Advanced Synthesis of 1-Boc-4,5-Pyrroline-2-Carboxylates for High-Value Pharmaceutical Intermediates
Advanced Synthesis of 1-Boc-4,5-Pyrroline-2-Carboxylates for High-Value Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust and scalable pathways for constructing complex heterocyclic scaffolds, particularly those serving as critical building blocks for antibiotics and antiviral agents. Patent CN103073475A introduces a transformative preparation method for 1-Boc-4,5-pyrroline-2-carboxyl compounds, addressing long-standing bottlenecks in the synthesis of this vital class of medical intermediates. These compounds are not merely academic curiosities; they serve as essential segments in the biosynthesis of Virginiamycin, a potent streptogramin antibiotic effective against gram-positive microorganisms, and act as key precursors for integrin VLA-4 antagonists used in treating asthma and multiple sclerosis. The disclosed technology leverages a strategic protecting group approach combined with low-temperature lithiation to achieve high yields under significantly milder conditions than previously reported methods.
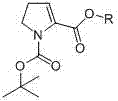
This innovation represents a pivotal shift from reliance on scarce, expensive precursors to the utilization of abundant, cost-effective starting materials like 1-Boc-3-hydroxy pyrrolidine. By establishing a unified synthetic platform where a single protected intermediate can be divergently functionalized into ethyl esters, tert-butyl esters, free acids, or benzyl esters, the patent offers a versatile solution for process chemists. The ability to access these dehydro-proline derivatives efficiently opens new avenues for the rapid assembly of peptidomimetics and macrocyclic antibiotics, ensuring a more reliable supply chain for downstream drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of 1-Boc-4,5-pyrroline-2-carboxylates was plagued by severe operational and economic constraints that hindered industrial adoption. As documented in the Russian Journal of Organic Chemistry (2008), existing protocols often necessitated extreme cryogenic conditions, specifically requiring reaction temperatures below -90°C to control regioselectivity and prevent byproduct formation. Maintaining such ultra-low temperatures on a multi-kilogram or tonne scale imposes immense energy costs and requires specialized refrigeration equipment that is not standard in many fine chemical facilities. Furthermore, the European Journal of Organic Chemistry (2009) described alternative routes that relied on raw materials which were not only prohibitively expensive but also difficult to source in bulk quantities, creating a fragile supply chain vulnerable to market fluctuations.
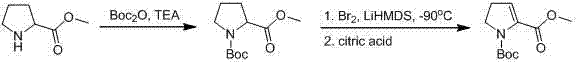
The complexity of these legacy methods often involved multi-step sequences with poor atom economy, leading to significant waste generation and lower overall yields. The reliance on harsh reagents and unstable intermediates at sub-zero temperatures increased the risk of safety incidents and made process validation a daunting task for quality assurance teams. Consequently, the market availability of high-purity 1-Boc-4,5-pyrroline-2-carboxyl compounds remained limited, driving up prices and restricting their application in the development of novel therapeutic candidates. These factors collectively created a pressing need for a methodology that could balance chemical efficiency with practical manufacturability.
The Novel Approach
The methodology outlined in CN103073475A circumvents these historical challenges by introducing a streamlined two-step sequence initiated from the readily available 1-Boc-3-hydroxy pyrrolidine. The core innovation lies in the strategic protection of the 3-hydroxyl group using tert-butyl dimethyl silyl (TBS) or methoxymethyl (MOM) groups, which serves a dual purpose: it prevents unwanted side reactions at the hydroxyl position and directs the subsequent lithiation to the desired 2-position of the pyrrolidine ring. This protective strategy allows the use of isobutyl-lithium at a much more manageable -78°C, a temperature easily achievable with standard dry ice-acetone baths, thereby drastically reducing the energy footprint and equipment complexity associated with the synthesis.
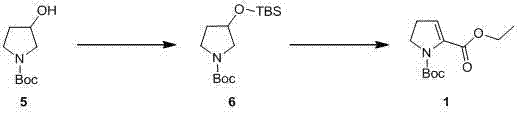
Following the lithiation step, the protocol demonstrates remarkable versatility by allowing the introduction of various functional groups through simple electrophilic quenching. Whether the target is an ethyl ester via reaction with vinyl chloroformate, a tert-butyl ester using Boc anhydride, a carboxylic acid via carbon dioxide insertion, or a benzyl ester using CbzCl, the same protected intermediate serves as the universal precursor. This modularity simplifies inventory management and reduces the need for maintaining multiple distinct synthetic lines for different derivatives. The result is a robust, high-yielding process that transforms a cheap commodity chemical into a high-value pharmaceutical intermediate with minimal operational friction.
Mechanistic Insights into Regioselective Lithiation and Protecting Group Effects
The success of this synthetic route hinges on the precise interplay between the N-Boc group and the 3-position protecting group (TBS or MOM) in directing the metalation event. In the absence of the 3-hydroxyl protection, lithiation of N-Boc pyrrolidines can often lead to mixtures of regioisomers or decomposition due to the acidity of the hydroxyl proton. By converting the hydroxyl group into a bulky silyl ether or a stable methyl ether, the molecule adopts a conformation that favors deprotonation at the C2 position adjacent to the nitrogen. The isobutyl-lithium reagent, being a strong base, selectively abstracts the proton at C2, generating a stabilized carbanion that is resonance-stabilized by the adjacent carbamate nitrogen. This regioselectivity is critical for ensuring that the subsequent double bond formation occurs exclusively between the C4 and C5 positions during the elimination or functionalization phase, preserving the integrity of the pyrroline scaffold.
Furthermore, the choice of electrophile in the quenching step dictates the final oxidation state and functionality of the product without altering the core cyclization mechanism. For instance, when vinyl chloroformate is employed, the lithiated species attacks the carbonyl carbon, followed by an elimination sequence that establishes the 4,5-unsaturation characteristic of the target pyrroline. In the case of carbon dioxide quenching, the organolithium intermediate acts as a nucleophile to form the carboxylate salt directly, which upon acidic workup yields the free acid. This mechanistic understanding allows process chemists to fine-tune reaction parameters, such as the rate of electrophile addition and stirring efficiency, to maximize conversion and minimize the formation of over-lithiated byproducts. The consistent yields observed across different electrophiles (ranging from 53% to nearly 80%) underscore the reliability of this mechanistic pathway.
How to Synthesize 1-Boc-4,5-pyrroline-2-carboxylates Efficiently
The execution of this synthesis requires strict adherence to anhydrous conditions and temperature control to ensure optimal performance of the organolithium reagent. The process begins with the protection of the starting hydroxy-pyrrolidine, followed by the critical lithiation step which must be performed under an inert atmosphere to prevent quenching by moisture or oxygen. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are essential for maintaining batch-to-batch consistency. The following guide outlines the critical operational phases derived from the patent embodiments.
- Protect the 3-hydroxyl group of 1-Boc-3-hydroxy pyrrolidine using TBSCl or MOM-Cl under basic conditions to form the silyl or ether protected intermediate.
- Perform regioselective lithiation at the 2-position using isobutyl-lithium at -78°C in anhydrous THF to generate the reactive organolithium species.
- Quench the lithiated intermediate with specific electrophiles such as vinyl chloroformate, Boc anhydride, dry ice, or benzyl chloroformate to yield the target 4,5-dehydro derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond simple chemical yield. By shifting the raw material base from obscure, high-cost precursors to commodity chemicals like 1-Boc-3-hydroxy pyrrolidine, manufacturers can significantly decouple their production costs from volatile specialty chemical markets. The simplification of the reaction conditions, specifically the move from -90°C to -78°C, translates directly into reduced utility costs and allows for the utilization of standard reactor fleets without the need for custom cryogenic engineering. This operational flexibility enhances the resilience of the supply chain, ensuring that production schedules are not disrupted by equipment limitations or excessive energy demands.
- Cost Reduction in Manufacturing: The elimination of expensive and hard-to-source starting materials fundamentally alters the cost structure of producing these intermediates. By utilizing a common protected intermediate that can be diverted into multiple product streams (esters, acids), facilities can achieve economies of scale that were previously unattainable with single-product dedicated lines. The removal of extreme cryogenic requirements further lowers the barrier to entry for contract manufacturing organizations, fostering a more competitive pricing environment for downstream API producers seeking reliable partners.
- Enhanced Supply Chain Reliability: The reliance on readily available reagents such as TBSCl, imidazole, and isobutyl-lithium ensures that raw material procurement is straightforward and less prone to geopolitical or logistical disruptions. The robustness of the two-step sequence minimizes the number of unit operations required, thereby reducing the cumulative risk of batch failures and delays. This streamlining allows for faster turnaround times from order to delivery, enabling pharmaceutical companies to accelerate their own development timelines for antibiotics and antiviral therapies without being bottlenecked by intermediate shortages.
- Scalability and Environmental Compliance: The process generates fewer byproducts and avoids the use of hazardous reagents associated with older elimination methods, aligning with modern green chemistry principles. The simplified workup procedures, often involving standard aqueous extractions and chromatography, facilitate easier waste management and solvent recovery. This environmental compatibility is crucial for maintaining regulatory compliance in increasingly stringent jurisdictions, ensuring long-term operational continuity without the threat of shutdowns due to environmental violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity for R&D and procurement teams evaluating this route for potential adoption.
Q: How does this new synthesis method improve upon previous Russian and European protocols?
A: Unlike the Russian method requiring extreme cryogenic conditions below -90°C which poses significant safety and equipment challenges, this patent utilizes a manageable -78°C lithiation temperature. Furthermore, it avoids the expensive and scarce raw materials cited in European literature, utilizing readily available 1-Boc-3-hydroxy pyrrolidine instead.
Q: What is the structural scope of compounds accessible via this lithiation strategy?
A: The method demonstrates high versatility, allowing for the synthesis of various esters (ethyl, tert-butyl, benzyl) and the free carboxylic acid. By simply changing the electrophile in the second step—such as vinyl chloroformate, Boc anhydride, CO2, or CbzCl—manufacturers can access a diverse library of 1-Boc-4,5-pyrroline-2-carboxyl derivatives from a single common intermediate.
Q: Is the starting material 1-Boc-3-hydroxy pyrrolidine commercially viable for large-scale production?
A: Yes, the patent explicitly highlights that the starting raw material is cheap and easy to obtain compared to precursors used in prior art. This availability, combined with the short two-step reaction sequence from the protected intermediate, significantly enhances the economic feasibility for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Boc-4,5-pyrroline-2-carboxylates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity 1-Boc-4,5-pyrroline-2-carboxylates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and security for your supply chain.
