative Amination for High-Purity 2-Aminoindole Intermediates
Scalable Metal-Free Electrooxidative Amination for High-Purity 2-Aminoindole Intermediates
The landscape of organic synthesis is undergoing a paradigm shift towards sustainable, metal-free methodologies, a trend exemplified by the groundbreaking technology disclosed in patent CN111676487A. This intellectual property introduces a novel electrooxidative amination strategy that directly constructs C-N bonds without the reliance on expensive transition metal catalysts. For R&D directors and process chemists seeking robust pathways for pharmaceutical intermediates, this invention offers a compelling alternative to traditional cross-coupling reactions. By leveraging electricity as a traceless reagent, the method operates within a green aqueous phase system, significantly simplifying downstream processing and waste management. The core innovation lies in the generation of nitrogen radicals via anodic oxidation, which subsequently attack the indole scaffold to form thermodynamically stable 2-amino substituted products. This approach not only aligns with modern green chemistry principles but also addresses critical supply chain vulnerabilities associated with precious metal dependency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aromatic amine structural fragments has relied heavily on transition metal-catalyzed cross-coupling reactions, such as the classic Ullmann condensation or the more modern Buchwald-Hartwig amination. As illustrated in the general reaction scheme below, these conventional pathways typically necessitate the pre-functionalization of the aromatic ring, often requiring halogenated precursors that add synthetic steps and cost.
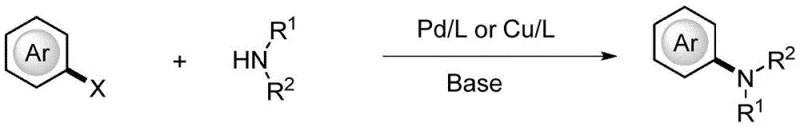
Furthermore, these methods invariably introduce heavy metal residues into the final product, necessitating rigorous and costly purification protocols to meet stringent regulatory standards for high-purity API intermediates. The requirement for specialized ligands, inert atmospheres, and anhydrous conditions further complicates the operational complexity, making scale-up challenging and economically burdensome for cost reduction in pharmaceutical manufacturing. The reliance on palladium or copper also exposes the supply chain to volatility in metal prices and availability, creating potential bottlenecks for continuous production lines.
The Novel Approach
In stark contrast, the electrooxidative method presented in CN111676487A bypasses these hurdles by utilizing direct C-H activation driven by anodic oxidation. The reaction proceeds through a radical mechanism where sulfonamides are oxidized at the anode to generate nitrogen-centered radicals, which then selectively attack the C2 position of the indole ring. This eliminates the need for pre-halogenation, streamlining the synthetic route and reducing the overall material footprint. The process operates in a mixed solvent system of 1,4-Dioxane and water, utilizing simple inorganic bases like potassium phosphate, which are inexpensive and readily available globally. By removing the transition metal catalyst entirely, the method inherently produces a cleaner crude profile, drastically reducing the burden on purification teams and enabling faster time-to-market for complex drug candidates.
Mechanistic Insights into Electrooxidative C-N Bond Formation
The mechanistic pathway of this transformation is a sophisticated interplay of electrochemical oxidation and radical chemistry, offering precise control over regioselectivity. Initially, the sulfonamide substrate undergoes deprotonation by the base to form a nitrogen anion, which is subsequently oxidized at the anode surface to generate a reactive nitrogen radical species. This radical acts as an electrophile, attacking the electron-rich C2 position of the indole nucleus to form a benzylic radical intermediate. This step is thermodynamically favored due to the stability of the resulting radical species adjacent to the aromatic system. Following this addition, the intermediate undergoes a second single-electron transfer (SET) oxidation at the anode to yield a cationic species. Finally, the loss of a proton restores aromaticity, delivering the desired 2-aminoindole derivative. This elegant cascade ensures high atom economy and minimizes the formation of over-oxidized byproducts often seen in chemical oxidant-mediated reactions.
From an impurity control perspective, the electrochemical nature of the reaction allows for fine-tuning of the oxidation potential by adjusting the current density, typically maintained at 4 mA in the optimized protocol. This precise control prevents the over-oxidation of sensitive functional groups that might be present on the indole or sulfonamide scaffolds, such as aldehydes or esters. The use of an open-air system further mitigates safety risks associated with hydrogen gas accumulation at the cathode, ensuring a safe operating environment for plant personnel. The absence of metal catalysts also means there is no risk of metal-induced decomposition of the product during storage, enhancing the long-term stability of the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2-Aminoindole Efficiently
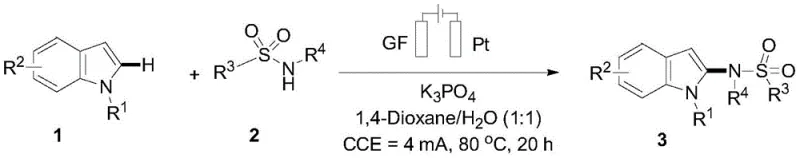
The practical implementation of this electrooxidative protocol is straightforward and requires standard laboratory or pilot-plant electrolysis equipment. The process begins with the dissolution of the substituted indole and sulfonamide reactants in a 1:1 volume ratio of 1,4-Dioxane and water, supplemented with potassium phosphate as the supporting electrolyte and base. The reaction is conducted under constant current electrolysis using a graphite felt anode and a platinum sheet cathode, maintaining a temperature of 80°C for approximately 20 hours to ensure complete conversion. Detailed standardized synthesis steps, including specific stoichiometry and workup procedures, are provided in the guide below.
- Prepare the reaction mixture by dissolving substituted indole and sulfonamide compounds in a 1: 1 mixture of 1,4-Dioxane and water with potassium phosphate base.
- Set up the electrolytic cell using a graphite felt (GF) anode and a platinum sheet cathode, maintaining a constant current of 4 mA at 80°C for 20 hours.
- Upon completion, remove solvents under vacuum, adsorb the residue on silica, and purify via column chromatography to isolate the 2-aminoindole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free electrochemical technology translates into tangible strategic advantages beyond mere technical novelty. The elimination of precious metal catalysts removes a significant variable cost component and decouples production from the volatile commodities market associated with palladium and copper. Furthermore, the simplified reaction setup, which does not require rigorous exclusion of moisture or oxygen, reduces the capital expenditure needed for specialized reactor infrastructure. This robustness facilitates easier technology transfer from R&D to manufacturing sites, ensuring consistent supply continuity even in resource-constrained environments.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of expensive transition metal catalysts and specialized ligands, which traditionally account for a substantial portion of raw material costs in C-N bond forming reactions. Additionally, the absence of metal residues eliminates the need for costly scavenging resins or complex extraction processes designed to lower metal content to ppm levels, thereby streamlining the downstream purification workflow. The use of inexpensive inorganic bases and common solvents further drives down the bill of materials, resulting in significant overall cost savings per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: By relying on electricity as the primary oxidant and readily available commodity chemicals like indoles and sulfonamides, the process mitigates risks associated with the supply of specialized reagents. The operational simplicity, characterized by an open-air system and ambient pressure conditions, reduces the likelihood of unplanned downtime due to equipment failure or safety incidents. This reliability is crucial for maintaining uninterrupted production schedules for critical pharmaceutical intermediates, ensuring that downstream API synthesis is never delayed by precursor shortages.
- Scalability and Environmental Compliance: The use of a water-containing solvent system aligns perfectly with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. The electrochemical nature of the reaction generates minimal hazardous waste, as the primary byproduct is hydrogen gas, which is safely vented in the open system configuration. This green profile simplifies waste disposal logistics and reduces the environmental compliance burden, making the process highly attractive for large-scale manufacturing facilities aiming to reduce their carbon footprint and achieve sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrooxidative amination process, derived directly from the experimental data and specifications outlined in the patent documentation. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this technology for integration into their existing manufacturing portfolios.
Q: Does this electrooxidative method require transition metal catalysts?
A: No, the process described in patent CN111676487A is entirely metal-free, utilizing electricity as the sole oxidant to drive the radical mechanism, thereby eliminating heavy metal contamination risks.
Q: What are the optimal reaction conditions for maximum conversion?
A: The patent specifies optimal conditions as a constant current of 4 mA, a temperature of 80°C, and a reaction time of 20 hours using a K3PO4 base in a 1,4-Dioxane/Water solvent system.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the use of simple electrode materials like graphite felt and platinum, combined with an open-air system and aqueous solvents, makes the process highly scalable and environmentally compliant for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electroorganic synthesis in modernizing the production of high-value chemical building blocks. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this metal-free amination can be seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering high-purity 2-aminoindole derivatives that empower your drug discovery and development programs.
We invite you to collaborate with our technical team to explore how this cost-effective and sustainable methodology can be tailored to your specific project needs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis for your target molecules. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your timeline and optimize your manufacturing budget.
