Advanced One-Step Synthesis of Tetrahydropyranyl Sulfonates for Commercial Scale-Up
Advanced One-Step Synthesis of Tetrahydropyranyl Sulfonates for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and scalable synthetic routes for critical intermediates. A pivotal advancement in this domain is documented in patent CN1409707A, which introduces a groundbreaking process for producing tetrahydropyranyl-4-sulfonate and its subsequent conversion into 4-aminotetrahydropyran compounds. This technology addresses long-standing inefficiencies in the production of these valuable scaffolds, which are essential building blocks in the synthesis of various pharmaceutical agents and agrochemical products. By leveraging a direct reaction between readily available 3-buten-1-ol, formaldehyde derivatives, and organic sulfonic acids, this method eliminates the need for unstable intermediates and complex multi-step sequences. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic and economic implications of this patent is crucial for optimizing supply chains and reducing overall manufacturing costs in competitive markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydropyranyl-4-sulfonates has been plagued by inefficiencies inherent in multi-step processes that rely on unstable precursors. Traditional methodologies, such as those cited in older literature like Chem. Ber. (1955), typically involve a two-step sequence starting from 3-buten-1-ol. First, the alcohol is converted into tetrahydropyran-4-ol, often achieving yields around 76 percent under acidic conditions. However, the subsequent sulfonation step to generate the methanesulfonate ester frequently suffers from poor conversion rates, with reported yields dropping drastically to approximately 35 percent. Furthermore, alternative routes attempting to utilize tetrahydropyran-4-one face severe challenges due to the compound's extreme instability towards alkali and its propensity to undergo ring-opening polymerization. These technical hurdles not only depress overall process yields but also introduce significant variability and safety risks during handling, making cost reduction in pharmaceutical intermediate manufacturing difficult to achieve with legacy technologies.
The Novel Approach
In stark contrast to these cumbersome traditional pathways, the novel approach detailed in the patent data offers a streamlined, one-step synthesis that fundamentally reshapes the production economics of these intermediates. By reacting 3-buten-1-ol directly with a formaldehyde source and an organic sulfonic acid, the process achieves the formation of tetrahydropyranyl-4-sulfonate in a single operational unit. Experimental data from the patent demonstrates that this method can achieve yields as high as 86 percent when using paraformaldehyde and methanesulfonic acid in toluene at mild temperatures. This dramatic improvement in efficiency eliminates the isolation of the unstable alcohol intermediate, thereby reducing waste generation and processing time. For supply chain heads focused on the commercial scale-up of complex pharmaceutical intermediates, this transition from a low-yielding two-step process to a high-yielding one-pot reaction represents a substantial opportunity to enhance production throughput and ensure consistent supply continuity without the bottlenecks associated with purification of unstable species.
Mechanistic Insights into Acid-Catalyzed Cyclization and Substitution
The core chemical innovation lies in the acid-catalyzed cyclization mechanism that concurrently installs the sulfonate leaving group. In this reaction, the organic sulfonic acid serves a dual role: it acts as a catalyst to promote the electrophilic addition of the formaldehyde derivative to the alkene double bond of 3-buten-1-ol, and simultaneously provides the sulfonate anion for nucleophilic capture. This concerted pathway avoids the formation of free tetrahydropyran-4-ol, which is prone to side reactions, and instead stabilizes the cyclic structure immediately as the sulfonate ester. The choice of solvent, such as toluene or benzene, plays a critical role in managing the solubility of the reagents and facilitating the removal of water generated during the condensation, thus driving the equilibrium towards the desired product. Understanding this mechanism is vital for R&D teams aiming to replicate high-purity OLED material or pharmaceutical intermediate standards, as precise control over the acid-to-alkene ratio and temperature ensures minimal formation of polymeric byproducts.
Following the formation of the sulfonate, the subsequent conversion to 4-aminotetrahydropyran derivatives proceeds via a classic nucleophilic substitution mechanism. The sulfonate group is an excellent leaving group, allowing various amines to displace it under relatively mild thermal conditions ranging from 40°C to 180°C. 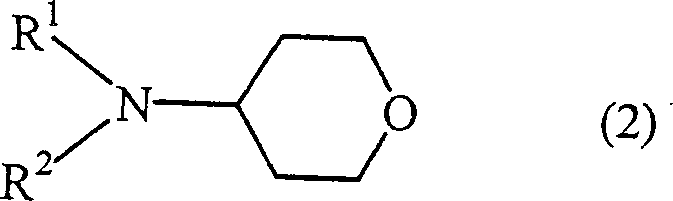 The versatility of this substitution is highlighted by the broad scope of amines compatible with the process, including primary, secondary, and cyclic amines. The structural integrity of the tetrahydropyran ring is maintained throughout this transformation, ensuring that the final products retain the stereochemical and conformational properties required for biological activity. For quality control purposes, the ability to tune the R1 and R2 groups on the nitrogen atom allows for the rapid generation of diverse libraries of compounds, facilitating the discovery of new active pharmaceutical ingredients while maintaining a robust and predictable impurity profile that simplifies downstream purification efforts.
The versatility of this substitution is highlighted by the broad scope of amines compatible with the process, including primary, secondary, and cyclic amines. The structural integrity of the tetrahydropyran ring is maintained throughout this transformation, ensuring that the final products retain the stereochemical and conformational properties required for biological activity. For quality control purposes, the ability to tune the R1 and R2 groups on the nitrogen atom allows for the rapid generation of diverse libraries of compounds, facilitating the discovery of new active pharmaceutical ingredients while maintaining a robust and predictable impurity profile that simplifies downstream purification efforts.
How to Synthesize Tetrahydropyranyl-4-Sulfonate Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and thermal management to maximize the benefits of the one-step design. The process begins by charging a reactor with 3-buten-1-ol and a formaldehyde source, such as paraformaldehyde or trioxane, in a suitable organic solvent like toluene. An organic sulfonic acid, preferably methanesulfonic acid or p-toluenesulfonic acid, is then added dropwise to control the exotherm and ensure uniform mixing. The reaction mixture is typically stirred at temperatures between 20°C and 60°C for several hours to reach completion. Detailed standardized synthesis steps see the guide below.
- Mix 3-buten-1-ol with a formaldehyde derivative (such as paraformaldehyde) and an organic sulfonic acid like methanesulfonic acid in a solvent like toluene.
- Maintain the reaction temperature between 20°C to 60°C under stirring to facilitate the one-step cyclization and sulfonation.
- Purify the resulting tetrahydropyranyl-4-sulfonate via standard workup procedures including extraction and distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented process offers transformative advantages for procurement and supply chain operations, primarily driven by the simplification of the manufacturing workflow. By consolidating two distinct chemical transformations into a single reaction vessel, manufacturers can significantly reduce capital expenditure on equipment and lower the operational overhead associated with intermediate isolation and storage. The elimination of the unstable tetrahydropyran-4-ol intermediate removes a major source of yield loss and safety hazard, leading to a more predictable and reliable production schedule. This stability is particularly beneficial for long-term supply contracts where consistency is paramount, allowing suppliers to offer more competitive pricing structures based on improved material efficiency rather than speculative market fluctuations.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the drastic simplification of the synthetic route, which inherently lowers the cost of goods sold. By avoiding the separate synthesis and purification of the alcohol intermediate, the process saves on solvent usage, energy consumption for heating and cooling cycles, and labor hours required for multiple workups. Furthermore, the high yields achieved in the one-step cyclization mean that less raw material is wasted, directly translating to better atom economy. Although specific percentage savings depend on local utility costs, the qualitative reduction in processing steps ensures a leaner manufacturing operation that is less susceptible to cost overruns common in multi-step syntheses involving unstable intermediates.
- Enhanced Supply Chain Reliability: The reliance on 3-buten-1-ol as a starting material significantly de-risks the supply chain, as this feedstock is widely available and commercially mature compared to specialized ketones or unstable alcohols. The robustness of the reaction conditions, which do not require extreme pressures or cryogenic temperatures, means that production can be maintained consistently across different manufacturing sites without the need for highly specialized infrastructure. This flexibility enhances supply continuity, ensuring that downstream customers receive their orders on time even during periods of high market demand. Additionally, the ability to recover and recycle unreacted amines in the second step further secures the supply of critical reagents, minimizing the impact of external raw material shortages.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward due to the mild reaction parameters and the absence of hazardous high-pressure hydrogenation steps often found in alternative routes. The use of common organic solvents like toluene allows for established waste management protocols to be applied, facilitating compliance with environmental regulations. Moreover, the high selectivity of the reaction reduces the burden on wastewater treatment facilities by minimizing the load of organic byproducts. This environmental efficiency not only supports corporate sustainability goals but also reduces the regulatory friction often associated with scaling up complex chemical processes, enabling faster time-to-market for new products derived from these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthesis technology, derived directly from the comparative data and experimental examples provided in the patent documentation. These insights are intended to clarify the practical benefits and limitations of the process for technical decision-makers evaluating its adoption for their specific manufacturing needs. Understanding these nuances is essential for conducting accurate feasibility assessments and ensuring that the technology aligns with your facility's capabilities and quality standards.
Q: Why is the conventional two-step method for tetrahydropyranyl sulfonates inefficient?
A: Conventional methods involve synthesizing tetrahydropyran-4-ol first, followed by sulfonation, resulting in cumulative yield losses and handling difficulties with unstable intermediates.
Q: What are the advantages of using 3-buten-1-ol as a starting material?
A: 3-Buten-1-ol is readily available and allows for a direct one-step conversion to the sulfonate, significantly simplifying the supply chain and reducing raw material costs.
Q: Can this process be scaled for industrial production of aminotetrahydropyran derivatives?
A: Yes, the process operates under mild conditions (20-60°C) without requiring complex high-pressure equipment, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydropyranyl-4-Sulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in maintaining a competitive edge in the global fine chemicals market. Our team of expert chemists has extensively evaluated the technology described in CN1409707A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative process to life. We are committed to delivering high-purity intermediates that meet stringent purity specifications, utilizing our rigorous QC labs to ensure every batch conforms to the highest industry standards. Our capability to handle complex acid-catalyzed cyclizations and nucleophilic substitutions ensures that we can reliably supply the tetrahydropyranyl scaffolds necessary for your next-generation pharmaceutical and agrochemical projects.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your specific product requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current supply chain challenges. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your production efficiency and reduce overall costs. Let us be your partner in transforming complex chemical challenges into commercial successes through superior technology and unwavering commitment to quality.
