Scalable Synthesis of 2-Chloro-7H-Pyrrolo[2,3-d]Pyrimidine via Selective Dechlorination
Scalable Synthesis of 2-Chloro-7H-Pyrrolo[2,3-d]Pyrimidine via Selective Dechlorination
The pharmaceutical industry's relentless pursuit of potent kinase inhibitors has placed a premium on high-quality heterocyclic building blocks, specifically 2-chloro-7H-pyrrolo[2,3-d]pyrimidine derivatives. As detailed in the groundbreaking patent CN102633802B, a novel synthetic methodology has emerged that fundamentally alters the economic and operational landscape for producing this critical scaffold. This technology addresses the longstanding bottlenecks of low yields, difficult purification, and hazardous reagents that have plagued traditional manufacturing processes. By leveraging a strategic protection-deprotection sequence coupled with selective catalytic hydrogenation, this approach delivers a robust pathway suitable for the rigorous demands of modern API supply chains. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloro-7H-pyrrolo[2,3-d]pyrimidine has been fraught with significant technical and economic challenges that hinder efficient commercialization. Prior art methods, such as those disclosed in WO2010/007114, often rely on complex reduction strategies utilizing expensive catalyst systems like sodium borohydride combined with palladium and bulky phosphine ligands such as BINAP. These legacy processes not only incur exorbitant raw material costs but also necessitate sophisticated purification techniques, frequently requiring preparative high-performance liquid chromatography (HPLC) to isolate the desired product from complex reaction mixtures. Furthermore, alternative routes involving Stille couplings with organotin reagents introduce severe toxicity concerns and environmental liabilities, complicating waste management and regulatory compliance. The cumulative effect of these inefficiencies is a process with low overall yields and poor scalability, rendering it unsuitable for the high-volume production required by the global pharmaceutical market.
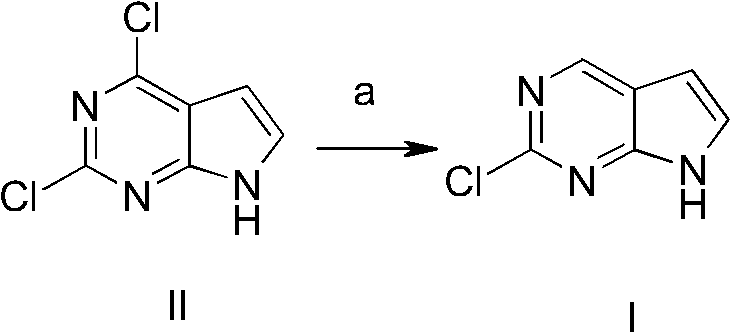
The Novel Approach
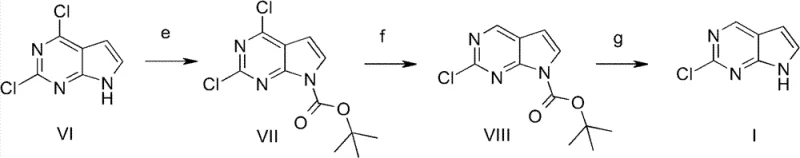
In stark contrast to these cumbersome legacy methods, the innovative strategy outlined in patent CN102633802B introduces a streamlined three-step sequence that prioritizes operational simplicity and chemical efficiency. The core of this breakthrough lies in the initial protection of the pyrrole nitrogen atom using di-tert-butyl dicarbonate, which effectively masks the reactive site and enables subsequent selective transformations. This is followed by a highly selective dechlorination step under mild atmospheric hydrogenation conditions, which precisely removes the unwanted chloro-substituent while preserving the critical 2-chloro functionality needed for downstream coupling reactions. The final deprotection step cleanly reveals the target molecule with exceptional purity, eliminating the need for labor-intensive chromatographic separations. This logical progression from protection to selective reduction and finally deprotection represents a paradigm shift in cost reduction in API manufacturing, transforming a previously difficult synthesis into a robust, industrial-grade operation.
Mechanistic Insights into Selective Catalytic Dechlorination
The heart of this synthetic innovation is the mechanistic precision achieved during the catalytic hydrogenation step, where the differentiation between the 2-chloro and 4-chloro positions on the pyrrolo-pyrimidine ring is paramount. The presence of the tert-butoxycarbonyl (Boc) protecting group on the pyrrole nitrogen plays a crucial electronic and steric role, modulating the electron density of the heterocyclic system to favor the reduction of the C4-Cl bond over the C2-Cl bond. Under atmospheric hydrogen pressure with a palladium on carbon catalyst, the reaction proceeds through a surface-mediated mechanism where the accessibility of the chlorine atoms to the active catalytic sites is key. The Boc group likely creates a steric environment or electronic distribution that renders the C4 position more susceptible to oxidative addition and subsequent reductive elimination of hydrogen chloride. This selectivity is vital because non-selective reduction would lead to the fully dechlorinated byproduct, drastically reducing yield and complicating purification. By fine-tuning reaction parameters such as solvent choice (e.g., ethyl acetate or THF) and the presence of acid scavengers like triethylamine, the process ensures that the reaction stops selectively at the mono-dechlorinated stage.
Furthermore, the mechanism of the protection and deprotection steps is designed to minimize side reactions and maximize throughput. The initial acylation of the pyrrole nitrogen using di-tert-butyl dicarbonate is facilitated by nucleophilic catalysts like DMAP, which accelerate the formation of the carbamate bond under mild alkaline conditions. This step is reversible, allowing for the final cleavage of the protecting group using strong acids such as trifluoroacetic acid or concentrated hydrochloric acid. The cleavage mechanism involves protonation of the carbamate oxygen, leading to the formation of a tert-butyl cation and the release of carbon dioxide, thereby regenerating the free NH group of the pyrrole ring. This chemical logic ensures that the intermediate remains stable throughout the hydrogenation process but can be rapidly converted to the final product under controlled acidic conditions. Such mechanistic control is essential for maintaining a clean impurity profile, ensuring that the final high-purity pharmaceutical intermediate meets the stringent specifications required for clinical and commercial drug substance production.
How to Synthesize 2-Chloro-7H-Pyrrolo[2,3-d]Pyrimidine Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and reaction conditions to replicate the high yields reported in the patent data. The process begins with the dissolution of the starting dichloro-material in a suitable organic solvent, followed by the controlled addition of the protecting group reagent and base. Once the protected intermediate is isolated, typically via crystallization, it undergoes the critical hydrogenation step where monitoring hydrogen uptake is essential to prevent over-reduction. The final step involves careful pH control during the acid workup to ensure complete precipitation of the product while removing inorganic salts. For a detailed breakdown of the specific molar ratios, temperatures, and workup procedures that guarantee success, please refer to the standardized protocol below.
- Protect the pyrrole nitrogen of 2,4-dichloro-7H-pyrrolo[2,3-d]pyrimidine using di-tert-butyl dicarbonate (Boc2O) with a base like triethylamine and DMAP catalyst to form the Boc-protected intermediate.
- Perform selective dechlorination under atmospheric hydrogen pressure using a palladium on carbon (Pd/C) catalyst to remove the 4-chloro group while retaining the 2-chloro substituent.
- Remove the Boc protecting group using an acid such as trifluoroacetic acid (TFA) or concentrated hydrochloric acid to yield the final 2-chloro-7H-pyrrolo[2,3-d]pyrimidine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers profound advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By eliminating the need for expensive transition metal ligands and toxic organotin reagents, the raw material costs are significantly reduced, making the final intermediate much more price-competitive in the global market. Moreover, the shift from complex chromatographic purification to simple crystallization and filtration drastically cuts down on solvent consumption and processing time, leading to substantial operational savings. This efficiency translates into a more reliable supply chain, as the simplified process is less prone to batch-to-batch variability and equipment bottlenecks. For procurement managers, this means securing a source of commercial scale-up of complex pharmaceutical intermediates that is both economically viable and environmentally sustainable.
- Cost Reduction in Manufacturing: The elimination of costly catalysts like BINAP and the removal of preparative HPLC purification steps result in a drastic simplification of the production workflow. This reduction in complexity lowers the barrier to entry for large-scale manufacturing, allowing for significant margin improvements without compromising on quality standards. The use of common, commodity chemicals for protection and deprotection further insulates the process from volatile raw material pricing, ensuring stable long-term costing models for budget planning.
- Enhanced Supply Chain Reliability: The reliance on atmospheric hydrogenation and standard heterogeneous catalysts means that the process can be executed in a wide range of manufacturing facilities without requiring specialized high-pressure equipment. This flexibility enhances supply continuity, as production can be easily scaled or shifted between different sites if necessary. Additionally, the high purity achieved through recrystallization reduces the risk of downstream failures in API synthesis, ensuring a steady flow of qualified material for drug development pipelines.
- Scalability and Environmental Compliance: The avoidance of toxic organotin byproducts and the reduction of solvent waste through crystallization-based purification align perfectly with modern green chemistry principles. This makes the process easier to permit and operate within strict environmental regulations, reducing the risk of shutdowns due to compliance issues. The robust nature of the reaction conditions allows for seamless scaling from pilot plant quantities to multi-ton commercial production, supporting the growing demand for kinase inhibitor therapies globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent's experimental data and comparative analysis. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for their specific production needs. The answers provided reflect the proven capabilities of the technology to overcome traditional synthesis hurdles.
Q: How does this new method improve purification compared to prior art?
A: Unlike previous methods requiring preparative HPLC or column chromatography, this novel route allows for simple recrystallization and filtration, significantly reducing solvent waste and processing time.
Q: What are the cost advantages of the selective dechlorination step?
A: The process utilizes atmospheric hydrogenation with standard Pd/C catalysts instead of expensive ligand-based systems or toxic organotin reagents, drastically lowering raw material costs and safety hazards.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the mild reaction conditions (20-60°C, atmospheric pressure) and high total yield of approximately 63% make it highly feasible for commercial scale-up without complex engineering controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-7H-Pyrrolo[2,3-d]Pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102633802B are fully realized in practical manufacturing settings. We are committed to delivering materials with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our capability to handle complex heterocyclic chemistry allows us to offer a secure supply of this vital intermediate, mitigating the risks associated with sourcing from less experienced vendors.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a partnership that drives both innovation and efficiency in your supply chain.
