Advanced Catalytic Hydrogenation Strategy for High-Purity UV-1130 Intermediate Manufacturing
Advanced Catalytic Hydrogenation Strategy for High-Purity UV-1130 Intermediate Manufacturing
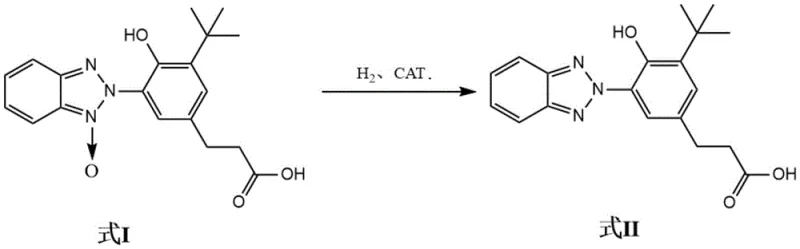
In the highly competitive landscape of specialty chemical manufacturing, the production of high-performance UV absorbers like UV-1130 demands precision, efficiency, and environmental compliance. A groundbreaking technical solution has emerged through patent CN114436981A, which details a superior preparation method for 3-[3-(2H-benzotriazol-2-yl)-5-tert-butyl-4-hydroxyphenyl]propionic acid, a critical precursor in the synthesis of advanced benzotriazole UV stabilizers. This innovation addresses long-standing challenges in the industry by replacing stoichiometric metal reductions with a sophisticated catalytic hydrogenation system. By leveraging a dual-catalyst approach involving Raney Nickel and Ruthenium Carbon, the process achieves yields exceeding 98.00% and purities greater than 99.00%, setting a new benchmark for quality in the supply chain of polymer additives and fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this vital UV absorber intermediate has been plagued by inefficient and environmentally hazardous methodologies. Prior art, such as Chinese patent CN201010567765.5, relied heavily on zinc powder reduction, a technique notorious for its low catalytic efficiency and the generation of massive quantities of solid metal waste that complicate disposal and increase environmental liability. Similarly, alternative routes utilizing aluminum powder, as seen in CN201310321244.5, suffer from poor atom economy, requiring multiple extraction steps that drive up solvent consumption and operational costs while still yielding suboptimal product recovery. Furthermore, literature reports utilizing Palladium on Carbon (Pd/C) catalysts have demonstrated a critical flaw: the excessive activity of palladium often leads to over-reduction, generating unwanted tetrahydro impurities that severely degrade the optical performance and purity of the final UV stabilizer, rendering the batch unsuitable for high-end applications.
The Novel Approach
The methodology disclosed in patent CN114436981A represents a paradigm shift towards sustainable and high-selectivity manufacturing. By employing a specific catalytic system under controlled hydrogen pressure, this novel approach circumvents the pitfalls of stoichiometric reductants. The process operates under mild conditions, typically between 25°C and 60°C, utilizing a synergistic catalyst blend that ensures the nitro group is reduced with pinpoint accuracy while leaving the sensitive benzotriazole ring intact. This selectivity is paramount for maintaining the structural integrity required for effective UV absorption. The result is a streamlined workflow that eliminates the need for tedious extractions, minimizes waste generation, and delivers a product with exceptional purity profiles, directly addressing the quality concerns of R&D directors and procurement managers alike.
Mechanistic Insights into Dual-Catalyst Hydrogenation Selectivity
The core of this technological breakthrough lies in the intricate interplay between the chosen catalysts and the reaction environment. The utilization of Raney Nickel, specifically formulated with promoting components like Chromium, Iron, and Copper, alongside Ruthenium Carbon, creates a unique electronic environment on the catalyst surface. This dual-catalyst system modulates the adsorption energy of the substrate, facilitating the cleavage of the N-O bonds in the nitro group while simultaneously suppressing the hydrogenation of the aromatic rings within the benzotriazole moiety. This mechanistic precision is what prevents the formation of the dreaded tetrahydro impurities observed with Pd/C catalysts. The presence of auxiliary agents, such as pyridine or sodium hydroxide, further fine-tunes the reaction medium's pH and polarity, stabilizing the transition states and ensuring that the reduction proceeds exclusively to the desired amine intermediate without side reactions.
Impurity control is rigorously managed through this selective mechanism. In conventional processes, the lack of selectivity leads to a complex impurity profile that requires extensive and costly purification steps, often involving recrystallization or column chromatography which reduce overall yield. In contrast, the catalytic hydrogenation method described herein inherently suppresses these side pathways. The high selectivity (>98%) means that the crude product emerging from the reactor is already of high purity, significantly reducing the burden on downstream purification units. For quality control teams, this translates to a much cleaner HPLC profile with fewer unknown peaks, ensuring that the final UV-1130 intermediate meets the stringent specifications required for use in sensitive polymer matrices and pharmaceutical formulations where trace impurities can have detrimental effects.
How to Synthesize 3-[3-(2H-benzotriazol-2-yl)-5-tert-butyl-4-hydroxyphenyl]propionic acid Efficiently
The operational protocol for this synthesis is designed for robustness and scalability, making it ideal for industrial adoption. The process begins with the precise charging of solvents, typically a mixture of xylene and methanol, along with the specific auxiliary agent and the substrate (Formula I) into a standard hydrogenation autoclave. The addition of the catalyst package follows, after which the system is sealed and subjected to a rigorous purge cycle to ensure an oxygen-free environment. The reaction is then driven by hydrogen pressure, maintained within the optimal range to balance reaction rate and safety. Upon completion, the workup is remarkably simple, involving filtration and pH adjustment, which allows for the direct isolation of the high-purity solid product. For detailed standard operating procedures and specific parameter optimization, please refer to the standardized synthesis guide below.
- Charge the hydrogenation autoclave with solvent (xylene/methanol), auxiliary agent (pyridine/NaOH), substrate (Formula I), and catalysts (Raney Ni/Ru-C).
- Seal the reactor, purge with nitrogen and hydrogen three times, and react at 25°C-60°C under 2-3MPa hydrogen pressure for 6-12 hours.
- Filter off the catalyst, remove solvent, wash with water, adjust pH to 5-6, and filter the solid to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic hydrogenation technology offers profound strategic advantages beyond mere technical superiority. The elimination of stoichiometric metal powders like zinc and aluminum removes a significant variable cost component and drastically reduces the logistical burden associated with handling and disposing of hazardous solid waste. This shift not only lowers the direct cost of goods sold (COGS) but also mitigates regulatory risks associated with heavy metal discharge, ensuring long-term operational continuity in regions with strict environmental enforcement. Furthermore, the simplified workup procedure, which avoids multiple solvent extractions, significantly shortens the batch cycle time, allowing manufacturing facilities to increase throughput and respond more agilely to market demand fluctuations without requiring additional capital investment in new equipment.
- Cost Reduction in Manufacturing: The economic impact of replacing stoichiometric reductants with reusable heterogeneous catalysts cannot be overstated. By eliminating the consumption of tons of zinc or aluminum powder per batch, the raw material costs are substantially reduced. Additionally, the ability to recover and reuse the solvent system after desolvation further drives down operational expenses. The high yield (>98%) ensures that raw material input is maximized into saleable product, minimizing waste loss and improving the overall margin structure for the production of these specialty chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of this new process. Traditional methods involving multiple extraction steps are prone to emulsion formation and phase separation issues, which can lead to batch failures and unpredictable delays. The new hydrogenation route relies on a straightforward filtration and precipitation workflow, which is highly reproducible and less susceptible to operator error. This reliability ensures consistent delivery schedules and reduces the risk of stockouts for downstream customers relying on a steady supply of high-purity UV absorber intermediates for their own production lines.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is seamless because it utilizes standard hydrogenation autoclaves found in most fine chemical plants. There is no need for specialized equipment required for handling large volumes of reactive metal powders. From an environmental perspective, the drastic reduction in solid waste and the minimization of solvent usage align perfectly with global sustainability goals. This 'green' manufacturing profile enhances the brand value of the supplier, making it a preferred partner for multinational corporations committed to reducing their Scope 3 emissions and adhering to strict ESG (Environmental, Social, and Governance) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of purity, yield, and operational safety. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this process for large-scale manufacturing of UV stabilizers and related fine chemicals.
Q: How does this new method prevent the formation of tetrahydro impurities?
A: Unlike traditional Palladium on Carbon (Pd/C) catalysts which are overly active and prone to over-reduction, the novel combination of Raney Nickel and Ruthenium Carbon offers superior selectivity. This specific catalytic system effectively reduces the nitro group without saturating the benzotriazole ring, thereby eliminating the formation of tetrahydro impurities that typically compromise product purity.
Q: What are the environmental advantages over zinc or aluminum powder reduction?
A: Conventional methods utilizing zinc or aluminum powder generate substantial amounts of solid metal waste and require complex extraction processes, leading to significant environmental pollution. The disclosed catalytic hydrogenation method utilizes reusable heterogeneous catalysts and produces minimal by-products, drastically simplifying the 'three wastes' treatment and aligning with modern green chemistry standards.
Q: Can the solvent system be recycled in this process?
A: Yes, the process is designed for efficiency. After the reaction is complete and the catalyst is filtered off, the reaction solution undergoes desolvation. The patent indicates that the solvent system can be recovered and reused, which not only lowers the raw material costs but also reduces the overall solvent consumption footprint of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-[3-(2H-benzotriazol-2-yl)-5-tert-butyl-4-hydroxyphenyl]propionic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing processes requires a partner with deep technical expertise and proven scale-up capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN114436981A are fully realized in a commercial setting. Our state-of-the-art facilities are equipped with high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-[3-(2H-benzotriazol-2-yl)-5-tert-butyl-4-hydroxyphenyl]propionic acid meets the highest international standards for polymer additives and specialty intermediates.
We invite you to collaborate with us to optimize your supply chain for UV absorber intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this catalytic hydrogenation route can improve your bottom line. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples to validate the superior quality of our commercially produced intermediates.
