Scalable Synthesis of Pitavastatin Key Intermediate via Novel Lewis Acid Catalysis for Commercial Manufacturing
Scalable Synthesis of Pitavastatin Key Intermediate via Novel Lewis Acid Catalysis for Commercial Manufacturing
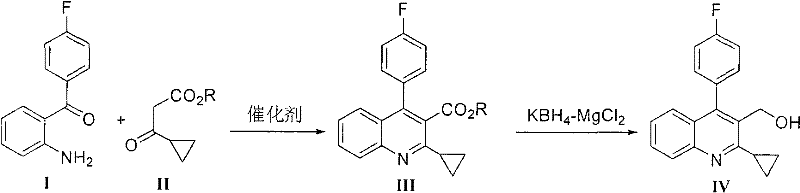
The global demand for potent HMG-CoA reductase inhibitors continues to drive innovation in the synthesis of their critical precursors, particularly for next-generation statins like Pitavastatin. Patent CN101747265B introduces a transformative methodology for preparing 2-cyclopropyl-4-(4'-fluorophenyl)quinoline-3-methanol, a pivotal building block in the lipid-lowering drug value chain. This technical disclosure moves away from traditional stoichiometric acid catalysis, proposing a greener, more efficient route utilizing catalytic Zinc Triflate (Zn(OTf)2). For pharmaceutical manufacturers and procurement strategists, this shift represents not merely a chemical optimization but a fundamental restructuring of cost drivers and supply chain resilience. By enabling catalyst recovery and simplifying downstream processing, this technology addresses the dual pressures of regulatory compliance and margin protection in the highly competitive API intermediate market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinoline core for Pitavastatin intermediates has relied heavily on harsh cyclization conditions employing stoichiometric amounts of strong Brønsted acids. Prior art, as detailed in the background of the patent, frequently utilizes catalysts such as p-toluenesulfonic acid (TsOH), methanesulfonic acid (MsOH), or sulfuric acid in molar equivalents relative to the substrate. These conventional approaches suffer from significant drawbacks, including the generation of large volumes of acidic waste streams that require costly neutralization and disposal procedures. Furthermore, the use of equimolar acid often leads to difficult work-up procedures where product isolation is complicated by salt formation or emulsion issues. In some documented cases, yields have been suboptimal, hovering around 74%, which directly impacts the cost of goods sold (COGS) and creates bottlenecks in large-scale production campaigns where material throughput is paramount.
The Novel Approach
In stark contrast, the methodology disclosed in CN101747265B leverages the unique properties of Lewis acid catalysis to achieve superior efficiency and environmental performance. The core innovation lies in the use of Zinc Triflate (Zn(OTf)2) at catalytic loadings ranging from 1 mol% to 40 mol%, with an optimal window identified between 5 mol% and 10 mol%. This drastic reduction in catalyst loading eliminates the need for massive acid neutralization steps. Moreover, the process allows for the direct use of the crude cyclization product in the subsequent reduction step, effectively telescoping two distinct chemical transformations. This operational simplicity reduces solvent consumption, minimizes equipment occupancy time, and significantly lowers the overall energy footprint of the manufacturing process, making it an ideal candidate for modern, sustainable chemical production facilities.
Mechanistic Insights into Zn(OTf)2-Catalyzed Cyclization
The mechanistic elegance of this route stems from the high turnover frequency and specific activation capability of the Zinc Triflate catalyst. As a water-tolerant Lewis acid, Zn(OTf)2 effectively activates the carbonyl group of the 3-cyclopropyl-3-oxopropionate ester, facilitating nucleophilic attack by the amino group of the 2-amino-4'-fluorobenzophenone. Unlike protic acids which can promote non-specific hydrolysis or degradation of sensitive functional groups, the Lewis acid pathway offers a controlled environment for ring closure. This selectivity is crucial for maintaining the integrity of the cyclopropyl moiety, which is prone to ring-opening under harsh acidic conditions. The mild nature of the catalytic cycle ensures that side reactions are minimized, resulting in a crude product profile that is remarkably clean. This inherent purity is a key enabler for the telescoped process, as it removes the necessity for intermediate crystallization or chromatographic purification before the reduction step.
Furthermore, the impurity control mechanism is intrinsically linked to the recyclability of the catalyst system. Upon completion of the cyclization, the reaction mixture is treated with water, allowing the zinc species to partition into the aqueous phase while the organic product remains in the organic layer. This phase separation serves a dual purpose: it isolates the product and simultaneously recovers the catalyst. The aqueous layer can be evaporated to dryness to reclaim Zn(OTf)2 for reuse in subsequent batches, creating a closed-loop system that prevents metal accumulation in the final API. This feature is particularly valuable for R&D directors focused on heavy metal specifications, as it inherently limits the risk of zinc contamination in the final active pharmaceutical ingredient, thereby simplifying the validation and quality control protocols required for regulatory filing.
How to Synthesize 2-Cyclopropyl-4-(4'-fluorophenyl)quinoline-3-methanol Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale operations, emphasizing reproducibility and safety. The process begins with the reflux of reactants in ethanol, a benign and widely available solvent, ensuring that the reaction proceeds to completion within a reasonable timeframe. Following the cyclization, the solvent is partially removed, and the residue is subjected to a liquid-liquid extraction to separate the catalyst. The resulting organic solution containing the crude quinoline ester is then directly introduced into the reduction reactor.
- Cyclization of 2-amino-4'-fluorobenzophenone with 3-cyclopropyl-3-oxopropionate using catalytic Zn(OTf)2 in ethanol.
- Isolation of the crude quinoline-3-carboxylate ester intermediate via solvent extraction and catalyst recovery.
- Reduction of the crude ester to the target methanol using KBH4 and MgCl2 in a THF-toluene system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology translates into tangible strategic advantages beyond simple yield improvements. The shift from stoichiometric to catalytic reagents fundamentally alters the raw material cost structure, removing the burden of purchasing and disposing of large quantities of strong acids. Additionally, the ability to recycle the zinc catalyst creates a buffer against raw material price volatility, as the effective consumption of this specialized reagent is negligible over the lifecycle of a production campaign. The streamlined workflow, which avoids intermediate isolation, also reduces the requirement for extensive drying and handling equipment, lowering capital expenditure requirements for new production lines dedicated to this intermediate.
- Cost Reduction in Manufacturing: The implementation of a catalytic cycle with recoverable reagents significantly lowers the variable costs associated with reagent consumption and waste treatment. By eliminating the need for stoichiometric amounts of expensive or hazardous acids, the process reduces the direct material input costs per kilogram of product. Furthermore, the simplified work-up procedure decreases the consumption of auxiliary materials such as neutralizing bases and washing solvents. This cumulative effect results in a leaner manufacturing process where the cost of goods is optimized through chemical efficiency rather than sheer volume scaling, providing a competitive edge in pricing negotiations for long-term supply contracts.
- Enhanced Supply Chain Reliability: The robustness of the Zn(OTf)2 catalyzed route enhances supply continuity by reducing the complexity of the manufacturing process. Fewer unit operations mean fewer points of failure and reduced risk of batch deviations that could lead to production delays. The use of common solvents like ethanol and toluene ensures that raw material sourcing is not dependent on niche suppliers, mitigating the risk of supply disruptions. Moreover, the high purity of the crude intermediate allows for flexible scheduling, as the pressure to perform rigorous intermediate quality checks is alleviated. This agility enables manufacturers to respond more rapidly to fluctuations in downstream API demand, ensuring consistent delivery performance to global pharmaceutical partners.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this methodology aligns perfectly with modern green chemistry principles and stringent regulatory standards. The reduction in acidic waste generation simplifies effluent treatment processes, lowering the environmental compliance burden and associated fees. The process is inherently safer due to the milder reaction conditions and the avoidance of corrosive bulk acids, which improves workplace safety metrics. Scalability is further supported by the exothermic profile of the reaction, which is manageable in large reactors, allowing for seamless technology transfer from pilot plants to multi-ton commercial production facilities without the need for extensive process re-engineering or safety modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What is the primary advantage of using Zn(OTf)2 over traditional acid catalysts?
A: Unlike stoichiometric acids like TsOH or MsOH which generate significant waste, Zn(OTf)2 is used in catalytic amounts (5-10 mol%) and can be easily recovered from the aqueous layer for reuse, drastically reducing environmental impact and raw material costs.
Q: Can the intermediate be purified before the reduction step?
A: The patent demonstrates that the crude cyclization product possesses sufficient purity to be directly subjected to the reduction step without additional purification, streamlining the workflow and improving overall throughput.
Q: What is the expected yield profile for this two-step sequence?
A: Experimental data within the patent indicates that the two-step sequence consistently achieves yields exceeding 80%, demonstrating high efficiency suitable for industrial scale-up compared to older methods yielding around 74%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyclopropyl-4-(4'-fluorophenyl)quinoline-3-methanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful commercialization of life-saving medications like Pitavastatin. Our technical team has extensively analyzed advanced synthetic routes, including the Zn(OTf)2 catalyzed methodology, to ensure our manufacturing capabilities meet the highest standards of efficiency and purity. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 2-cyclopropyl-4-(4'-fluorophenyl)quinoline-3-methanol delivered meets the exacting requirements of global regulatory bodies.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and operational excellence can drive value and security for your pharmaceutical manufacturing operations.
