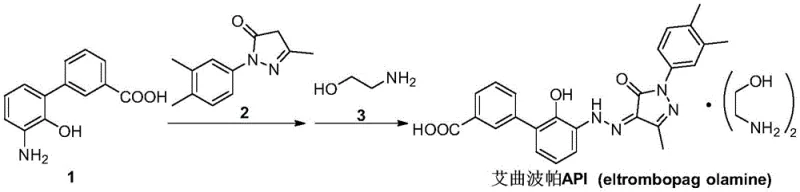
Advanced Synthetic Route for Eltrombopag Intermediate Enhancing Purity and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic pathways for critical active pharmaceutical ingredient (API) precursors, and the recent disclosure of patent CN115322110A presents a significant advancement in the production of Eltrombopag intermediates. This specific technology focuses on the efficient synthesis of 3'-amino-2'-hydroxybiphenyl-3-carboxylic acid, a pivotal building block required for the assembly of Eltrombopag olamine, which is widely used in treating thrombocytopenia. The innovation lies in a meticulously designed four-step sequence that prioritizes safety, environmental compliance, and economic feasibility without compromising on chemical purity. By leveraging a novel heteropolyacid ionic liquid catalyst system, the method overcomes historical bottlenecks associated with toxic reagents and difficult purification processes. For R&D directors and procurement strategists, understanding this technological shift is crucial for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent regulatory standards. The following analysis dissects the technical merits and commercial implications of this patented approach.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
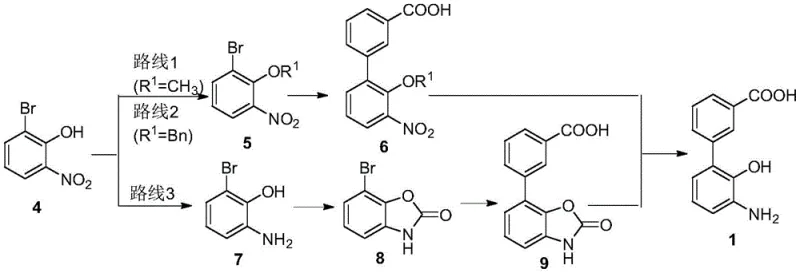
Historically, the manufacturing landscape for this specific biphenyl carboxylic acid derivative has been plagued by hazardous chemical dependencies and inefficient reaction conditions that hinder scalability. Traditional strategies, such as those utilizing methyl iodide for hydroxyl protection, introduce severe safety risks due to the toxicity and volatility of the alkylating agents involved. Furthermore, alternative routes requiring benzyl protection often necessitate high-pressure hydrogenation operations, which demand specialized equipment and pose significant explosion hazards in a plant environment. Another common pathway involves the use of N,N'-carbonyldiimidazole, a reagent known for its instability and difficulty in storage and transport, adding logistical friction to the supply chain. These legacy methods also frequently suffer from poor coupling yields and require harsh acidic conditions for deprotection, leading to complex waste streams that are costly to treat and dispose of responsibly. Consequently, these factors collectively inflate the cost of goods and limit the ability to achieve consistent large-scale production.
The Novel Approach
In stark contrast, the methodology outlined in patent CN115322110A introduces a streamlined protocol that eliminates the need for dangerous alkylating agents and high-pressure infrastructure. The new route initiates with a mild nitro reduction using safe hydrogen sources like ammonium formate, followed by a groundbreaking cyclization step mediated by a reusable heteropolyacid ionic liquid. This catalytic system not only accelerates the reaction kinetics but also allows for straightforward recovery and recycling, fundamentally altering the economic model of the synthesis. The subsequent Suzuki coupling employs readily available palladium carbon, which can be filtered and reused, further driving down material costs and minimizing heavy metal contamination in the final product. Finally, the hydrolysis deprotection is achieved under controlled alkaline conditions that ensure high conversion rates without generating excessive corrosive waste. This holistic redesign of the synthetic tree represents a substantial leap forward in cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Heteropolyacid Ionic Liquid Catalyzed Cyclization
The core technical differentiator of this process is the application of heteropolyacid ionic liquids during the cyclization protection step, which warrants a deep mechanistic understanding for technical stakeholders. These catalysts function by providing a highly acidic microenvironment that facilitates the condensation between the amino-phenol intermediate and the aldehyde derivative without requiring volatile organic solvents. The ionic nature of the catalyst ensures that it remains in a distinct phase or can be easily precipitated, allowing for physical separation from the reaction mixture through simple filtration rather than energy-intensive distillation. This mechanism effectively suppresses side reactions that typically lead to isomeric impurities, thereby enhancing the overall purity profile of the intermediate before it even reaches the coupling stage. Moreover, the structural stability of the heteropolyanion ensures that the catalytic activity remains consistent over multiple cycles, providing a predictable and robust performance profile essential for GMP manufacturing environments. Such precision in catalytic design directly translates to reduced batch-to-batch variability and higher confidence in quality control outcomes.
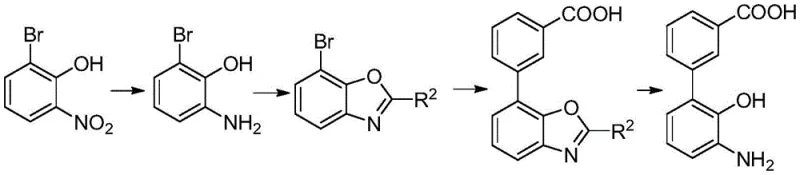
Impurity control is another critical aspect where this novel mechanism excels, particularly regarding the management of halogenated byproducts and residual metals. By avoiding the use of methyl iodide and benzyl bromide, the process inherently prevents the formation of genotoxic alkyl halides that are notoriously difficult to purge to acceptable limits. The Suzuki coupling step is optimized to minimize homocoupling of the boronic acid, a common side reaction that can compromise the purity of high-purity pharmaceutical intermediates. The use of specific bases and solvent mixtures in the coupling stage further fine-tunes the reaction selectivity, ensuring that the desired biphenyl bond is formed with maximal efficiency. Additionally, the final hydrolysis step is designed to cleave the protecting group cleanly without affecting the sensitive amino or carboxylic acid functionalities on the molecule. This comprehensive approach to impurity management ensures that the final API intermediate meets the rigorous specifications demanded by global regulatory bodies.
How to Synthesize 3'-Amino-2'-Hydroxybiphenyl-3-Carboxylic Acid Efficiently
Implementing this synthesis requires adherence to specific operational parameters to maximize yield and safety while maintaining the integrity of the catalytic systems. The process begins with the reduction of the nitro group, followed by the critical cyclization where temperature and catalyst loading must be precisely controlled to activate the ionic liquid effectively. Subsequent coupling reactions require an inert atmosphere to protect the palladium catalyst from oxidation, ensuring consistent turnover numbers throughout the batch. The final hydrolysis demands careful pH adjustment to precipitate the product while keeping impurities in the aqueous phase. Detailed standardized operating procedures covering these nuances are essential for technology transfer and successful commercial adoption. The complete technical workflow is summarized below for immediate reference by process engineering teams.
- Perform nitro reduction on 2-bromo-6-nitrophenol using a hydrogen source and metal catalyst to obtain 2-amino-6-bromophenol.
- Execute cyclization protection using a heteropolyacid ionic liquid catalyst to form the benzoxazole intermediate.
- Conduct Suzuki coupling reaction with 3-carboxyphenylboronic acid using palladium carbon catalyst.
- Finalize with hydrolysis deprotection under alkaline conditions to yield the target carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented technology offers compelling advantages that directly address the pain points of modern pharmaceutical supply chains, particularly regarding cost stability and vendor reliability. By eliminating the dependency on scarce and regulated toxic reagents, manufacturers can secure a more resilient supply of raw materials that is less susceptible to geopolitical or regulatory disruptions. The ability to recycle expensive catalysts multiple times significantly lowers the variable cost per kilogram, allowing for more competitive pricing structures without sacrificing margin quality. Furthermore, the simplified workup procedures reduce the burden on utility systems and waste treatment facilities, contributing to substantial cost savings in overhead and environmental compliance fees. These efficiencies collectively enhance the commercial viability of the intermediate, making it an attractive option for long-term supply agreements.
- Cost Reduction in Manufacturing: The elimination of high-cost protecting group reagents like methyl iodide and the implementation of recyclable ionic liquid catalysts create a leaner cost structure that is highly resistant to raw material price volatility. The ability to recover and reuse palladium carbon catalysts further diminishes the consumption of precious metals, which are often a major driver of production expenses in cross-coupling reactions. Additionally, the high conversion rates observed in the hydrolysis step minimize material loss, ensuring that the theoretical yield is closely approached in practical operations. These factors combine to deliver a manufacturing process that is economically superior to legacy methods, offering significant value to procurement managers seeking to optimize their bill of materials.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is streamlined because the starting materials, such as 2-bromo-6-nitrophenol, are commodity chemicals with established global supply networks. The avoidance of specialized, hazardous reagents reduces the regulatory burden on logistics providers, ensuring smoother transportation and fewer delays at customs or storage facilities. The robustness of the reaction conditions also means that production is less likely to be interrupted by equipment failures or safety incidents, guaranteeing a steady flow of material to downstream API manufacturers. This reliability is critical for reducing lead time for high-purity pharmaceutical intermediates and maintaining continuous production schedules.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, as it avoids high-pressure hydrogenation and extreme temperatures that limit reactor size and throughput. The green chemistry principles embedded in the method, such as catalyst recycling and reduced solvent usage, align perfectly with increasingly strict environmental regulations, future-proofing the manufacturing site against compliance risks. Waste streams are simpler to treat due to the absence of heavy halogenated organics, lowering the cost and complexity of effluent management. This environmental compatibility ensures sustainable production capabilities that meet the ESG goals of modern multinational corporations.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to the most pertinent questions raised by industry experts and potential partners. These responses are grounded in the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details is essential for making informed decisions regarding process adoption and supplier qualification. The following section addresses key concerns regarding safety, cost, and scalability.
Q: Why is this new synthetic route considered safer than conventional methods?
A: Conventional routes often rely on highly toxic methyl iodide or corrosive hydrogen bromide, whereas this patent utilizes safer hydrogen sources and recyclable ionic liquid catalysts, significantly reducing operational hazards and waste treatment complexity.
Q: How does the heteropolyacid ionic liquid catalyst impact production costs?
A: The catalyst exhibits high activity and can be recovered and reused multiple times through simple filtration, which drastically reduces the consumption of expensive catalytic materials and lowers the overall cost of goods sold.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process features mild reaction conditions, high conversion rates, and avoids high-pressure hydrogenation steps found in other routes, making it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3'-Amino-2'-Hydroxybiphenyl-3-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly evaluated the route described in patent CN115322110A and confirmed its potential for robust, large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of Eltrombopag intermediate meets the highest international standards. Partnering with us means gaining access to a supply chain that is both technically sophisticated and commercially reliable.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener, more efficient route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume and timeline. Let us help you engineer a more resilient and cost-effective supply chain for your critical pharmaceutical ingredients.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
