Scalable Synthesis of Borane Methyl Tert-Butylphosphine Hydrogen for Industrial Catalysis
The chemical industry is witnessing a significant paradigm shift in the production of phosphochiral intermediates, driven by the urgent need for safer, more economical, and scalable synthetic routes. Patent CN108409784B, published in July 2020, introduces a groundbreaking preparation method for borane methyl tert-butylphosphine hydrogen, a critical precursor for high-performance chiral ligands. This technology fundamentally alters the supply chain dynamics for pharmaceutical and agrochemical manufacturers by replacing hazardous, expensive starting materials with ubiquitous industrial commodities like phosphorus trichloride. For R&D directors and procurement strategists, this patent represents a viable pathway to reduce dependency on fragile supply chains associated with specialized organophosphorus reagents. The methodology outlined in this intellectual property not only streamlines the synthetic sequence but also enhances operational safety profiles, making it an ideal candidate for large-scale commercial adoption in the fine chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of borane methyl tert-butylphosphine hydrogen has been plagued by severe technical and economic bottlenecks that hindered its widespread industrial application. Early methodologies, such as those reported by Imamoto in 2000, relied heavily on the use of thiol-based chiral inducing reagents and required the cleavage of C-S bonds using lithium naphthalene, a reagent that is notoriously difficult to handle on a multi-kilogram scale due to its pyrophoric nature and high cost. Furthermore, alternative routes proposed in 2002 utilized borane tert-butylphosphine as a starting material, which is inherently unstable, dangerous to prepare, and poses significant safety risks during storage and transport. Other approaches involving the direct reaction of tert-butylphosphine dichloride with methyl Grignard reagents suffered from poor selectivity, often leading to over-alkylation and complex mixtures that were arduous to purify. These legacy processes resulted in low atom economy, generated substantial hazardous waste, and created unpredictable lead times for downstream users of chiral catalysts.
The Novel Approach
In stark contrast to these problematic legacy routes, the novel approach detailed in CN108409784B leverages a robust four-step sequence initiated by the abundant feedstock phosphorus trichloride. This strategy ingeniously bypasses the need for dangerous phosphane precursors by constructing the phosphorus-carbon framework through controlled Grignard substitutions and esterifications. The process introduces a stable ethyl tert-butylphosphinate intermediate, which serves as a pivotal junction for introducing the methyl group with high regioselectivity. By shifting the chemistry towards stable phosphine oxides prior to the final reduction step, the method effectively mitigates the risks associated with handling air-sensitive free phosphines until the very last stage. This structural redesign of the synthetic pathway ensures that the most hazardous transformations are confined to controlled laboratory conditions or managed via stable intermediates, thereby drastically improving the feasibility of commercial scale-up.
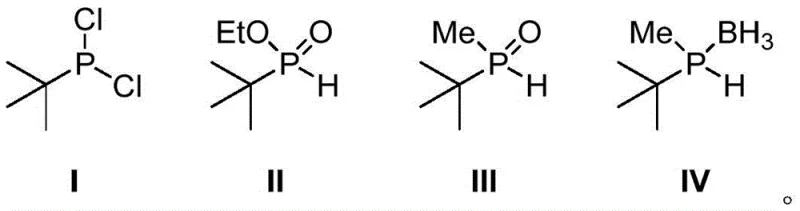
Mechanistic Insights into Grignard Substitution and Borane Protection
The core mechanistic advantage of this technology lies in the strategic manipulation of phosphorus oxidation states and leaving groups to ensure high fidelity in bond formation. The initial substitution of phosphorus trichloride with tert-butyl magnesium chloride exploits the high electrophilicity of the phosphorus center to install the bulky tert-butyl group, forming tert-butyl phosphine dichloride. Subsequent esterification with ethanol converts the remaining reactive P-Cl bonds into a P-OEt moiety, creating ethyl tert-butylphosphinate. This ester functionality is crucial because it acts as a masked electrophile that reacts cleanly with methyl Grignard reagents to form the P-C bond without the competing side reactions observed in dichloride systems. The resulting methyl tert-butyl phosphine oxide is thermodynamically stable and resistant to oxidation, allowing for isolation and quality control before the final reduction. This stepwise control over reactivity prevents the formation of di-methylated byproducts and ensures that the stereochemical integrity required for subsequent chiral ligand synthesis is preserved throughout the sequence.
Furthermore, the final transformation involves a sophisticated one-pot reduction and boranation protocol that converts the phosphine oxide directly into the borane-protected phosphine. This tandem operation eliminates the need to isolate the highly air-sensitive free phosphine intermediate, which would otherwise require inert atmosphere glovebox techniques. The reduction agents, such as borane-THF complexes or mixed hydride systems like LiAlH4-NaBH4-CeCl3, facilitate the deoxygenation of the P=O bond while simultaneously capping the resulting P-H bond with a borane group. This protective group strategy not only stabilizes the molecule against spontaneous oxidation but also simplifies the purification process, as the borane adducts often exhibit distinct crystallization properties compared to their unprotected counterparts. The ability to perform this conversion in a single vessel significantly reduces solvent consumption and processing time, contributing to the overall green chemistry metrics of the process.
How to Synthesize Borane Methyl Tert-Butylphosphine Hydrogen Efficiently
Implementing this synthesis requires precise control over reaction temperatures and stoichiometry to maximize yield and minimize impurity profiles. The process begins with the careful addition of Grignard reagents to phosphorus trichloride at controlled temperatures to manage exotherms, followed by a room temperature esterification that can utilize the alcohol reactant as the solvent itself. The methylation step demands anhydrous conditions and specific solvent choices like THF or ether to maintain Grignard reactivity, while the final reduction step offers flexibility in reagent selection depending on available infrastructure. Detailed standard operating procedures regarding quenching protocols, phase separation techniques, and crystallization parameters are essential for reproducing the high purity levels described in the patent examples. For a comprehensive breakdown of the specific molar ratios, temperature ramps, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- React phosphorus trichloride with tert-butyl magnesium chloride to form tert-butyl phosphine dichloride.
- Perform esterification with ethanol to generate ethyl tert-butylphosphinate.
- Execute methylation using a methyl Grignard reagent to produce methyl tert-butyl phosphine oxide.
- Conduct a one-pot reduction and boranation to yield the final borane-protected product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages that directly address the pain points of procurement managers and supply chain directors in the fine chemical industry. The substitution of exotic, high-cost starting materials with commodity chemicals like phosphorus trichloride and ethanol fundamentally reshapes the cost structure of the final intermediate. By eliminating the reliance on reagents like lithium naphthalene or pre-formed borane phosphines, manufacturers can decouple their production costs from the volatile pricing of niche organometallic suppliers. This shift towards bulk chemical feedstocks ensures a more predictable cost base and reduces the risk of supply disruptions caused by the limited production capacity of specialized reagent vendors. Additionally, the generation of simple inorganic salts and acids as byproducts simplifies waste treatment protocols, potentially lowering environmental compliance costs and facilitating faster regulatory approvals for new manufacturing sites.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the drastic simplification of the raw material basket. By utilizing phosphorus trichloride, a mass-produced industrial chemical, instead of custom-synthesized phosphane derivatives, the direct material costs are significantly lowered. The process avoids the use of expensive chiral auxiliaries or hazardous cleavage reagents that typically inflate the price of chiral intermediates. Moreover, the high atom economy of the route means that a larger proportion of the input mass is converted into the desired product, reducing the effective cost per kilogram of active material. The ability to use the alcohol reactant as the solvent in the esterification step further reduces solvent procurement and recovery costs, contributing to a leaner and more competitive manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved due to the widespread availability of the key reagents. Phosphorus trichloride, ethanol, and common Grignard reagents are produced by multiple global suppliers, eliminating single-source dependencies that often plague the supply of complex organophosphorus compounds. The stability of the intermediate methyl tert-butyl phosphine oxide allows for stockpiling and inventory management strategies that were previously impossible with air-sensitive precursors. This stability enables manufacturers to produce intermediates in large batches and store them for extended periods without degradation, ensuring continuous supply to downstream customers even during fluctuations in demand. The robustness of the process also reduces the likelihood of batch failures, leading to more consistent delivery schedules and stronger contractual reliability.
- Scalability and Environmental Compliance: The operational simplicity of this route makes it highly amenable to scale-up from pilot plant to multi-ton commercial production. The reactions proceed under moderate conditions without the need for extreme cryogenic temperatures or high-pressure equipment, reducing capital expenditure requirements for new production lines. The byproducts generated are primarily inorganic salts and simple organic acids, which are easier to treat and dispose of compared to the complex sulfur-containing waste streams of older methods. This alignment with green chemistry principles facilitates easier permitting and reduces the environmental footprint of the manufacturing process. The one-pot nature of the final reduction step minimizes solvent transfers and exposure risks, enhancing overall plant safety and operational efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to ensure accuracy and relevance for potential licensees or buyers. Understanding these nuances is critical for evaluating the fit of this technology within existing manufacturing portfolios.
Q: Why is the new PCl3-based route superior to previous methods?
A: Unlike prior art requiring dangerous lithium naphthalene or unstable borane tert-butylphosphine, this method utilizes cheap, industrially available phosphorus trichloride and generates stable intermediates.
Q: What are the key purification advantages of this process?
A: The process generates simple inorganic salt byproducts and acids, allowing for straightforward filtration and extraction workups without complex chromatography.
Q: Is the intermediate methyl tert-butyl phosphine oxide stable?
A: Yes, the patent highlights that methyl tert-butyl phosphine oxide is insensitive to air and moisture, significantly simplifying storage and handling compared to free phosphines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Borane Methyl Tert-Butylphosphine Hydrogen Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route for the global supply of chiral ligand precursors. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this chemistry are fully realized in a GMP-compliant environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of borane methyl tert-butylphosphine hydrogen meets the exacting standards required for asymmetric catalysis in pharmaceutical and agrochemical applications. We are committed to leveraging this efficient technology to provide our partners with a secure, high-quality source of this critical intermediate.
We invite forward-thinking organizations to collaborate with us to unlock the value of this innovative process. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how switching to this PCl3-based methodology can optimize your supply chain and enhance your competitive edge in the market.
