Advanced Manufacturing of DpCTA Pigment Intermediates via Safe Glycol Dispersion Technology
Advanced Manufacturing of DpCTA Pigment Intermediates via Safe Glycol Dispersion Technology
The global demand for high-performance organic pigments, particularly quinacridone derivatives, necessitates robust and scalable synthetic routes for their key precursors. Patent CN101844996A introduces a groundbreaking methodology for the preparation of 2,5-di(p-chloroanilino)-terephthalic acid (DpCTA), a critical intermediate in the production of premium red and violet pigments. This technology addresses long-standing inefficiencies in traditional synthesis by replacing volatile organic solvents with a stable ethylene glycol-water dispersion system. For R&D directors and procurement specialists seeking a reliable pigment intermediate supplier, this innovation represents a significant leap forward in process safety, yield optimization, and environmental compliance. By leveraging the unique solubility characteristics of dimethyl succinyl succinate (DMSS) in heated glycol mixtures, the process achieves near-quantitative conversion rates while drastically simplifying downstream purification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of DpCTA has been plagued by significant operational challenges inherent to the use of low-boiling organic solvents such as methanol and ethanol. In these traditional liquid-solid systems, the solubility of the key starting material, DMSS, is severely limited at ambient and moderate temperatures, leading to incomplete reactions and the entrapment of unreacted raw materials within the solid product matrix. Furthermore, the generation of water as a by-product during condensation complicates solvent recovery, often requiring energy-intensive distillation processes that increase the overall carbon footprint. To compensate for poor solubility, conventional methods frequently resort to pressurized operations at lower temperatures, which introduces substantial security risks related to vessel integrity and potential solvent leakage. The resulting heterogeneous reaction mixtures are difficult to stir and control, often yielding products with inconsistent particle size distributions and lower purity profiles that require extensive recrystallization.
The Novel Approach
The innovative process detailed in the patent fundamentally re-engineers the reaction environment by utilizing a dispersion medium composed of ethylene glycol and water. This strategic shift exploits the high boiling point of ethylene glycol to facilitate reaction temperatures between 110 ℃ and 140 ℃ under manageable pressure conditions (0.1 MPa to 0.6 MPa). At these elevated temperatures, DMSS exhibits dramatically increased solubility, allowing it to dissolve completely into the p-chloroaniline phase to form a homogeneous solution prior to the onset of condensation. As the reaction proceeds, the product precipitates as a fine yellow solid suspended in the glycol medium, creating an ideal liquid-solid system that is easy to stir and separate. This transition from a difficult solid-solid or limited solubility regime to a controlled precipitation process ensures thorough reactant contact and maximizes yield.
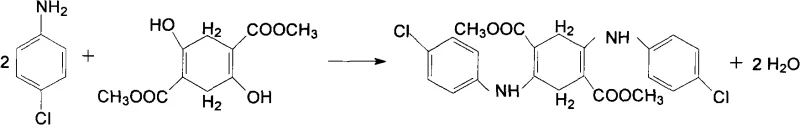
As illustrated in the reaction scheme above, the condensation of DMSS with p-chloroaniline proceeds efficiently under acid catalysis to form the 2,5-di(p-chloroanilino)-3,6-dihydro dimethyl terephthalate intermediate. This intermediate is then seamlessly carried forward into the subsequent oxidation stage without the need for isolation, streamlining the workflow and minimizing material handling losses.
Mechanistic Insights into Condensation and Oxidative Hydrolysis
The core of this synthesis lies in the precise orchestration of two distinct chemical transformations: acid-catalyzed condensation followed by alkaline oxidative hydrolysis. In the first stage, the protonation of the carbonyl groups in DMSS activates the molecule for nucleophilic attack by the amino group of p-chloroaniline. The use of catalysts such as sulfuric acid, hydrochloric acid, or acetic acid at concentrations ranging from 0.1% to 1.0% relative to DMSS ensures rapid kinetics. The reaction exotherm is carefully managed within the glycol dispersion, preventing thermal runaway while maintaining the system above the melting point of the reactants to ensure homogeneity. The resulting dihydro intermediate precipitates out of the solution due to its insolubility in the aqueous glycol mixture, driving the equilibrium forward according to Le Chatelier's principle and ensuring high conversion efficiency.
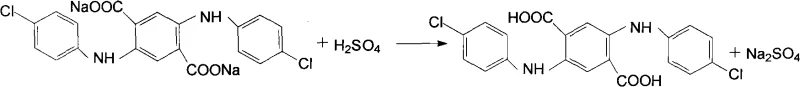
Following condensation, the slurry undergoes a simultaneous hydrolysis and oxidation reaction upon the addition of a mineral alkali and an oxidizing agent. As shown in the diagram, the ester groups are hydrolyzed to carboxylates while the central cyclohexadiene ring is aromatized to form the stable terephthalate structure. Oxidants such as m-nitrobenzene sulfonate, nitrobenzene, or p-nitrotoluene are employed in molar ratios of roughly 3:1 relative to DMSS to drive this aromatization to completion. The resulting disodium or dipotassium salt is highly soluble in the hot glycol-water phase, allowing for effective filtration of any unreacted solids which can be recycled. Subsequent acidification of the purified filtrate to a pH of 2 to 6 precipitates the final DpCTA product as a bright red crystalline powder, as depicted in the structural formula below.
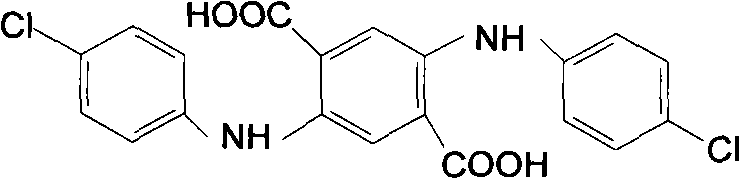
Impurity control is rigorously maintained through an adsorption step utilizing agents like activated carbon or diatomite prior to acidification. This step is critical for removing trace colored by-products and residual oxidants, ensuring the final pigment intermediate meets the stringent colorimetric standards required for high-grade automotive and industrial coatings. The ability to recycle the mother liquor further enhances the process economics, as the glycol and water phases can be recovered and reused, minimizing waste discharge and raw material costs.
How to Synthesize 2,5-Di(p-chloroanilino)-terephthalic Acid Efficiently
Implementing this synthesis route requires careful attention to temperature gradients and stoichiometric ratios to maximize the benefits of the glycol dispersion system. The process begins with the precise batching of DMSS and p-chloroaniline in a molar ratio of approximately 1:2.1, dispersed in ethylene glycol and water. Operators must ensure the dissolution phase reaches at least 110 ℃ to guarantee a single-phase mixture before catalyst addition. The subsequent condensation and oxidation steps are exothermic and require robust temperature control systems to maintain the optimal window of 100 ℃ to 140 ℃. Detailed standardized operating procedures regarding pressure limits and agitation speeds are essential for safe scale-up.
- Batch Mixing and Dissolution: Mix DMSS and p-chloroaniline with ethylene glycol and water, then heat to 110-140°C to form a homogeneous solution.
- Condensation Reaction: Add an acid catalyst (e.g., sulfuric acid) and maintain temperature at 100-140°C under pressure to form the dihydro intermediate precipitate.
- Hydrolysis and Oxidation: Treat the slurry with mineral alkali and an oxidant at 90-140°C to convert the intermediate into the soluble terephthalate salt.
- Purification and Acidification: Filter the solution, adsorb impurities with activated carbon, and acidify the filtrate to pH 2-6 to precipitate the final DpCTA product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this glycol-based synthesis route offers transformative benefits in terms of cost stability and operational reliability. By eliminating the reliance on volatile, low-boiling solvents like methanol, the process significantly reduces the capital expenditure associated with explosion-proof infrastructure and complex solvent recovery units. The ability to operate at atmospheric or near-atmospheric pressures with non-flammable glycol mixtures drastically lowers insurance premiums and safety compliance costs. Furthermore, the high solubility of intermediates in the reaction medium minimizes raw material loss, directly translating to improved atom economy and reduced cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The elimination of expensive solvent recovery distillation columns and the ability to recycle the ethylene glycol-water mother liquor lead to substantial operational savings. The high yield reported in the patent embodiments, consistently exceeding 98%, ensures that raw material utilization is maximized, reducing the frequency of replenishment orders for key starting materials like DMSS and p-chloroaniline. Additionally, the simplified workup procedure reduces labor hours and energy consumption per kilogram of product produced.
- Enhanced Supply Chain Reliability: The robustness of this chemical process against minor fluctuations in temperature and pressure makes it highly suitable for continuous or large-batch production, ensuring consistent delivery schedules for downstream pigment manufacturers. The use of commodity chemicals such as ethylene glycol and common mineral acids reduces dependency on specialized reagents, mitigating supply chain risks associated with raw material shortages. This stability allows for better inventory planning and long-term contracting strategies.
- Scalability and Environmental Compliance: The reduction in wastewater volume and the absence of toxic volatile organic compound (VOC) emissions align perfectly with increasingly strict global environmental regulations. The process generates significantly less hazardous waste compared to traditional methods, simplifying effluent treatment requirements and lowering disposal fees. This environmental advantage facilitates easier permitting for capacity expansion and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of DpCTA intermediates. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating this technology's fit within your supply chain.
Q: What are the primary advantages of using ethylene glycol over methanol in DpCTA synthesis?
A: Ethylene glycol has a significantly higher boiling point than methanol, allowing the condensation reaction to proceed at elevated temperatures (110-140°C) without requiring extreme pressure. This improves the solubility of DMSS, ensures a more thorough reaction, and eliminates the safety hazards associated with volatile organic solvents.
Q: How does this process handle impurity removal effectively?
A: The process incorporates a dedicated adsorption step using agents like activated carbon, diatomite, or molecular sieves on the purified mother liquor before acidification. This effectively removes colored impurities and by-products, resulting in a bright red pressed powder with high purity suitable for premium quinacridone pigments.
Q: Can the solvent system be recycled to reduce waste?
A: Yes, the ethylene glycol and water dispersion system is designed for recyclability. After solid-liquid separation during the acidification step, the mother liquor containing the glycol and water can be separated, purified, and returned to the reactor for the next batch, significantly reducing wastewater generation and raw material consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Di(p-chloroanilino)-terephthalic Acid Supplier
At NINGBO INNO PHARMCHEM, we understand that the consistency of pigment intermediates is paramount for the performance of the final colorant. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the laboratory-level efficiencies of the glycol dispersion method are faithfully reproduced at an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify every batch of DpCTA against critical parameters such as particle size, color strength, and chemical purity.
We invite you to collaborate with us to optimize your pigment supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can enhance your product quality while reducing overall procurement costs.
