Revolutionizing Aromatic Amide Synthesis: Electrochemical C-H Activation for Commercial Scale Production
Introduction to Patent CN111270260A
The landscape of organic synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, driven by the urgent need to reduce environmental impact and operational costs in the fine chemical industry. Patent CN111270260A, published in June 2020, presents a groundbreaking approach to the ortho-alkenylation of aromatic amide compounds, a critical transformation in the construction of complex molecular scaffolds for pharmaceuticals and agrochemicals. Unlike traditional methods that rely heavily on stoichiometric chemical oxidants, this invention leverages weak electric current as a clean, traceless oxidant to drive a Rhodium(III)-catalyzed C-H activation process. This electrochemical strategy not only circumvents the generation of hazardous metal waste but also operates efficiently in an aqueous medium, marking a significant advancement in atom economy and process safety. For R&D directors and process chemists, this technology offers a robust alternative to classical Heck reactions, providing high regioselectivity and moderate-to-excellent yields under mild conditions. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative protocols is essential for maintaining competitiveness in a market that increasingly values sustainability and efficiency.
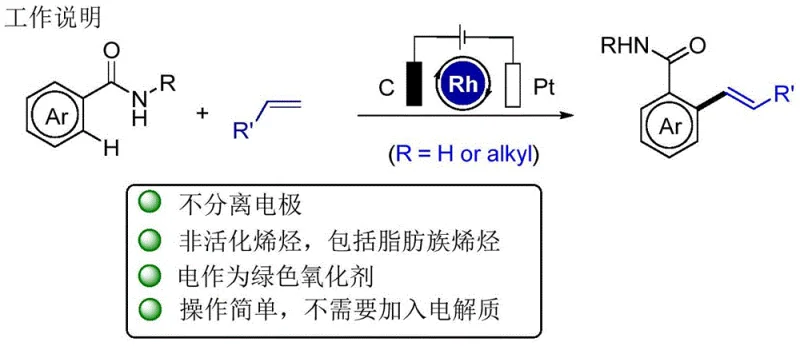
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of aromatic rings via C-H activation has been dominated by transition metal-catalyzed reactions that necessitate the use of substantial quantities of inorganic or organic oxidants to regenerate the active catalytic species. As illustrated in the general reaction scheme below, these conventional directing group-assisted processes often require harsh oxidative conditions and produce significant amounts of waste metals, posing severe challenges for environmental compliance and waste management. The reliance on stoichiometric oxidants such as silver salts, copper salts, or hypervalent iodine reagents not only inflates the raw material costs but also complicates the downstream purification processes due to the presence of metal residues. Furthermore, many of these traditional protocols demand anhydrous conditions and specialized solvents, limiting their applicability in large-scale manufacturing where safety and cost are paramount concerns. The accumulation of toxic byproducts and the difficulty in recycling expensive noble metal catalysts have long been bottlenecks in the commercial scale-up of complex pharmaceutical intermediates.

The Novel Approach
In stark contrast to these legacy methods, the electrochemical protocol disclosed in CN111270260A redefines the oxidative power source by utilizing electrons directly from an external circuit. This novel approach dissolves aromatic amides and non-activated olefins in a water-compatible solvent system, employing a weak current to facilitate the catalytic cycle without the need for external chemical oxidants. The reaction design cleverly integrates the anodic oxidation of the reduced Rh(I) species back to the active Rh(III) state, ensuring a continuous and efficient catalytic turnover. By operating in a mixture of tert-amyl alcohol and water, the method capitalizes on the inherent conductivity of the aqueous phase, thereby eliminating the necessity for additional supporting electrolytes which often complicate product isolation. This shift from chemical to electrochemical oxidation represents a fundamental improvement in green chemistry metrics, offering a cleaner reaction profile with simplified workup procedures. For procurement managers, this translates to a streamlined supply chain where the dependency on volatile oxidant markets is removed, enhancing overall process stability and cost predictability.
Mechanistic Insights into Rhodium-Catalyzed Electrochemical C-H Activation
The mechanistic elegance of this transformation lies in the seamless integration of organometallic catalysis with electrochemistry. The cycle initiates with the formation of an active Cp*Rh(III) species in the presence of a carboxylate additive, which subsequently coordinates with the aromatic amide substrate via the directing group to activate the ortho-C-H bond, forming a stable rhodacycle intermediate A. Following this activation, the non-activated olefin undergoes migratory insertion into the Rh-C bond, generating a new alkyl-rhodium species C. The critical step of beta-hydride elimination then releases the desired alkenylated product while reducing the metal center to a Rh(I) state. In traditional thermal catalysis, this Rh(I) species would require a chemical oxidant to return to Rh(III); however, in this electrochemical system, the Rh(I) intermediate is anodically oxidized at the graphite felt electrode surface, regenerating the active Rh(III) catalyst and closing the loop. This anodic regeneration is the cornerstone of the method's sustainability, as it replaces chemical waste with electrical energy. Understanding this cycle is vital for R&D teams aiming to optimize reaction parameters such as current density and electrode material to maximize turnover numbers and minimize energy consumption.
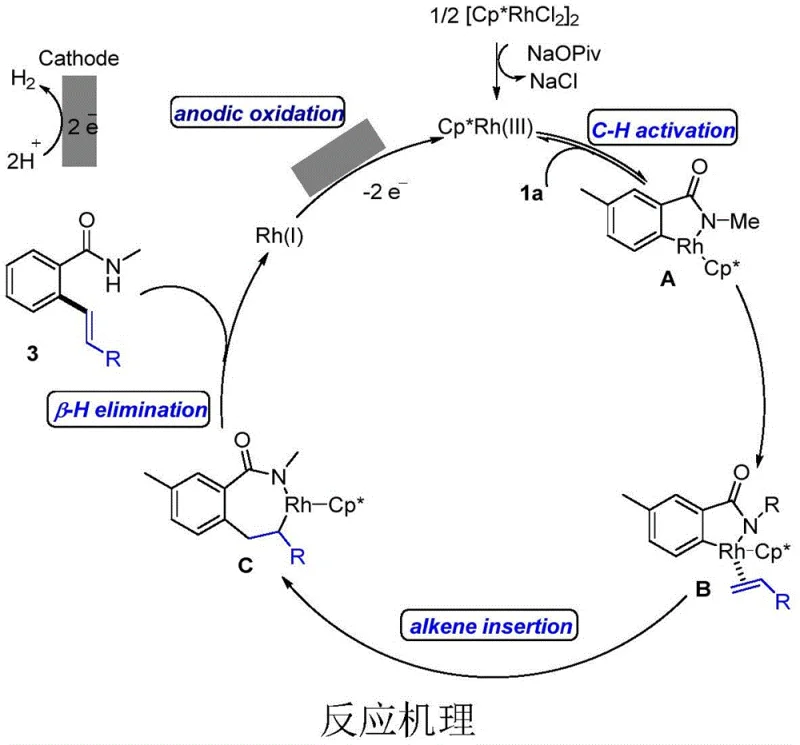
Furthermore, the control of impurities in this electrochemical system is inherently superior due to the mildness of the oxidative potential applied. Unlike strong chemical oxidants that can indiscriminately attack sensitive functional groups on the substrate, the controlled potential at the anode selectively targets the metal center for regeneration. This selectivity ensures that the integrity of the aromatic amide and the olefin partners is preserved, leading to a cleaner crude reaction mixture with fewer side products. The use of water as a co-solvent also plays a crucial role in stabilizing charged intermediates and facilitating proton transfer steps, which contributes to the high regioselectivity observed in the ortho-position. For quality control laboratories, this means that the burden of removing complex oxidative byproducts is significantly reduced, allowing for more straightforward analytical characterization and faster release of high-purity batches. The mechanistic robustness of this Rh-catalyzed cycle underscores its potential as a versatile platform for synthesizing diverse libraries of bioactive molecules.
How to Synthesize Ortho-Alkenylated Aromatic Amides Efficiently
Implementing this electrochemical synthesis route requires careful attention to reactor configuration and parameter optimization to ensure reproducibility and safety. The patent outlines a straightforward procedure where the aromatic amide, olefin, and sodium pivalate additive are dissolved in a specific ratio of tert-amyl alcohol and water, creating a conductive medium that supports the electrochemical process without extra salts. The choice of electrode materials is critical, with graphite felt serving as the anode to provide a high surface area for oxidation and platinum acting as the cathode for efficient hydrogen evolution. Maintaining a constant current of approximately 4 mA at a temperature of 100°C allows the reaction to proceed to completion within 12 to 24 hours, as monitored by TLC. While the laboratory-scale protocol is well-defined, scaling this process requires adapting the electrode surface area and current density to maintain the same electrochemical environment. The detailed standardized synthesis steps for replicating this high-yielding transformation are provided in the guide below.
- Dissolve aromatic amide, non-activated olefin, and NaOPiv additive in a t-AmOH/H2O solvent mixture.
- Add [Cp*RhCl2]2 catalyst and set up the electrochemical cell with a Graphite Felt anode and Platinum cathode.
- Apply a constant current of 4 mA at 100°C for 12-24 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrochemical alkenylation technology offers profound advantages for procurement and supply chain operations, primarily driven by the simplification of the input material list and waste management protocols. By replacing stoichiometric chemical oxidants with electricity, manufacturers can drastically reduce the volatility of their raw material costs, as electrical energy is generally more stable in price and availability compared to specialized reagents like silver carbonate or copper acetate. This shift also mitigates the risks associated with the storage and handling of hazardous oxidizing agents, enhancing workplace safety and reducing insurance and compliance overheads. For supply chain heads, the ability to run this reaction in an aqueous medium simplifies solvent recovery and reduces the volume of organic waste requiring disposal, leading to substantial cost savings in waste treatment facilities. The elimination of heavy metal waste from chemical oxidants further streamlines the purification workflow, potentially shortening the overall production cycle time and improving throughput capacity without compromising product quality.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the removal of expensive stoichiometric oxidants and the reduction of catalyst loading through efficient electrochemical recycling. Traditional methods often require greater than one equivalent of oxidant, which represents a direct material cost that is completely eliminated here. Additionally, the use of water as a co-solvent reduces the consumption of expensive anhydrous organic solvents, lowering the overall solvent purchase and recovery costs. The simplified workup, which avoids the need for extensive metal scavenging treatments, further reduces the operational expenditure associated with downstream processing. These factors combine to create a leaner manufacturing process that is highly attractive for cost-sensitive large-volume production of fine chemicals.
- Enhanced Supply Chain Reliability: Relying on electricity as a reagent decouples the production process from the supply chain fluctuations of niche chemical oxidants, which can sometimes face shortages or quality inconsistencies. The substrates used, such as aromatic amides and styrenes, are commodity chemicals with robust global supply networks, ensuring consistent availability. The robustness of the electrochemical setup, which tolerates the presence of water and does not require rigorous exclusion of air, makes the process less sensitive to environmental variations, thereby reducing the risk of batch failures. This reliability is crucial for maintaining steady delivery schedules to downstream customers in the pharmaceutical and agrochemical sectors, fostering stronger long-term partnerships.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the feasibility of gram-scale synthesis, proving that the reaction kinetics and mass transfer issues can be managed effectively beyond the milligram scale. The use of graphite felt electrodes is particularly advantageous for scale-up, as this material is inexpensive, chemically inert, and available in large dimensions suitable for industrial electrochemical cells. From an environmental standpoint, the process aligns perfectly with increasingly stringent global regulations on heavy metal discharge and organic solvent emissions. By minimizing waste generation and utilizing a greener solvent system, manufacturers can easily meet ISO 14001 standards and other environmental certifications, avoiding potential fines and reputational damage while appealing to eco-conscious clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this electrochemical alkenylation technology, derived directly from the experimental data and specifications within the patent documentation. These insights are intended to clarify the practical aspects of the reaction for technical teams evaluating its adoption. Understanding these details is key to assessing the feasibility of integrating this method into existing production lines or R&D pipelines for new drug candidates.
Q: What are the advantages of using electricity over chemical oxidants in this Heck-type reaction?
A: Using electricity eliminates the need for stoichiometric amounts of expensive and hazardous chemical oxidants (like Ag or Cu salts), significantly reducing waste generation and raw material costs while simplifying the purification process.
Q: Is this electrochemical method scalable for industrial production?
A: Yes, the patent demonstrates successful gram-scale synthesis (Example 4) with maintained yields around 70%, indicating strong potential for commercial scale-up using standard electrochemical flow or batch reactors.
Q: Does this method require additional electrolytes?
A: No, the method utilizes a water-containing solvent system (t-AmOH/H2O) which provides sufficient conductivity, removing the need for extra supporting electrolytes and further reducing downstream separation complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Amide Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of high-value pharmaceutical intermediates. Our technical team has extensively analyzed the capabilities of Patent CN111270260A and is fully prepared to leverage this Rh-catalyzed C-H activation technology to deliver superior ortho-alkenylated products. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory curiosity to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of monitoring the specific purity specifications required for these complex molecules, guaranteeing that every batch meets the stringent standards expected by global innovator companies.
We invite you to collaborate with us to explore how this green chemistry approach can optimize your specific supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how switching to this electrochemical method can improve your margins. Please contact our technical procurement team today to request specific COA data for similar structures and to discuss route feasibility assessments for your upcoming projects. Together, we can build a more sustainable and cost-effective future for fine chemical manufacturing.
