Revolutionizing 4-ADPA Production: Scalable Phase Transfer Catalysis for Global Supply Chains
The chemical landscape for producing critical intermediates like 4-aminodiphenylamine (4-ADPA) is undergoing a significant transformation driven by the need for more sustainable and cost-efficient manufacturing processes. Patent CN100546972C introduces a groundbreaking method that leverages phase transfer catalysis to optimize the coupling of aniline and nitrobenzene, addressing long-standing challenges in yield and selectivity. This technology is particularly vital for industries relying on 4-ADPA as a precursor for antiozonants and antioxidants used in rubber protection, as well as for various pharmaceutical applications requiring high-purity aromatic amines. By utilizing a mixture comprising a strong base, a suitable phase transfer catalyst, and an oxidant, the process achieves superior reaction kinetics under milder conditions compared to traditional anaerobic methods. The strategic implementation of this patented approach allows manufacturers to bypass the limitations of earlier techniques, such as the reliance on expensive crown ethers or complex solvent systems, thereby establishing a new benchmark for industrial efficiency. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic advancements is crucial for securing a competitive edge in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-aminodiphenylamine derivatives has been plagued by inefficiencies associated with conventional catalytic systems, particularly those relying on crown ethers like 18-crown-6. While effective in facilitating the reaction between aniline and nitrobenzene in the presence of alkali metal hydroxides, crown ethers present substantial economic and environmental drawbacks that hinder large-scale commercial viability. The initial cost of crown ethers is prohibitively high, and their high toxicity poses significant safety risks for plant operators and environmental compliance teams. Furthermore, their poor solubility in water complicates recovery and recycling processes, often leading to substantial losses in the organic product stream or requiring energy-intensive distillation steps for separation. Another critical issue is the tendency of crown ethers to act as chelating agents, which can irreversibly bind to expensive hydrogenation catalyst metals, leading to unacceptable catalyst deactivation and increased operational expenditures. These factors collectively create a bottleneck for cost reduction in fine chemical manufacturing, necessitating a shift towards more robust and economically feasible catalytic alternatives.
The Novel Approach
The innovative methodology described in the patent data circumvents these issues by employing specific quaternary ammonium salts as phase transfer catalysts, which offer a compelling combination of performance and economic practicality. Unlike crown ethers, these catalysts, such as tetramethylammonium chloride, are readily available, less toxic, and can be effectively managed within aqueous reaction systems. The process allows for the reaction to proceed in the presence of strong inorganic bases like potassium hydroxide, facilitating the formation of 4-aminodiphenylamine intermediates with excellent yield and selectivity. A key feature of this novel approach is the flexibility to operate under partially oxidative conditions, where the introduction of free oxygen or peroxides enhances selectivity towards the desired nitroso and nitro intermediates. This capability not only simplifies the reaction setup by removing the strict requirement for anaerobic atmospheres but also aligns with modern green chemistry principles by reducing waste and energy consumption. For supply chain leaders, this translates to a more resilient production model capable of commercial scale-up of complex polymer additives and rubber chemicals without the baggage of hazardous reagent handling.
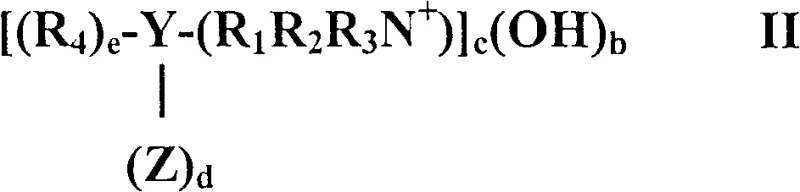
Mechanistic Insights into Phase Transfer Catalyzed Coupling
The core of this technological advancement lies in the precise interaction between the phase transfer catalyst and the reactive species within the biphasic or aqueous reaction medium. The catalyst, generally defined by structures such as Formula I shown in the patent documentation, functions by transporting the anionic species generated by the strong base into the organic phase where the nitrobenzene and aniline reside. This transport mechanism dramatically increases the local concentration of reactive ions at the interface, thereby accelerating the nucleophilic attack of the aniline on the nitrobenzene. The structural versatility of the catalyst, allowing for variations in alkyl chain lengths and anionic counterions, enables fine-tuning of the reaction kinetics to match specific production requirements. For instance, the use of tetraalkylammonium halides has been observed to provide a periodic trend in efficiency, with fluoride and chloride salts often outperforming their heavier halide counterparts in terms of conversion rates. This level of control is essential for R&D directors focused on purity and impurity profiles, as it minimizes the formation of unwanted side products like phenazines or azobenzenes that complicate downstream purification.
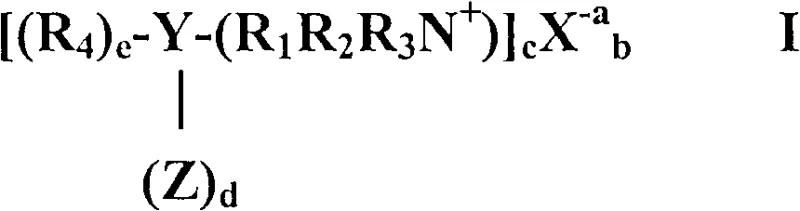
Furthermore, the mechanism incorporates a sophisticated balance of oxidative and reductive steps to ensure the final product meets stringent quality standards. The initial coupling reaction produces intermediates such as 4-nitrosodiphenylamine (p-NDPA) and 4-nitrodiphenylamine (4-NDPA), the ratio of which can be manipulated by adjusting the stoichiometry of the reactants and the presence of oxidizing agents. Experimental data indicates that maintaining an excess of aniline favors the formation of p-NDPA, while excess nitrobenzene acts as an internal oxidant to improve overall selectivity. The subsequent reduction of these intermediates to 4-ADPA is typically achieved through hydrogenation using standard catalysts like platinum on carbon, a step that benefits significantly from the high purity of the precursor stream generated by the PTC method. By eliminating the chelating agents found in older technologies, the process ensures that the hydrogenation catalyst remains active for longer cycles, reducing downtime and maintenance costs. This mechanistic clarity provides a solid foundation for developing robust standard operating procedures that guarantee consistent batch-to-batch quality.
How to Synthesize 4-Aminodiphenylamine Efficiently
Implementing this synthesis route requires careful attention to the preparation of the reaction mixture and the control of process parameters to maximize efficiency. The procedure begins with the reactive contacting of aniline or its derivatives with nitrobenzene in a defined zone, ensuring that the molar ratios are optimized to favor the desired intermediate formation. Detailed standardized synthesis steps are critical for reproducibility, involving the precise addition of strong bases like potassium hydroxide and phase transfer catalysts such as tetramethylammonium chloride under controlled temperature conditions ranging from 20°C to 150°C. The management of water content is also pivotal, as aqueous systems can enhance the solubility of the base and facilitate the phase transfer mechanism, provided that the water-to-nitrobenzene ratio is kept within optimal limits to prevent hydrolysis or dilution effects. Operators must also monitor the oxidative environment, potentially introducing air or peroxides at specific intervals to boost selectivity without causing over-oxidation.
- Prepare a reaction mixture comprising aniline or its derivatives and nitrobenzene or its derivatives in a defined zone.
- React the mixture in the presence of a strong base, an oxidizing agent, and a specific phase transfer catalyst at temperatures between 20°C and 150°C.
- Reduce the resulting 4-aminodiphenylamine intermediate product, such as 4-nitrodiphenylamine, to produce the final 4-aminodiphenylamine or substituted derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this phase transfer catalysis method offers profound benefits that extend beyond mere technical feasibility, directly impacting the bottom line and supply chain stability. The elimination of expensive and toxic crown ethers results in substantial cost savings regarding raw material procurement and waste disposal, making the process economically attractive for high-volume production. Additionally, the use of commodity chemicals like potassium hydroxide and tetraalkylammonium salts ensures a stable supply of reagents, mitigating the risk of shortages that often plague specialty chemical markets. The ability to operate in aqueous media further reduces the dependency on volatile organic solvents, lowering both the environmental footprint and the costs associated with solvent recovery and emission control systems. For procurement managers, this means negotiating contracts with greater confidence, knowing that the underlying manufacturing process is built on a foundation of readily available and cost-effective inputs. The streamlined workflow also supports faster turnaround times, enabling suppliers to respond more agilely to fluctuating market demands.
- Cost Reduction in Manufacturing: The substitution of high-cost crown ethers with economical quaternary ammonium salts drastically lowers the direct material costs associated with the catalytic system. Moreover, the prevention of hydrogenation catalyst poisoning by chelating agents extends the lifespan of precious metal catalysts, leading to significant long-term savings in catalyst replacement and regeneration expenses. The simplified workup procedures, facilitated by the aqueous compatibility of the reagents, reduce energy consumption related to distillation and solvent exchange, contributing to a leaner manufacturing overhead. These cumulative efficiencies allow for a more competitive pricing structure without compromising on the quality of the final high-purity OLED material or pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: By relying on widely available inorganic bases and organic salts, the manufacturing process becomes less vulnerable to supply chain disruptions caused by the scarcity of exotic reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures, ensures that production can continue even under variable utility conditions, enhancing overall plant reliability. This stability is crucial for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the risk of batch failures and production delays. Suppliers can thus maintain consistent inventory levels and meet delivery commitments more reliably, fostering stronger partnerships with downstream customers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively in both batch and continuous modes, which supports the transition from pilot scale to full commercial production with minimal technical risk. The reduced use of hazardous organic solvents and toxic catalysts aligns with increasingly stringent environmental regulations, simplifying the permitting process and reducing the liability associated with chemical handling. Waste streams are easier to treat due to the lower toxicity profile, facilitating compliance with local and international environmental standards. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible partner in the global chemical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, providing clarity for stakeholders evaluating its potential integration into their operations. These insights are derived directly from the experimental data and embodiments disclosed in the patent, ensuring that the information is grounded in verified scientific evidence. Understanding these nuances is essential for making informed decisions about process adoption and supplier selection.
Q: What are the preferred phase transfer catalysts for 4-ADPA synthesis?
A: The patent highlights tetraalkylammonium salts, specifically tetramethylammonium chloride and tetramethylammonium hydroxide, as highly effective catalysts that significantly improve yield and selectivity compared to traditional methods.
Q: Can this process be operated in an aqueous environment?
A: Yes, the method is particularly advantageous when using water-soluble bases like potassium hydroxide, allowing for reactions in aqueous solutions which simplifies workup and reduces solvent costs.
Q: How does the addition of oxidants affect the reaction selectivity?
A: Introducing oxidizing agents such as free oxygen (air) or hydrogen peroxide during specific phases of the reaction significantly improves selectivity towards the desired 4-ADPA intermediates while minimizing by-product formation like azobenzene.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminodiphenylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the pharmaceutical and specialty chemical industries. Our expertise as a CDMO partner allows us to leverage technologies like the phase transfer catalysis method described in CN100546972C to deliver superior results for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from development to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 4-aminodiphenylamine meets the highest standards of quality and consistency required for sensitive applications. By partnering with us, you gain access to a team of experts dedicated to optimizing process parameters and resolving any technical challenges that may arise during scale-up.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific supply chain needs. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this more efficient manufacturing process. We are prepared to provide specific COA data and route feasibility assessments to support your decision-making process. Let us help you secure a reliable supply of high-quality intermediates while driving down costs and enhancing your operational efficiency. Contact us today to initiate a conversation about your next project and discover the NINGBO INNO PHARMCHEM advantage.
